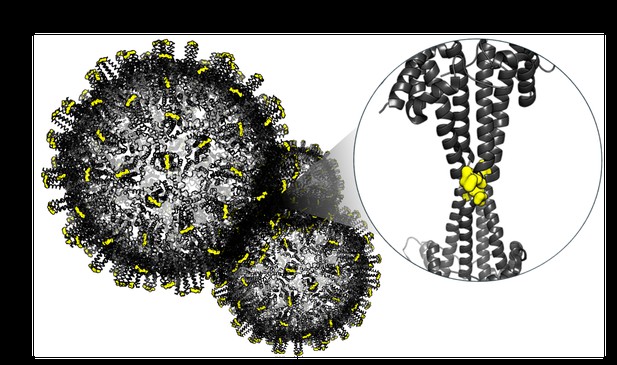
Peptides (yellow) binding to an unconventional site on the Hepatitis B virus capsid (grey), induce aggregation and offer a fresh strategy for antiviral therapy. Image credit: Hans Michael Maric (CC BY 4.0)
New and better strategies to treat hepatitis B are urgently needed. Many people worldwide remain unvaccinated against the disease, leaving them vulnerable to infection and serious liver problems. Children are particularly at risk of developing long-term illness. Treatments exist to help manage the condition, but they can rarely cure it.
Hepatitis B virus is protected by a spiky shell called the capsid, made of HBc proteins. This structure is critical for survival, and therefore a promising therapeutic target. Current approaches rely on compounds disrupting the assembly or stability of this structure by binding onto the HBc protein. So far, most of these drug candidates target the same location at the base of the capsid’s spikes. Until now, other binding pockets on the capsid remained largely unexplored.
To investigate whether these sites could be potential drug targets, Khayenko et al. developed two types of molecules, peptides and geranyl dimers, that could theoretically attach to the capsid – the former at the tip of the spikes and the latter in their middle section. In vitro and in living cells, the compounds not only latched onto these sites, but caused HBc proteins to clump together, preventing the capsid from forming properly. A similar mode of action is observed with existing drug candidates that, however, all bind to the pocket at the base of the spikes.
These findings highlight alternative strategies for targeting hepatitis B. Future studies will need to determine how well these molecules work in clinical conditions, and whether they could complement or improve existing treatments.




