Postprandial sleep mechanics in Drosophila
Figures
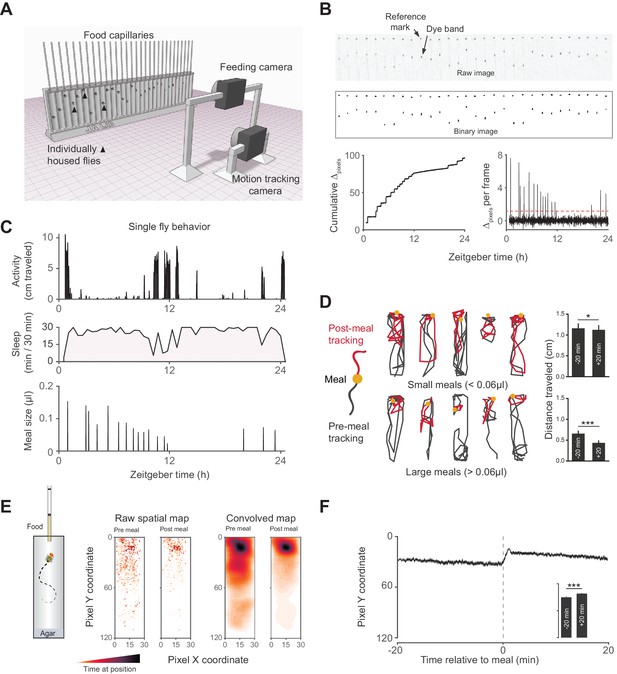
Activity Recording CAFE (ARC) facilitates simultaneous, high resolution measurements of food intake and motion in individual flies.
(A) Schematic of ARC apparatus. Independent computer-controlled cameras record images of capillaries for feeding measurements and of flies for activity and sleep determination. (B) Raw image (one out of 1440 taken over 24 hr) of capillaries containing liquid food (top). Capillaries have an external reference mark and an internal dyed oil band overlaid the aqueous food. Images are thresholded to binary, from which cumulative pixel distances between the reference mark and dye band can be calculated from the image series (bottom). Δpixels per frame reveals individual feeding events, selected as events greater than 3.5 standard deviations above noise (red dashed line). (C) Activity, sleep (black shading) and feeding (µl) of an individual fly (activity and feeding in 1 min bins, sleep in 30 min bins). (D) Example motion traces of individuals before (black) and after (red) a meal (yellow circle). Motion traces and averages associated with small (<0.06 µl) or large (≥0.06 µl) meals are shown. n = 661 meals from 30 flies, w1118; *p<0.05, ***p<0.001, Wilcoxon matched-pairs sign rank test. (E) Raw and convolved spatial heat map showing time spent at each pixel coordinate in the 20 min before and after meals (291 feeding events from 15 w1118 flies, Gaussian convolution). (F) Kymograph of average vertical position in time relative to meals (shaded line represents mean ± s.e.m.). The inset graph shows the average position over 20 min pre-meal (−20) and post-meal (+20) with the axis scaled to the parent graph. ***p<0.001, Wilcoxon matched-pairs sign rank test. All bars represent mean ± s.e.m.
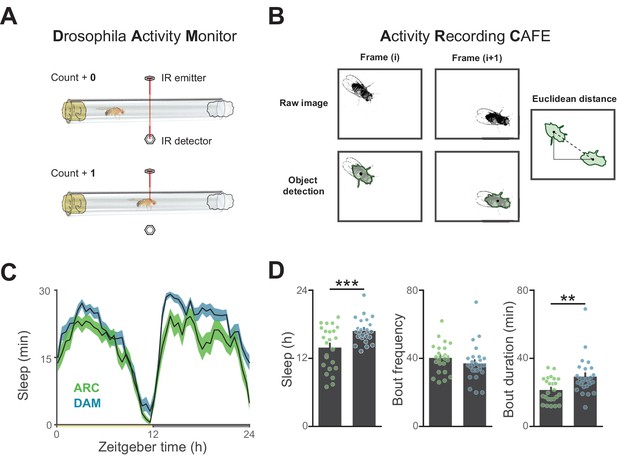
Influence of recording apparatus on sleep architecture.
(A) Illustration of motion estimation using the Drosophila Activity Monitor in which the number of times a fly breaks an infrared beam in a glass tube is counted. (B) Illustration of motion quantification by first detecting an object and determining the frame-frame distance of the object centroids. (C) A comparison of circadian sleep profiles derived from the Drosophila Activity Monitor versus object tracking reveals general conservation of sleep patterning between systems. (D) Measurement of total sleep and sleep duration are lower using video tracking, indicating a higher resolution of waking motion. 25–27 flies per condition, **p<0.01, ***p<0.001, Student’s t-test.
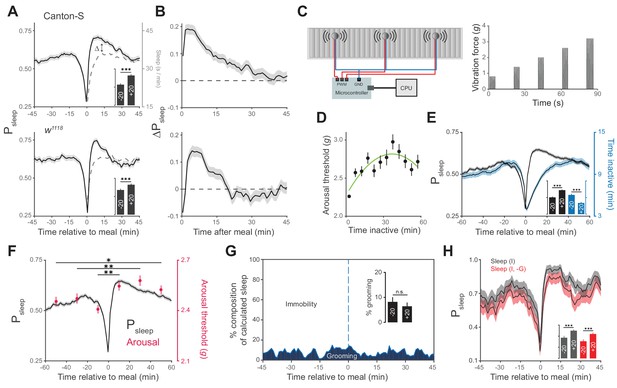
Animals exhibit increased sleep and arousal threshold after eating.
(A) Probability of sleep (Psleep) preceding or following each meal (t = 0) in Canton-S (top) or w1118 (bottom) males. Data are shown as averages of 1 min bins. ΔPsleep is defined as the difference between postprandial Psleep and the corresponding time-matched pre-meal Psleep (i.e. t1 – t−1, t2 – t−2, …). For clarity, a mirror image of pre-meal Psleep is replotted in the postprandial period (dashed line). Inset graphs show average Psleep (± s.e.m.) for the 20 min before (−20) and after (+20) meals, with the axis scaled to the parent graph. n = 757 meals from 50 flies, Canton-S; 661 meals from 30 flies, w1118; ***p<0.001, Wilcoxon matched-pairs sign rank test. (B) ΔPsleep calculated from data in A. (C) Diagram of stimulus delivery system showing shaft-less vibration motors attached to the back of ARC chamber. Increasing vibrations are delivered to the chamber via a microcontroller using pulse width modulation. (D) Arousal threshold shows an initial increase with the time an animal is inactive. 180 flies, 11,479 arousal events, Canton-S; 5 min bins, circles represent mean ± s.e.m., a secondorder polynomial trendline is shown. (E) Superimposition of time inactive (blue) over Psleep (black) relative to meals during periodic vibrational stimuli. The inset graph shows Psleep and time inactive in the 20 min before and after each meal. n = 2245 meals from 180 flies, Canton-S; ***p<0.001, Wilcoxon matched-pairs sign rank test. (F) Stimulus response from 0–20, 20–40, and 40–60 min pre- and post-meal (red) superimposed onto Psleep (black). Arousal events are filtered to 5 min intervals for prior time inactive to control for sleep depth (0–5 mins shown, minimum 1 s inactivity). Circles represent mean ± s.e.m.; n = 2245 meals from 180 flies, Canton-S; *p<0.05, **p<0.01, Mann Whitney test. (G) Percent of calculated sleep that is actual immobility versus grooming. The inset graph shows the percent grooming in the 20 min before and after each meal. n = 55 meals from seven flies, Canton-S; p=0.69, Wilcoxon matched-pairs sign rank test. (H) Comparison of Psleep before and after meals calculated using immobility criteria "I" versus immobility criteria paired with grooming criteria "I, -G". Inset shows average Psleep (± s.e.m.) in the 20 min before and after each meal. ***p<0.001, Wilcoxon matched-pairs sign rank test.
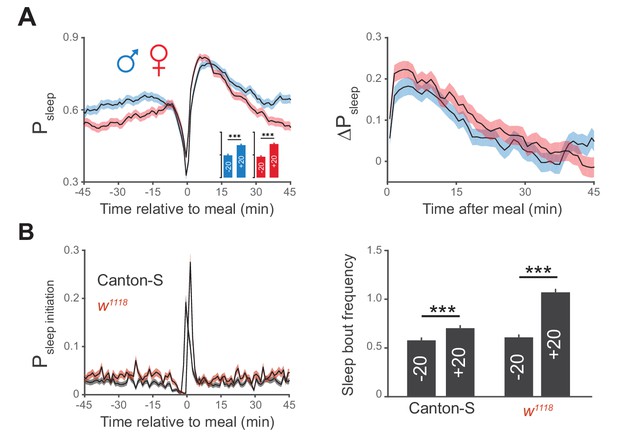
Postprandial sleep is sex independent.
(A) Canton-S males (blue) and females (red) demonstrate similar increases in postprandial sleep. n = 672 meals from 30 males; 730 meals from 30 females; ***p<0.001, Wilcoxon matched-pairs sign rank test. (B) Probability of sleep initiation in 1 min windows surrounding feeding events for Canton-S (gray) and w1118 (red), with a quantification of sleep bout frequency in the 20 min before (+20) and after (−20) feeding events. ***p<0.001, Wilcoxon matched-pairs sign rank test.
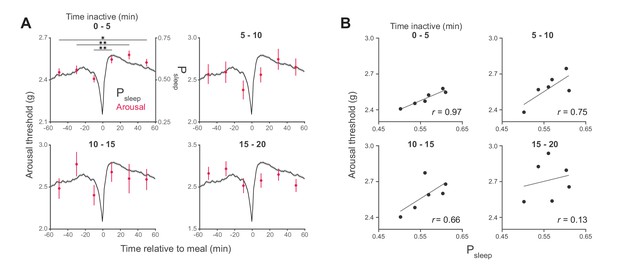
Arousal threshold surrounding meals with increasing sleep duration state.
(A) Stimulus response from 0–20, 20–40, and 40–60 min pre- and post-meal (red) superimposed onto Psleep (black) representing the entire data set. Arousal events are filtered to 5-min intervals for prior time inactive (0–5 min duplicated from Figure 2) to control for sleep depth. (B) Correlation between Psleep and arousal threshold for 0–20, 20–40, and 40–60 min bins surrounding feeding events (Pearson correlation, six time-bins derived from 2245 meals from 180 flies, Canton-S).

Grooming event influence on immobility based sleep.
Ethograms of individual fly behavior in the 20 min before and after each meal, including immobility (purple), sleep (grey), grooming (blue), and recalculated sleep (red). n = 55 meals from seven flies, Canton-S.
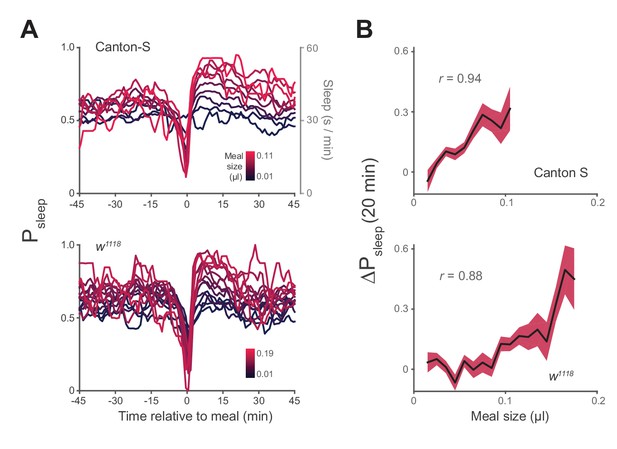
Postprandial sleep correlates with food intake quantity.
(A) Sleep probability of Canton-S and w1118 surrounding meals of varying size (lines represent mean; color grading corresponds to meal size). Only groupings with n > 7 are shown for visualization. (B) 20 min ΔPsleep as a function of meal size for each grouping (Pearson correlation: p<0.001 for Canton-S and w1118.) Shaded lines represent mean ± s.e.m.
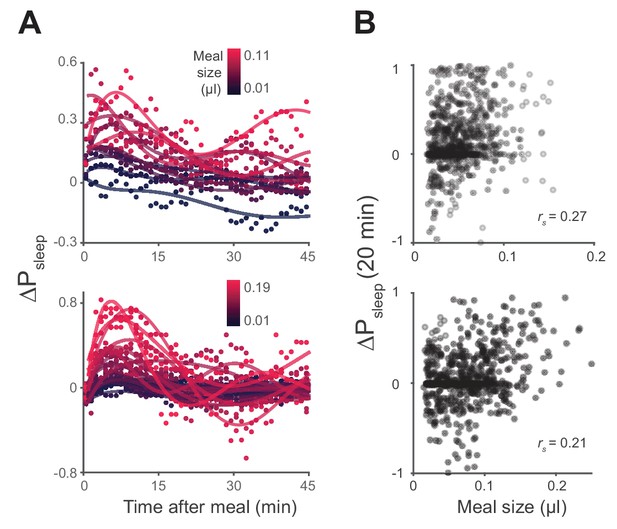
Sleep probability and ΔPsleep response to meal size.
(A) Time-course of ΔPsleep for meals grouped by volume (0.01 µl meal groupings, circles represent 1 min binned averages, lines represent spline fit). (B) Correlation between meal size and ΔPsleep in 20 min after feeding events for Canton-S and w1118 (Spearman rank-order correlation: p=9.0 × 10−14, Canton-S; p=4.1 × 10−8, w1118).
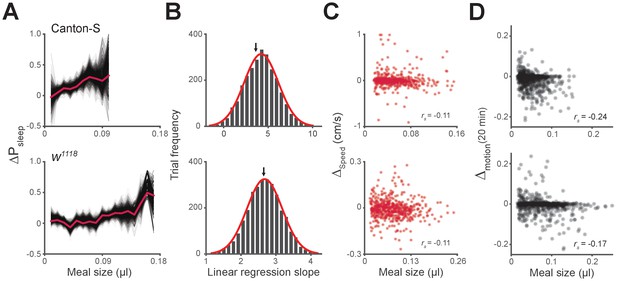
Unequal meal sampling frequency and motor ability effects on meal size-sleep correlation.
(A) Monte Carlo simulation of ΔPsleep as a function of meal size using fixed sampling frequency from individual flies. Simulations contained 3000 trials of randomly sampled meals, where thin black lines represent individual trials, and the red line represents the entire data set (three samples/fly, Canton-S; eight samples/fly, w1118; 0.01 µl meal groupings). (B) Histogram of linear regression slope for each trial fit with a Gaussian distribution (red line). The black arrow indicates the slope value of the entire data set. (C) Average Δspeed for the 20 min relative to meals versus meal size (Spearman rank-order correlation: p=0.0096, Canton-S; p=0.0095, w1118).
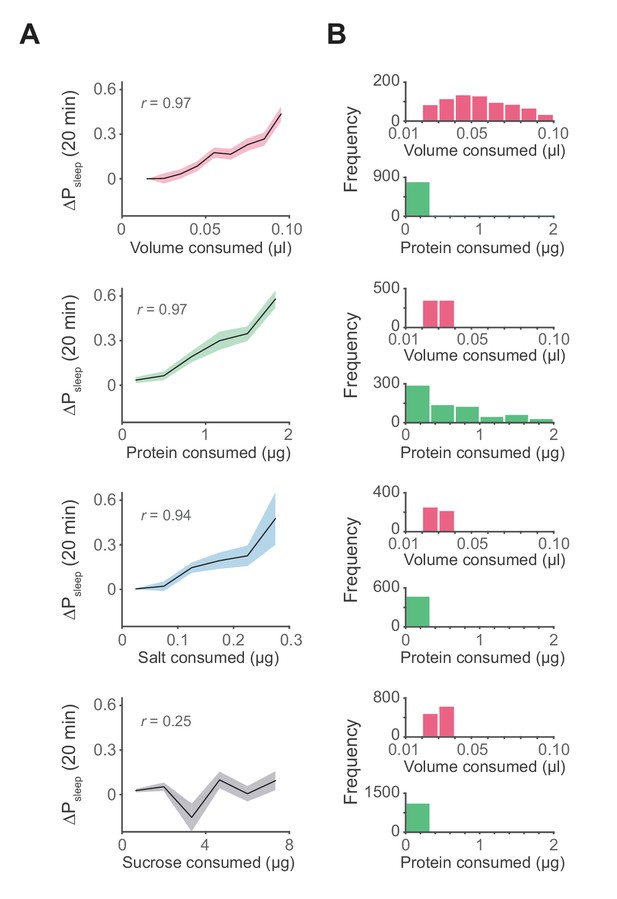
Influence of meal components on postprandial sleep.
(A) Average 20 min ΔPsleep as a function of each meal component (Canton-S, Pearson correlation: p<0.001, volume; p<0.001, protein; p<0.005, salt; p=0.065, sucrose). Shaded lines represent mean ± s.e.m. (B) Histograms representing the distribution of volume and protein consumed in each meal for data from A.
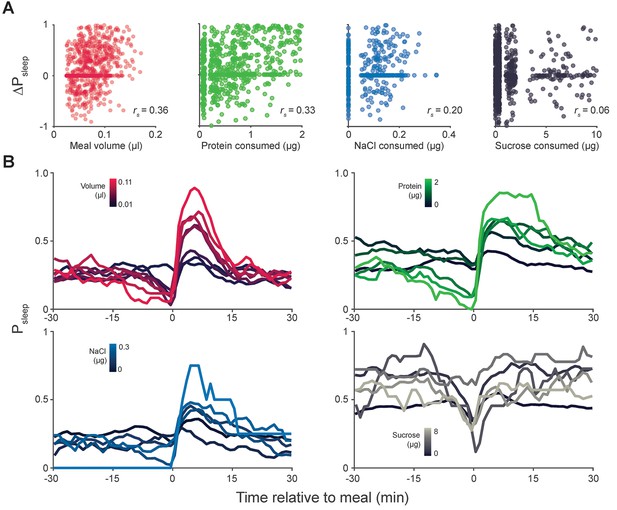
Meal component correlates to postprandial sleep.
(A) Scatter plots representing individual meals showing 20 min ΔPsleep versus meal volume or consumed protein, salt (NaCl), or sucrose. Non-normal data in each plot can arise from the tendency for fully awake animals to stay awake (ΔPsleep = 0) and from low variation in nutrient consumption within animals given a low nutrient, fixed diet (protein, NaCl, or sucrose consumption = 0 µg). Spearman rank-order correlation: p=1.52 × 10−25, volume; p=1.73 × 10−19, protein; p=6.84 × 10−6, NaCl; p=0.055, sucrose). (B) Time-courses of sleep probability for component groupings (lines represent mean for each grouping).
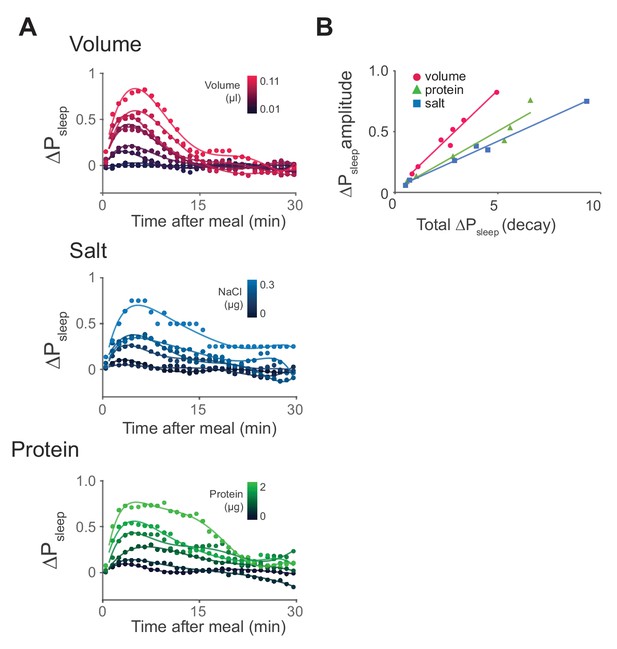
Time-course analysis of sleep in response to meal components reveals differential kinetics.
(A) Time-course analysis of ΔPsleep for meals of graded volume, protein, or salt consumption (circles represent 1 min binned averages, lines represent spline fit). n = 811 meals, volume; 714, protein; 478, salt; 1131, sucrose. (B) ΔPsleep amplitude for gradings of volume, protein, and salt plotted against total ΔPsleep during the decay phase 20 min following the maximum.
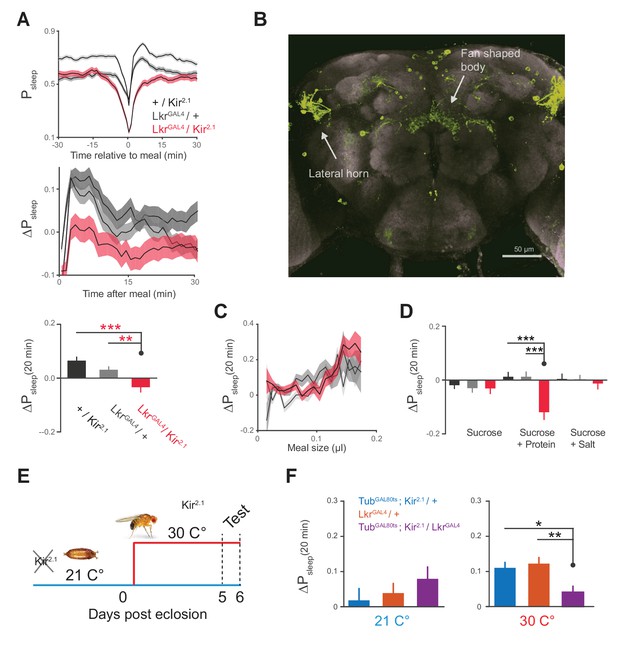
Leucokinin receptor neurons regulate protein-induced postprandial sleep.
(A) Overexpression of Kir2.1 channel in cells labeled by LkrGAL4 (R65C07) results in a defective postprandial sleep response (20 flies per genotype, n = 697 meals, Kir2.1 / wCS; 762, LkrGAL4 / wCS; 450, Kir2.1 / LkrGAL4; *p<0.05; ***p<0.001, Kruskal-Wallis test followed by Dunn’s multiple comparisons). (B) LkrGAL4 drives mCD8-GFP expression in neurons innervating the dorsal fan-shaped body, stemming from cell bodies in the lateral horn (scale bar = 50 µm). (C) Average 20 min ΔPsleep for Kir2.1-silenced LkrGAL4 animals and controls given low nutrient food (1% sucrose, 0.25% tryptone) for observing volumetric effects (n = 1589–1736 meals per genotype). (D) Average 20 min ΔPsleep for Kir2.1-silenced LkrGAL4 animals and controls given salt (2.5% sucrose + 1% salt) or protein (2.5% sucrose + 1.7% tryptone) diet to test meal component influences on postprandial sleep. The silenced line shows a negative response to protein supplemented diet (***p<0.001, Kruskal-Wallis test followed by Dunn’s multiple comparisons). n = 257–444 meals per genotype. (E) Expression of Kir2.1 can be restricted by using temperature-sensitive GAL80ts to suppress GAL4 activity at 21°C throughout development. (F) Silencing of LkrGAL4-labeled cells in adulthood (30°C) is sufficient to reduce postprandial sleep (24 flies per genotype, n = 493 meals, TubGAL80ts; Kir2.1/ wCS; 499, LkrGAL4/ wCS; 528, TubGAL80ts; Kir2.1/ LkrGAL4; *p<0.05; **p<0.01, Kruskal-Wallis test followed by Dunn’s multiple comparisons).
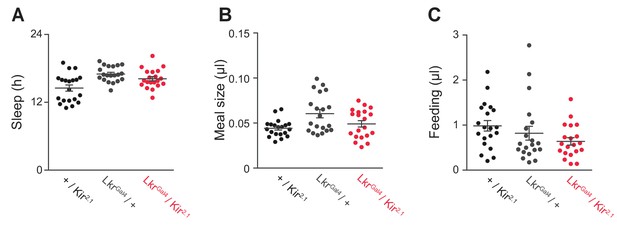
General behavior in Lkr neuronal-silenced animals.
(A) Total sleep, (B) meal size, and (C) feeding of LkrGAL4 / Kir2.1 and controls on sucrose + protein diet (Student’s t-test or one-way ANOVA followed by Tukey-Kramer post hoc test for multiple comparisons). Points represent individual animals and lines represent mean ± s.e.m.
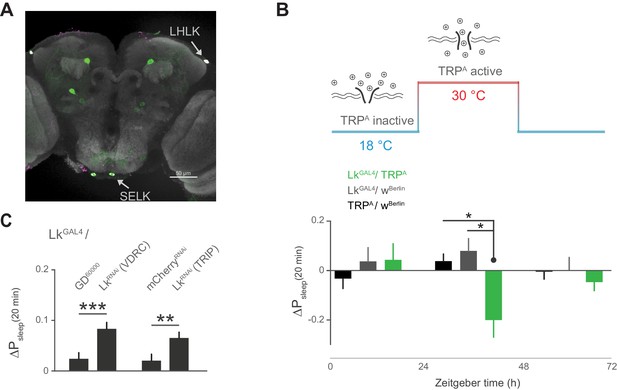
Leucokinin neurons inhibit postprandial sleep.
(A) Confocal reconstruction of immunostaining for anti-Lk (magenta) in the brain of LkGAL4>mCD8::GFP reveals Lk co-localization with GFP-expressing neurons (green) in the lateral horn (LHLK) and suboesophageal ganglion (SELK). The neuropil marker nc82 (gray) is used as background (scale bar = 50 µm). (B) Stimulation of Lk neurons at 30°C by expressing Transient receptor potential channel, TrpA, causes a reduction in postprandial sleep in comparison to the unstimulated state at 18°C (10 flies per genotype, 30°C, n = 83 meals, TrpA/ wBerlin; 63, LkGAL4 / wBerlin; 25, LkGAL4 / TrpA; *p<0.05, Kruskal-Wallis test followed by Dunn’s multiple comparisons). (C) Downregulation of Lk in LkGAL4-labeled cells, using two independent RNAi lines, increases postprandial sleep. (60 flies per genotype, n = 893 meals, LkGAL4 / GD60000; 841, LkGAL4 / LkRNAi (VDRC); 953, LkGAL4 /mCherryRNAi; 1060, LkGAL4 / LkRNAi (TRIP); **p<0.01, ***p<0.001 Mann-Whitney test).
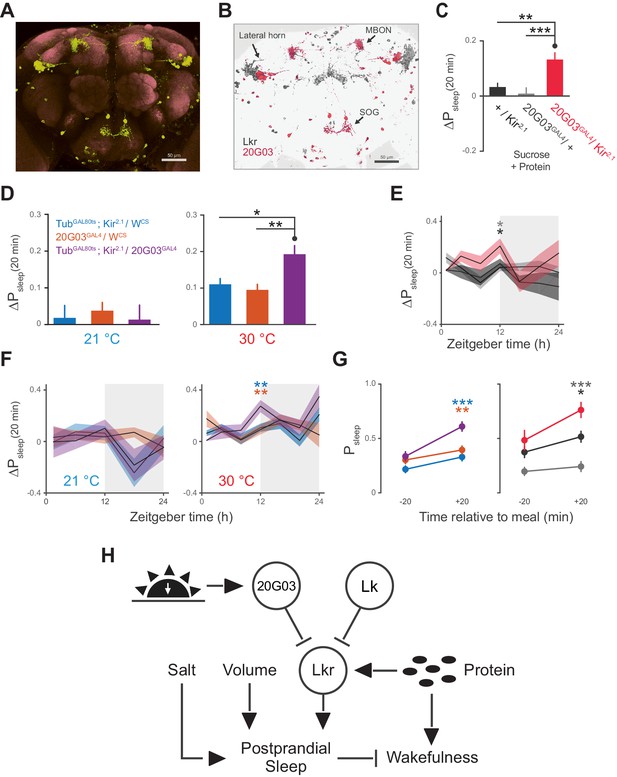
20G03 neurons inhibit postprandial sleep in a circadian manner.
(A) 20G03GAL4 drives mCD8-GFP expression in neurons with cell bodies positioned proximal to cell arborizations of those labeled by LkrGAL4. (B) Overlaid images of 20G03GAL4 and LkrGAL4 expression show close proximity between cell types in the lateral horn and SOG. (C) Overexpression of Kir2.1 channel in 20G03GAL4-labeled cells results in an enhanced postprandial sleep response (30 flies per genotype, n = 805 meals, Kir2.1 / w1118; 488, 20G03GAL4 / w1118; 285, Kir2.1 / 20G03GAL4; **p<0.01; ***p<0.001, Kruskal-Wallis test followed by Dunn’s multiple comparisons). (D) Conditional silencing of 20G03GAL4-labeled cells in adulthood is sufficient to increase postprandial sleep (24 flies per genotype, n = 493 meals, TubGAL80ts; Kir2.1/ wCS; 575, 20G03GAL4 / wCS; 528, TubGAL80ts; Kir2.1/20G03GAL41; *p<0.05; **p<0.01, Kruskal-Wallis test followed by Dunn’s multiple comparisons). (E) Time-course of postprandial sleep reveals that effect of 20G03GAL4 silencing is most prominent from ZT 10–14 (time-course partitioned into 4 hr bins, shaded region indicates dark period; *p<0.05, Kruskal-Wallis test followed by Dunn’s multiple comparisons). (F) Conditional silencing effects are also stronger at dusk ZT 10–14 (**p<0.01, Kruskal-Wallis test followed by Dunn’s multiple comparisons). (G) Comparison of Psleep in the 20 min before and after each meal for both 20G03GAL4 manipulations (color codes from C and D; *p<0.05; **p<0.01, ***p<0.001, Kruskal-Wallis test followed by Dunn’s multiple comparisons). (H) Proposed model for regulation of postprandial sleep by dietary components and neuronal circuitry. Meal volume, ingested salt, and protein drive postprandial sleep. Sleep induced by ingested protein acts through Lkr neurons. Protein also induces a waking response independent of Lkr neuronal activity. Leucokininergic (Lk) or non-leucokininergic (20G03) cell populations can independently inhibit postprandial sleep, possibly through modulation of Lkr neuronal activity.
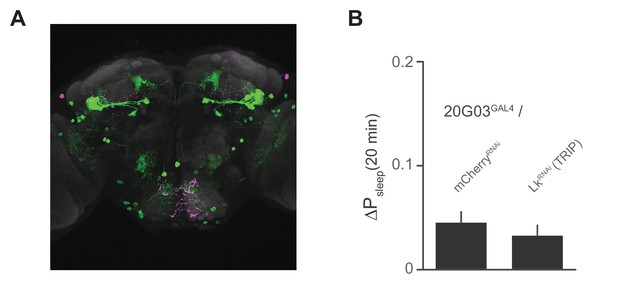
Lk immunostaining and knockdown in 20G03 neurons.
(A) Confocal reconstruction of 20G03GAL4>mCD8::GFP. GFP-expressing neurons (green) with immunostaining for anti-Lk (magenta) and neuropil marker nc82 (gray), shows no co-localization of Lk in 20G03GAL4-labeled neurons. (B) RNAi of Lk in 20G03GAL4 neurons does not affect postprandial sleep (60 flies per genotype; n = 1118 meals, 20G03GAL4 / mCherryRNA; 1249, 20G03GAL4 / LkRNAi [TRIP]).
Additional files
-
Supplementary file 1
Meal selection algorithm.
Flowchart depicting the algorithm used for processing dye-reference mark pixel distance data to identify feeding events.
- https://doi.org/10.7554/eLife.19334.020





