Suppressed prefrontal neuronal firing variability and impaired social representation in IRSp53-mutant mice
Figures
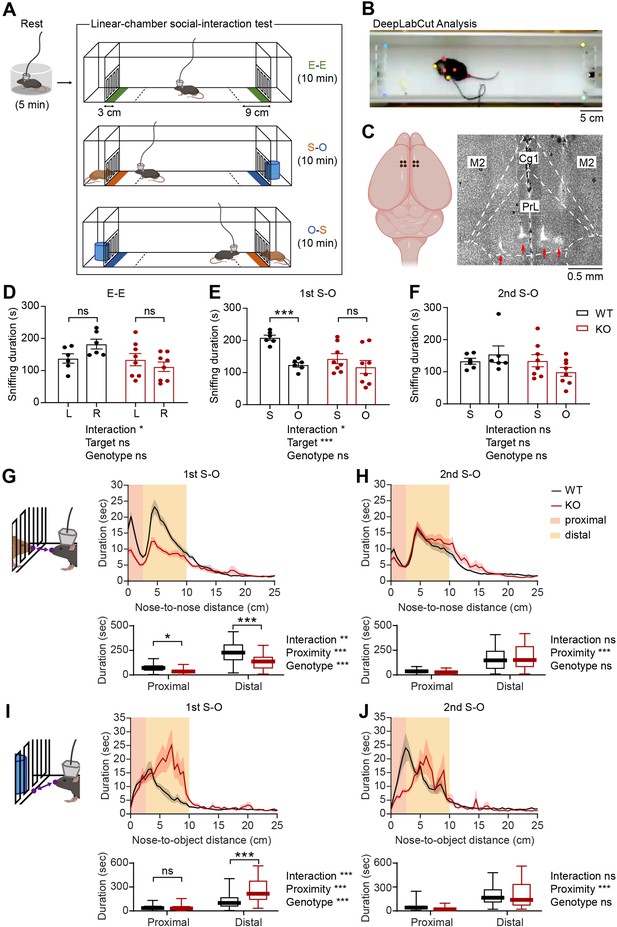
Social impairments in IRSp53-KO mice in the linear-chamber social-interaction test.
(A) Schematic diagram of the linear-chamber social-interaction test used to measure social approach towards a novel conspecific mouse (S, social) versus a novel non-social target (O, object). A tetrode-implanted mouse was first placed in the rest box for 5 min and moved to the linear social-interaction chamber to perform the following three sessions: empty-empty (E–E) session, social-object (first S-O) session, and object-social (second S-O) session. The in-zone area, falling within 9 cm from the front face of the chambers, is indicated by a dashed line. The sniffing zone, falling within 3 cm from the front face of the chambers, is indicated by green, orange, and blue colors. (B) An example video frame of subject mouse and social target mouse body parts automatically tracked by the DeepLabCut program. (C) Schematic (left) and a representative coronal brain section (right) showing the locations of the implanted tetrodes. PrL, prelimbic cortex; IL, infralimbic cortex; Cg1, cingulate cortex, area 1; M2, secondary motor cortex. (D–F) Mean (± standard error of mean/SEM) sniffing duration for left (L) vs. right (R) empty targets during the E-E session (D) and the social (S) vs. object (O) targets during the first S-O (E) and second S-O (F) sessions. (n=6 mice [WT], 8 mice [IRSp53-KO], *P<0.05, ***P<0.001, ns, not significant, two-way repeated-measures (RM)-ANOVA with Sidak’s multiple comparisons test). (G and H) Mean (± SEM) duration of the interaction between subject and social target mice as a function of the distance between the noses of the two mice during the first (G) and second (H) S-O sessions (top). Cumulative duration of proximal and distal social interactions (bottom). (n=47 experiments from 6 mice [WT], 58, 8 [IRSp53-KO], *p<0.05, **p<0.01, ***p<0.001, ns, not significant, two-way RM-ANOVA with Sidak’s multiple comparison test). (I and J) Mean (± SEM) duration of the interaction between subject mice and object target as a function of the distance between the subject mouse’s nose and the center of the object chamber face during the first (I) and second (J) S-O session (top). Cumulative duration of proximal and distal object interactions (bottom). (n=47 experiments from 6 mice [WT], 58, 8 [IRSp53-KO], ***p<0.001, ns, not significant, two-way RM-ANOVA with Sidak’s multiple comparison test). See Supplementary file 2 for statistics. Numerical data used to generate the figure are available in the Figure 1—source data 1.
-
Figure 1—source data 1
Source file for mouse behavior data in Figure 1.
The excel file contains the numerical data used to generate Figure 1D–J.
- https://cdn.elifesciences.org/articles/74998/elife-74998-fig1-data1-v2.xlsx
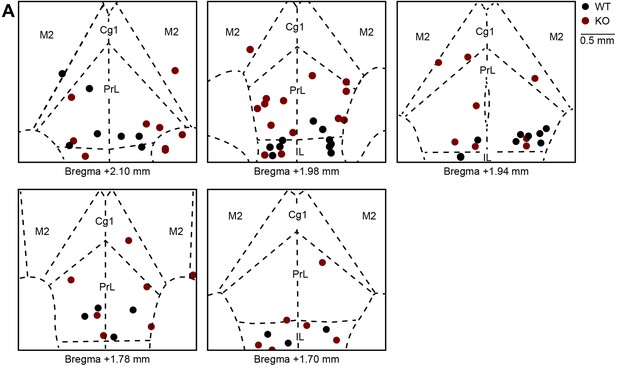
Locations of implanted tetrodes in the mPFC of WT and IRSp53-KO mice.
(A) Final location of tetrodes implanted into the mPFC of WT (black) and IRSp53-KO (red) mice. Coronal sections of the mPFC shown in this figure represent +1.70 –+2.10 mm away from the bregma. PrL, prelimbic cortex; IL, infralimbic cortex; Cg1, cingulate cortex, area 1; M2; secondary motor cortex.
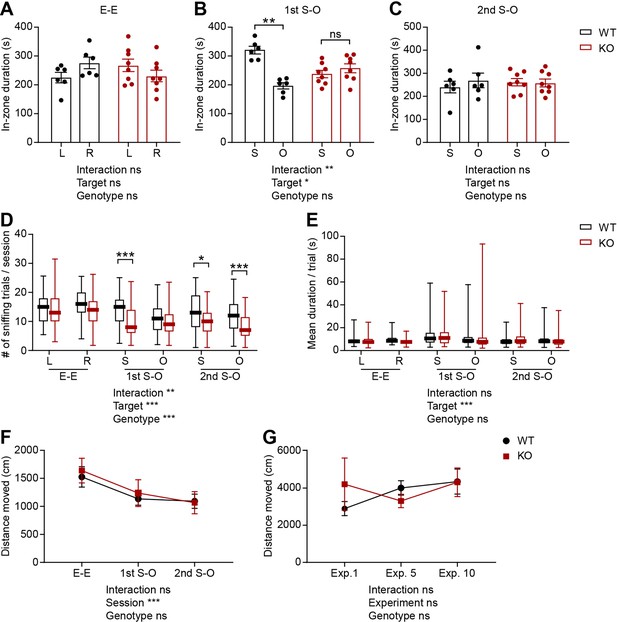
Social impairments, as judged by in-zone durations, and unaltered mean duration of each visit or locomotor activity in IRSp53-KO mice in the linear social-interaction chamber.
(A–C) Mean (± SEM) in-zone duration for left (L) vs. right (R) empty targets during the E-E session and social (S) vs. object (O) targets during the first and second S-O sessions. (n=6 mice [WT], 8 mice [IRSp53-KO], *p<0.05, **p<0.01, ns, not significant, two-way RM-ANOVA with Sidak’s multiple comparison test). (D and E) The mean number of sniffing visits (D) and mean duration of time spent sniffing per valid sniffing trial (E) for each target during the E-E, first S-O, and second S-O sessions. (n=57 experiments from 6 mice [WT], 69, 8 [IRSp53-KO], *p<0.05, **p<0.01, ***p<0.001, ns, not significant, two-way RM-ANOVA with Sidak’s multiple comparison test). (F and G) The mean (± SEM) distance moved in the linear social-interaction test across three consecutive sessions (E-E, first S-O, and second S-O) in an experiment (F) and across different recording experiments (1st, 5th, and 10th experiments used as examples; G). (n=6 mice [WT], 8 mice [IRSp53-KO], ***p<0.001, ns, not significant, two-way RM-ANOVA with Sidak’s multiple comparison test). See Supplementary file 2 for statistics. Numerical data used to generate the figure are available in the Figure 1—figure supplement 2—source data 1.
-
Figure 1—figure supplement 2—source data 1
Source file for mouse behavior data in Figure 1—figure supplement 2.
The excel file contains the numerical data used to generate Figure 1—figure supplement 2A–G.
- https://cdn.elifesciences.org/articles/74998/elife-74998-fig1-figsupp2-data1-v2.xlsx
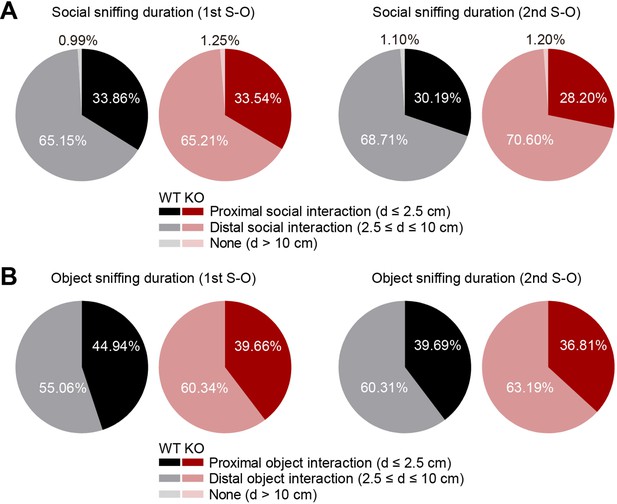
Comparable composition of behavior in social and object target sniffing zones in WT and IRSp53-KO mice in the linear social-interaction chamber.
(A) Mean composition of behavior in social target sniffing zones according to proximity (d, distance) between the noses of the subject mouse and the social target mouse during the first and second S-O sessions (n=47 experiments from 6 mice [WT], 58, 8 [IRSp53-KO]). (B) Mean composition of behavior in object target sniffing zones according to proximity between the nose of the subject mouse and the center of the object chamber face during the first and second S-O sessions (n=47 experiments from 6 mice [WT], 58, 8 [IRSp53-KO]). See Supplementary file 2 for statistics. Numerical data used to generate the figure are available in the Figure 1—figure supplement 3—source data 1.
-
Figure 1—figure supplement 3—source data 1
Source file for mouse behavior data in Figure 1—figure supplement 3.
The excel file contains the numerical data used to generate Figure 1—figure supplement 3A and B.
- https://cdn.elifesciences.org/articles/74998/elife-74998-fig1-figsupp3-data1-v2.xlsx
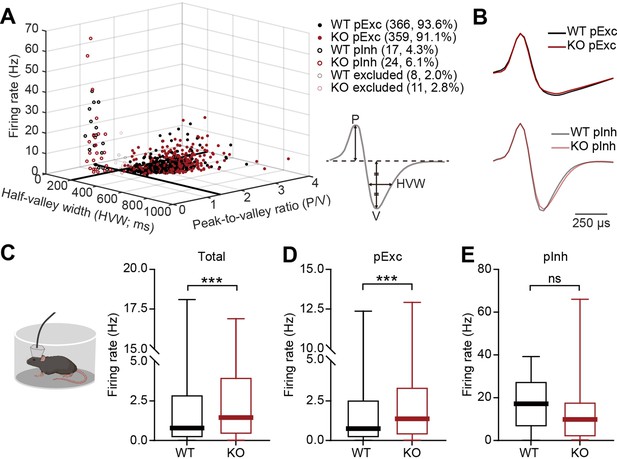
Increased resting firing rate in IRSp53-KO pExc mPFC neurons.
(A) Classification of recorded neurons into putative excitatory (pExc) and putative inhibitory (pInh) neurons based on the half-valley width (200ms) and peak-to-valley ratio (1.4). P, peak; V, valley; HVW, half-valley width. (B) Average waveforms of WT and IRSp53-KO pExc (top) and pInh (bottom) neurons (n=366 [WT-pExc], 359 [KO-pExc], 17 [WT-pInh], 24 [KO-pInh]). The waveforms of each neuron were normalized by their peak values. (C–E) Firing rates of WT and IRSp53-KO total (C), pExc (D), and pInh (E) neurons in the mPFC during the 5-min rest period. (n=391 [WT-total], 394 [KO-total], 366 [WT-pExc], 359 [KO-pExc], 17 [WT-pInh], 24 [KO-pInh], ***p<0.001, ns, not significant, Mann-Whitney test). See Supplementary file 2 for statistics. Numerical data used to generate the figure are available in the Figure 2—source data 1.
-
Figure 2—source data 1
Source file for resting firing rate data in Figure 2.
The excel file contains the numerical data used to generate Figure 2A–E.
- https://cdn.elifesciences.org/articles/74998/elife-74998-fig2-data1-v2.xlsx
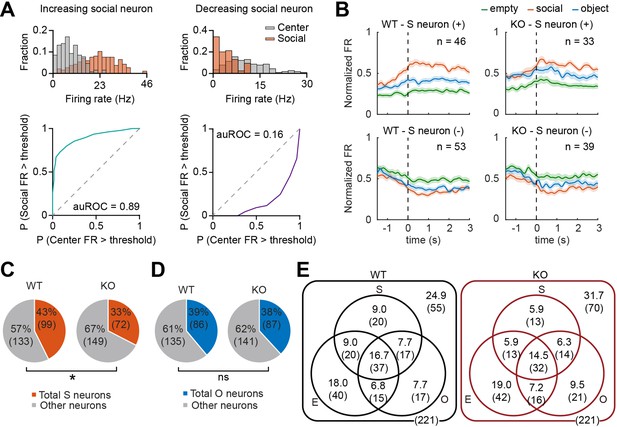
Fewer social pExc mPFC neurons in IRSp53-KO mice.
(A) Distributions of instantaneous firing rates (FR) during social sniffing and center zone (top) and the receiver operating characteristic curves (ROCs; bottom) of increasing (left) and decreasing (right) social neuron examples. (B) Average spike density functions (SDFs) of firing rate responses to empty (green), social (orange), and object (blue) targets (aligned to the onset of sniffing) for all social (S) neurons. Social neurons are divided by genotype (WT left, IRSp53-KO right) and response direction (increasing (+) top, decreasing (-) bottom). Total numbers of neurons are indicated at the upper right corner of each SDF. Shading indicates ± SEM. (C and D) Proportions of total social (C) and object (D) neurons (both increasing and decreasing neurons) out of the total recorded neurons. (*p<0.05, ns, not significant, Fisher’s exact test). (E) Venn diagram summary of empty (E), social (S), and object (O) neuronal proportions for WT (left) and IRSp53-KO (right) pExc neurons. Numbers indicate neuronal proportion % (n neurons). See Supplementary file 2 for statistics.
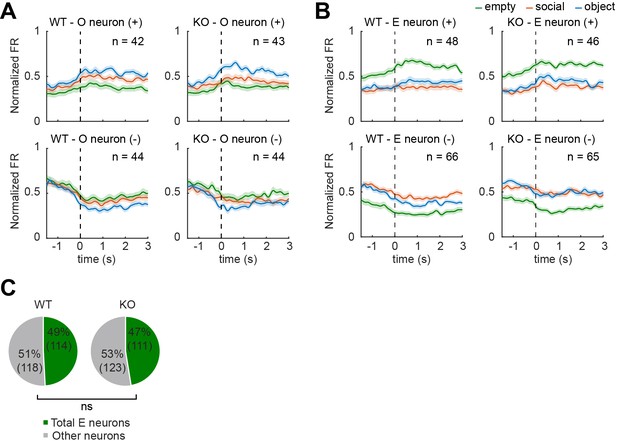
Normal number of empty pExc mPFC neurons in IRSp53-KO mice.
(A and B) Average spike density functions (SDFs) of firing rate responses to object empty (green), social (orange), and object (blue) targets (aligned to the onset of sniffing) for all object (O; A) and empty (E; B) neurons. Target neurons are divided by genotype (WT left, IRSp53-KO right) and response direction (increasing (+) top, decreasing (-) bottom). Total numbers of neurons are indicated at the upper right corner of each SDF. Shading indicates ± SEM. (C) Proportion of total empty neurons out of the total recorded neurons. (ns, not significant, Fisher’s exact test).
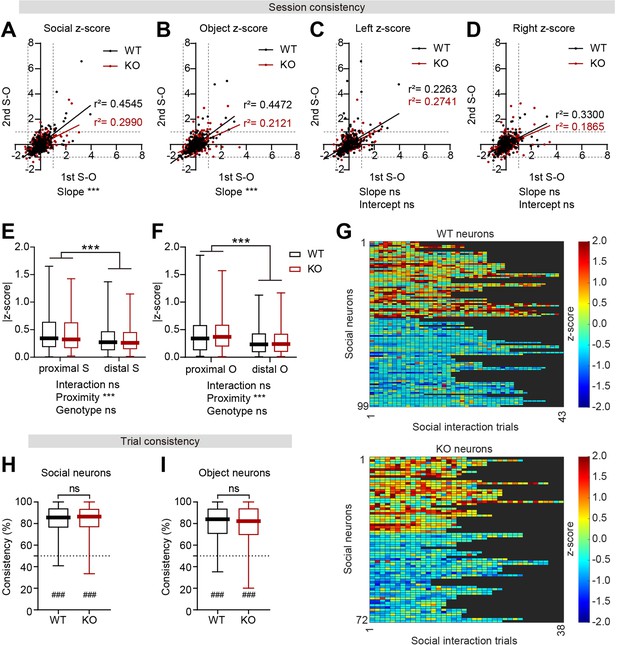
Robust target-dependent responses of WT and IRSp53-KO pExc mPFC neurons.
(A–D) Scatterplot of firing responses (in z-score) to social (A) and object (B) targets and left (C) and right (D) sidedness during the first S-O session against those during the second S-O session. Solid lines indicate simple linear regressions for WT (black) and KO (red) neurons. (n=233 [WT-pExc] and 258 [KO-pExc]), ***p<0.001, ns, not significant, simple linear regression with slope comparison test (see Methods). (E and F) Firing rate response magnitude (in |z-score|) to social (E) and object (F) targets according to proximity. (n=198 [WT-pExc] and 214 [KO-pExc], ***p<0.001, ns, not significant, two-way RM-ANOVA). (G) Heatmap showing trial-by-trial activity (in z-score) of WT (top) and IRSp53-KO (bottom) increasing and decreasing social neurons during proximal social target interactions. (n=53 [WT-pExc] and 32 [KO-pExc]). (H and I) Trial consistency (%) during social (H) and object (I) interaction trials in social and object neurons, respectively. (n=99, 86 [social, object neurons WT-pExc] and 72, 83 [social, object neurons KO-pExc], ns, not significant, Mann-Whitney test, ###p<0.001 (above chance level), One sample t-test). See Supplementary file 2 for statistics. Numerical data used to generate the figure are available in the Figure 3—figure supplement 2—source data 1.
-
Figure 3—figure supplement 2—source data 1
Source file for target neuron data in Figure 3—figure supplement 2.
The excel file contains the numerical data used to generate Figure 3—figure supplement 2A–I.
- https://cdn.elifesciences.org/articles/74998/elife-74998-fig3-figsupp2-data1-v2.xlsx
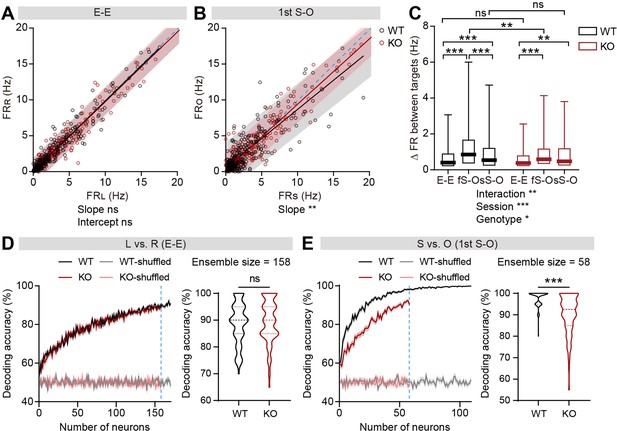
Limited discriminability between social and object targets in IRSp53-KO pExc mPFC neurons.
(A and B) Scatterplot of left in-zone firing rate (FRL) against right in-zone firing rate (FRR) during the E-E session (A) and social in-zone firing rate (FRS) against object in-zone firing rate (FRO) during the first S-O session (B) for WT and IRSp53-KO pExc neurons. Solid lines indicate simple linear regressions for WT (black) and KO (red) neurons. Shaded areas indicate the 95% confidence intervals for the WT (black) and KO (red) firing rates. Blue dashed lines are 45 degree lines. (n=233 [WT-pExc] and 258 [KO-pExc]), **p<0.01, ns, not significant, simple linear regression with slope comparison test (see Methods). (C) Absolute changes in left versus right in-zone firing rates (E-E session) and social versus object in-zone firing rates (first (fS-O) and second (sS-O) S-O sessions) for WT and IRSp53-KO pExc neurons. (n=233 [WT-pExc] and 258 [KO-pExc], *p<0.05, **p<0.01, ***p<0.001, ns, not significant, two-way RM-ANOVA with Sidak’s test). (D and E) Neural decoding of left versus right sidedness during the E-E session (D) and social versus object target during the first S-O session (E) as a function of ensemble size (left) and their decoding performance at maximum comparable ensemble size (indicated by blue dashed line) (right). Ensemble sizes vary due to the limitation of sniffing trials in some experiments (minimum trial number for the SVM decoding was set to 10 per target). Note that the decoding accuracies of WT (pink) and KO (grey) neurons remain similar to chance level (50%) across all tested ensemble sizes after target shuffling. (n=100 decoding trials for 170 and 109 pExc neurons in the E-E and first S-O sessions, respectively [WT] and 100, 158, 58 [KO], ***p<0.001, ns, not significant, Mann-Whitney test). See Supplementary file 2 for statistics. Numerical data used to generate the figure are available in the Figure 4—source data 1.
-
Figure 4—source data 1
Source file for firing-rate discriminability data in Figure 4.
The excel file contains the numerical data used to generate Figure 4A–E.
- https://cdn.elifesciences.org/articles/74998/elife-74998-fig4-data1-v2.xlsx
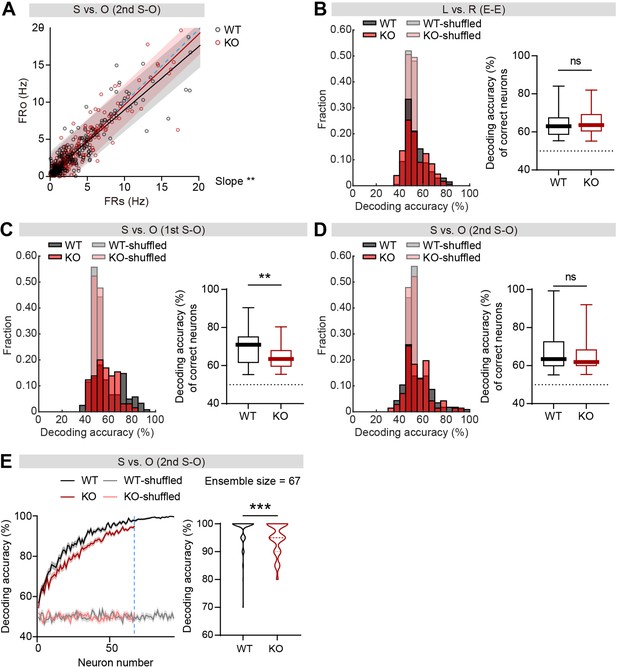
Comparable firing-rate discriminability between social and object targets in individual IRSp53-KO pExc mPFC neurons in the second S-O session.
(A) Scatterplot of social in-zone firing rate (FRS) against object in-zone firing rate (FRO) during the second S-O session for WT and IRSp53-KO pExc neurons. Solid lines indicate simple linear regressions for WT (black) and KO (red) neurons. Shaded areas indicate the 95% confidence intervals for WT (black) and KO (red) firing rates. Blue dashed lines are 45 degree lines. (n=233 [WT-pExc] and 258 [KO-pExc], **p<0.01, simple linear regression with slope comparison test). (B–D) Histogram plotting the decoding performance of left versus right sidedness during the E-E session (B) social versus object target during the first S-O session (C) and second S-O session (D) in individual neurons (left) and the decoding performance of correct neurons (right). The correct neurons were defined as those with decoding accuracies >55% because decoding accuracies fall within 45% and 55% after target shuffling. (n=78, 73, and 47 correct pExc neurons in the E-E, first S-O, and second S-O sessions, respectively [WT] and 69, 34, and 37 [KO], **p<0.01, ns, not significant, Mann-Whitney test (B, D) and unpaired t-test (C)). (E) Neural decoding of social versus object target during the second S-O session as a function of ensemble size (left) and their decoding performance at maximum comparable ensemble size (indicated by blue dashed line) (right). (n=100 decoding trials for 94 pExc neurons in the second S-O session [WT] and 100, 67 [KO], ***p<0.001, ns, not significant, Mann-Whitney test). See Supplementary file 2 for statistics. Numerical data used to generate the figure are available in the Figure 4—figure supplement 1—source data 1.
-
Figure 4—figure supplement 1—source data 1
Source file for firing-rate discriminability data in Figure 4—figure supplement 1.
The excel file contains the numerical data used to generate Figure 4—figure supplement 1A–E.
- https://cdn.elifesciences.org/articles/74998/elife-74998-fig4-figsupp1-data1-v2.xlsx
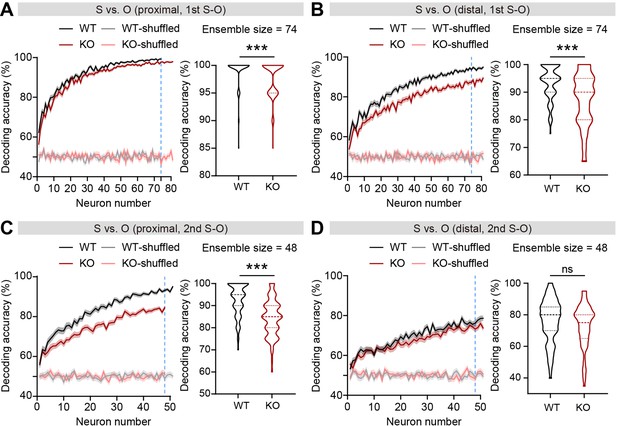
Increased firing-rate discriminability between social and object targets during proximal target interaction compared to distal target interaction.
(A and B) Neural decoding of social versus object target during proximal (A) and distal (B) target interaction in the first S-O session as a function of ensemble size (left) and their decoding performance at maximum comparable ensemble size (right). The blue dashed line indicates the maximum comparable ensemble size based on the first S-O session. Ensemble sizes over 80 neurons were not shown for better comparison between proximal and distal conditions. (n=100 decoding trials for 74 and 145 pExc neurons during proximal and distal interactions [WT] and 100, 89, 177 [KO], ***p<0.001, Mann-Whitney test). (C and D) Neural decoding of social versus object target during proximal (C) and distal (D) target interaction in the second S-O session as a function of ensemble size (left) and their decoding performance at maximum comparable ensemble size (right). The blue dashed line indicates the maximum comparable ensemble size based on the first S-O session. Ensemble sizes over 50 neurons were not shown for better comparison between proximal and distal conditions. (n=100 decoding trials for 66 and 145 pExc neurons during proximal and distal interactions [WT] and 100, 151, 151 [KO], ***p<0.001, ns, not significant, Mann-Whitney test). See Supplementary file 2 for statistics. Numerical data used to generate the figure are available in the Figure 4—figure supplement 2—source data 1.
-
Figure 4—figure supplement 2—source data 1
Source file for firing-rate discriminability data in Figure 4—figure supplement 2.
The excel file contains the numerical data used to generate Figure 4—figure supplement 2A–D.
- https://cdn.elifesciences.org/articles/74998/elife-74998-fig4-figsupp2-data1-v2.xlsx
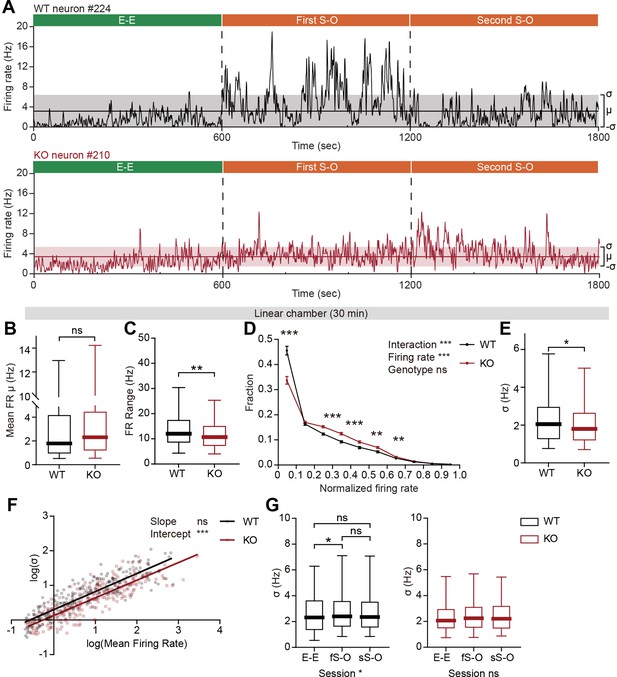
Limited firing-rate range and variability in IRSp53-KO pExc mPFC neurons during linear chamber exploration.
(A) Instantaneous firing-rate traces of representative WT (top) and IRSp53-KO (bottom) pExc neurons (3 s window advanced in 1 s steps) during a sample linear chamber experiment (30 min). Solid horizontal lines indicate the overall mean firing rates (μ). Shaded regions indicate one standard deviation (, sigma). (B) Mean firing rate of WT and IRSp53-KO pExc neurons during the 30 min linear chamber test. (n=233 [WT-pExc] and 258 [KO-pExc], ns, not significant, Mann-Whitney test). (C) Firing-rate ranges (maximum – minimum instantaneous firing rate) of WT and IRSp53-KO pExc neurons during the linear chamber test (n=233 [WT-pExc] and 258 [KO-pExc], **p<0.01, Mann-Whitney test). (D) Mean (± SEM) histograms of normalized instantaneous firing rate during the linear chamber test. For each neuron, instantaneous firing rates were normalized by its maximum instantaneous firing rates. (n=233 [WT-pExc] and 258 [KO-pExc], **p<0.01, ***p<0.001, ns, not significant, two-way RM-ANOVA with Bonferroni’s multiple comparisons test). (E) Sigma values of the instantaneous firing rates of WT and IRSp53-KO pExc neurons during the linear chamber test. (n=233 [WT-pExc] and 258 [KO-pExc], *p<0.05, Mann-Whitney test). (F) Log-scale scatter plot of sigma values against mean firing rates of WT and IRSp53-KO pExc neurons during the linear chamber test. Solid lines indicate simple linear regression of WT (black) and KO (red) values. (n=233 [WT-pExc] and 258 [KO-pExc], ***p<0.001, ns, not significant, slope comparison test (see Methods)). (G) Sigma values for the instantaneous firing rates of WT (left) and IRSp53-KO (right) pExc neurons during the E-E, first S-O (fS-O), and second S-O (sS-O) sessions of the linear chamber test. (n=233 [WT-pExc] and 258 [KO-pExc], *p<0.05, ns, not significant, Friedman test followed by Dunn’s multiple comparisons test). See Supplementary file 2 for statistics. Numerical data used to generate the figure are available in the Figure 5—source data 1.
-
Figure 5—source data 1
Source file for instantaneous firing rate data in Figure 5.
The excel file contains the numerical data used to generate Figure 5A–G.
- https://cdn.elifesciences.org/articles/74998/elife-74998-fig5-data1-v2.xlsx
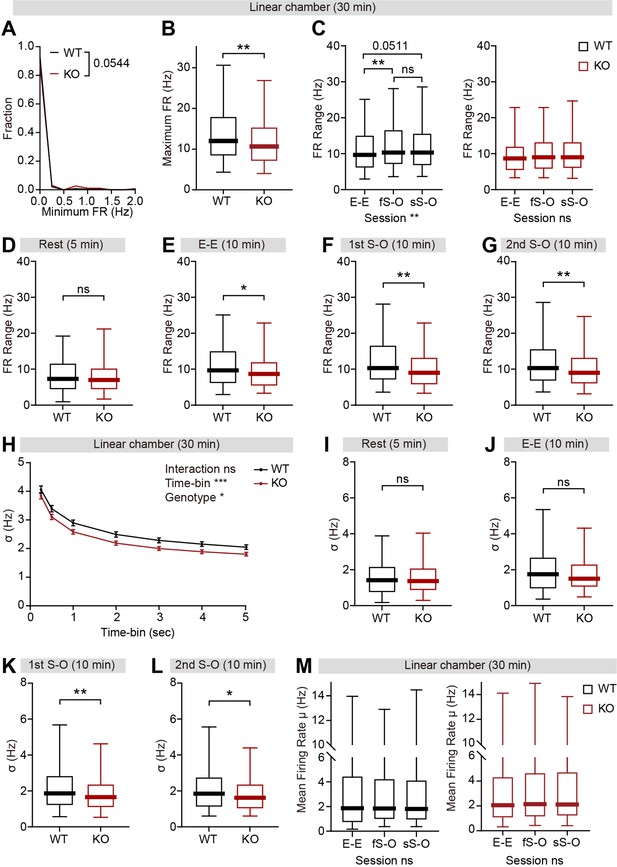
Decreased firing-rate range and variability in IRSp53-KO pExc mPFC neurons selectively during linear chamber exploration.
(A and B) Minimum (A) and maximum (B) instantaneous firing rates of WT and IRSp53-KO pExc neurons during the 30 min linear chamber test. (n=233 [WT-pExc] and 258 [KO-pExc], **p<0.01, Mann-Whitney test). (C) Firing-rate ranges of WT (left) and IRSp53-KO (right) pExc neurons during the E-E, first S-O (fS-O), and second S-O (sS-O) sessions of the linear chamber test. (n=233 [WT-pExc] and 258 [KO-pExc], **p<0.01, ns, not significant, Friedman test). (D–G) Comparison of firing-rate ranges of WT and IRsp53-KO pExc neurons during 5 min rest period (D), E-E (E), first S-O (F), and second S-O (G) sessions of the linear chamber test. (n=233 [WT-pExc] and 258 [KO-pExc], *p<0.05, **p<0.01, ns, not significant, Mann-Whitney test). (H) Mean (± SEM) sigma values of instantaneous firing rates during the 30 min linear chamber test calculated using different time-bin sizes (0.25–5 s). Window sizes were set to be the same as the time-bin size. Note that the overall sigma values of IRSp53-KO neurons are significantly smaller than those of the WT neurons for all analyzed time-bin sizes. (n=233 [WT-pExc] and 258 [KO-pExc], *p<0.05, ***p<0.001, ns, not significant, two-way RM-ANOVA). (I–L) Sigma values for the instantaneous firing rates (3 s window advanced in 1 s steps) during the 5-min rest period (I), E-E (J), first S-O (K), and second S-O (L) sessions in WT and IRSp53-KO pExc neurons. (n=233 [WT-pExc] and 258 [KO-pExc], *p<0.05, **p<0.01, ns, not significant, Mann-Whitney test). (M) Mean instantaneous firing rates of WT (left) and IRSp53-KO (right) pExc neurons during the E-E, first S-O, and second S-O sessions of the linear chamber test. (n=233 [WT-pExc] and 258 [KO-pExc], ns, not significant, Friedman test). See Supplementary file 2 for statistics. Numerical data used to generate the figure are available in the Figure 5—figure supplement 1—source data 1.
-
Figure 5—figure supplement 1—source data 1
Source file for instantaneous firing rate data in Figure 5—figure supplement 1.
The excel file contains the numerical data used to generate Figure 5—figure supplement 1A–M.
- https://cdn.elifesciences.org/articles/74998/elife-74998-fig5-figsupp1-data1-v2.xlsx
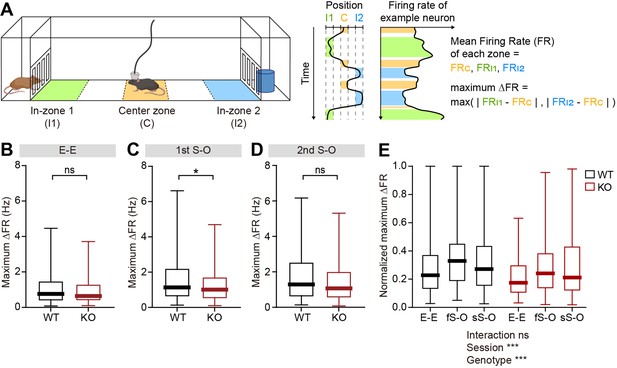
Limited firing-rate changes in response to social and object targets in IRSp53-KO pExc mPFC neurons.
(A) Definition of in-zone and center zone. The first and fifth of the equally divided five 9-cm-long areas were defined as in-zones (I1 and I2, respectively) while the third area was defined as the center zone (C). For each neuron, the maximum Δ firing rate is defined as the higher value among the firing rate differences between the center zone and two in-zones (left and right for E-E session, social and object for S-O sessions). (B–D) Maximum Δ firing rates of WT and IRSp53-KO pExc neurons during the E-E (B), first S-O (C), and second S-O (D) sessions. (n=233 [WT-pExc] and 258 [KO-pExc], *p<0.05, ns, not significant, Mann-Whitney test). (E) Normalized maximum Δ firing rates of WT and IRSp53-KO pExc neurons during the E-E, first S-O (fS-O), and second S-O sessions (sS-O). Normalized maximum Δ firing rates = max (|(FRI1 - FRC) / (FRI1 +FRC)|, |(FRI2 - FRC) / (FRI2 +FRC)|). (n=233 [WT-pExc] and 258 [KO-pExc], ***p<0.001, ns, not significant, two-way RM-ANOVA). See Supplementary file 2 for statistics. Numerical data used to generate the figure are available in the Figure 5—figure supplement 2—source data 1.
-
Figure 5—figure supplement 2—source data 1
Source file for firing-rate change data in Figure 5—figure supplement 2.
The excel file contains the numerical data used to generate Figure 5—figure supplement 2B–E.
- https://cdn.elifesciences.org/articles/74998/elife-74998-fig5-figsupp2-data1-v2.xlsx
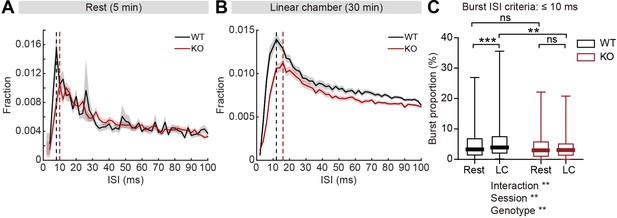
Decreased burst firing and spike variability in IRSp53-KO pExc mPFC neurons during linear chamber exploration.
(A and B) Mean (± SEM) histogram of interspike intervals (ISI) during the 5-min rest (A) and the 30 min linear chamber (B) periods. Dashed lines denote the peaks of mean ISI distributions for WT (black) and IRSp53-KO (red) neurons (8ms [WT] and 10ms [KO] for rest, 12ms [WT] and 16ms [KO] for linear chamber periods). (n=233 [WT-pExc] and 258 [KO-pExc]). (C) Burst proportion (proportion of burst spikes out of total spikes) of WT and IRSp53-KO pExc neurons during the rest and linear chamber (LC) periods for burst ISI threshold of 10ms. (n=233 [WT-pExc] and 258 [KO-pExc], **p<0.01, ***p<0.001, ns, not significant, two-way RM-ANOVA with Sidak’s multiple comparisons test). See Supplementary file 2 for statistics. Numerical data used to generate the figure are available in the Figure 6—source data 1.
-
Figure 6—source data 1
Source file for ISI and burst firing data in Figure 6.
The excel file contains the numerical data used to generate Figure 6A–C.
- https://cdn.elifesciences.org/articles/74998/elife-74998-fig6-data1-v2.xlsx
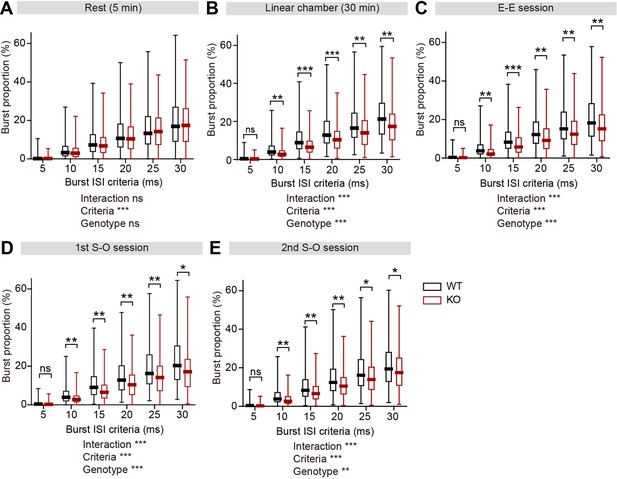
Decreased burst firing in IRSp53-KO pExc mPFC neurons during linear chamber exploration.
(A–E) Burst proportions of WT and IRSp53-KO pExc neurons during the rest (A) and linear chamber (B) periods, as well as, E-E (C), first (D) and second (E) S-O sessions, for different burst ISI thresholds. (n=233 [WT-pExc] and 258 [KO-pExc], *p<0.05, **p<0.01, ***p<0.001, ns, not significant, two-way RM-ANOVA with Sidak’s multiple comparisons test). See Supplementary file 2 for statistics. Numerical data used to generate the figure are available in the Figure 6—figure supplement 1—source data 1.
-
Figure 6—figure supplement 1—source data 1
Source file for burst firing data in Figure 6—figure supplement 1.
The excel file contains the numerical data used to generate Figure 6—figure supplement 1A–E.
- https://cdn.elifesciences.org/articles/74998/elife-74998-fig6-figsupp1-data1-v2.xlsx
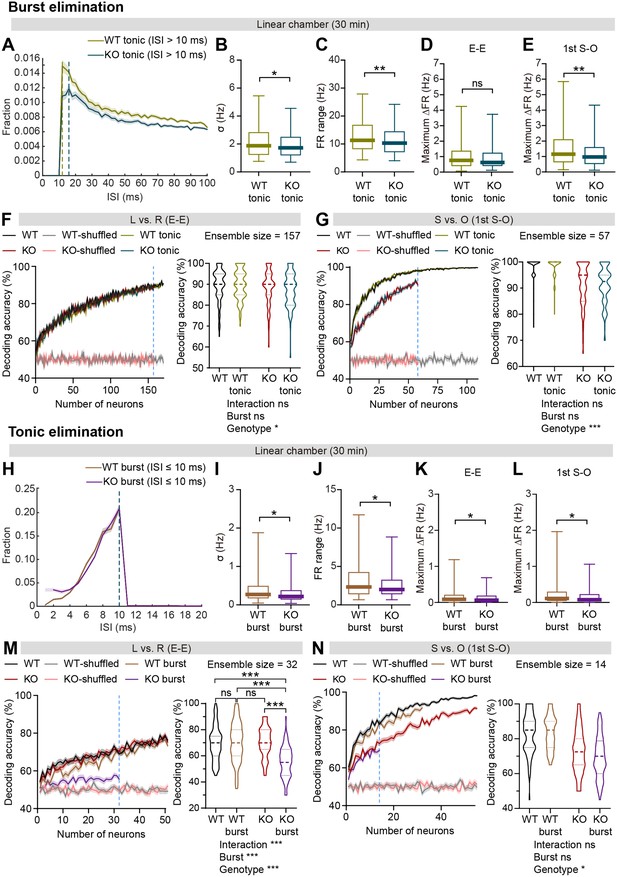
Burst firing is sufficient for discrimination of social and object targets.
(A) Mean (± SEM) histogram of interspike intervals (ISI) during 30 min linear chamber sessions after burst spike (ISI ≤10ms) elimination. Note that the peaks of mean ISI distributions for WT (12ms, green) and IRSp53-KO (16ms, blue) neurons are indicated by dashed lines. (n=233 [WT-pExc] and 258 [KO-pExc]). (B and C) Firing-rate (FR) variability (B) and range (C) during 30 min linear chamber sessions after burst spike elimination. (n=233 [WT-pExc] and 258 [KO-pExc], *p<0.05, **p<0.01, Mann-Whitney test). (D and E) Maximum Δ firing rates of WT and IRSp53-KO pExc neurons during the E-E (D) and first S-O (E) sessions after burst spike elimination. (n=233 [WT-pExc] and 258 [KO-pExc], *p<0.05, Mann-Whitney test). (F and G) Neural decoding of left versus right sidedness during the E-E session (F) and social versus object target during the first S-O session (G) as a function of ensemble size (left) and their decoding performance at maximum comparable ensemble size (indicated by blue dashed line) (right) after burst spike elimination. Note that the decoding accuracies of WT (pink) and KO (grey) neurons remain similar to chance level (50%) across all tested ensemble sizes after target shuffling. (n=100 decoding trials for 170, 109 pExc neurons before burst elimination and 168, 107 pExc neurons after burst elimination in the E-E and first S-O sessions, respectively [WT] and 100, 158, 58, 157, 57 [KO], *p<0.05, ***p<0.001, ns, not significant, two-way RM-ANOVA). (H) Mean (± SEM) histogram of interspike intervals (ISI) during 30 min linear chamber sessions after tonic spike (ISI >10ms) elimination. Note that the peaks of mean ISI distributions for WT (10ms, brown) and IRSp53-KO (10ms, purple) neurons are indicated by dashed lines. (n=233 [WT-pExc] and 258 [KO-pExc]). (I and J) Firing-rate variability (I) and range (J) during 30 min linear chamber sessions after tonic spike elimination. (n=233 [WT-pExc] and 258 [KO-pExc], *p<0.05, Mann-Whitney test) (K and L) Maximum Δ firing rates of WT and IRSp53-KO pExc neurons during the E-E (K) and first S-O (L) sessions after tonic spike elimination. (n=233 [WT-pExc] and 258 [KO-pExc], *p<0.05, Mann-Whitney test). (M and N) Neural decoding of left versus right sidedness during the E-E session (M) and social versus object target during the first S-O session (N) as a function of ensemble size (left) and their decoding performance at maximum comparable ensemble size (right) after tonic spike elimination. (n=100 decoding trials for 50, 54 pExc neurons before burst elimination and 50, 32 pExc neurons after burst elimination in the E-E and first S-O sessions, respectively [WT] and 100, 50, 54, 32, 14 [KO], ***p<0.001, ns, not significant, two-way RM-ANOVA with Tukey’s multiple comparisons test). See Supplementary file 2 for statistics. Numerical data used to generate the figure are available in the Figure 6—figure supplement 2—source data 1.
-
Figure 6—figure supplement 2—source data 1
Source file for firing rate data in Figure 6—figure supplement 2.
The excel file contains the numerical data used to generate Figure 6—figure supplement 2A–N.
- https://cdn.elifesciences.org/articles/74998/elife-74998-fig6-figsupp2-data1-v2.xlsx
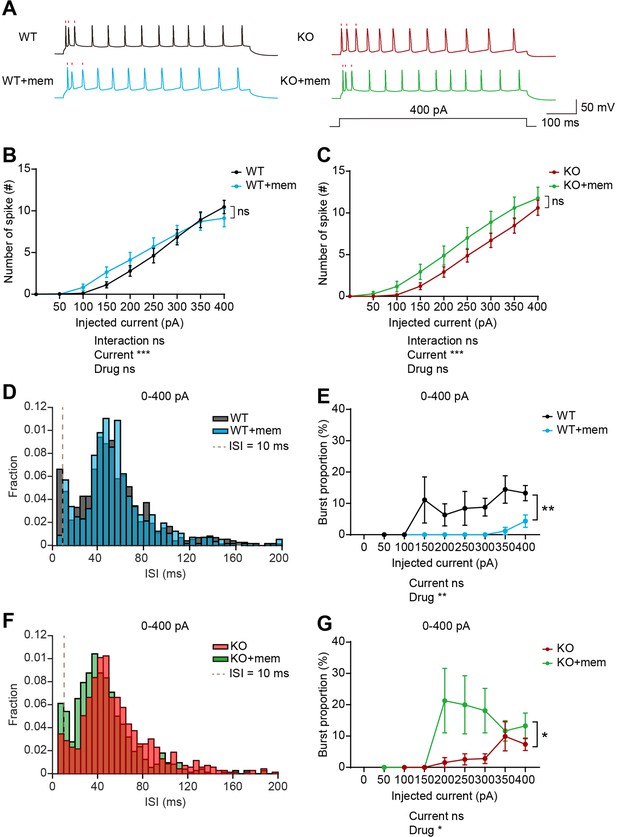
Memantine treatment increases and decreases burst firing in WT and IRSp53-KO prefrontal neurons, respectively.
(A) Examples of firing traces induced by current injections in memantine-untreated and memantine-treated (+mem) WT and IRSp53-KO layer 5 pyramidal neurons in mouse brain slices containing the prelimbic region of the mPFC. Timings of the first three action potentials are indicated by raster plots (red). (B and C) Differences in intrinsic excitability between memantine-untreated and memantine-treated layer 5 pyramidal neurons in the prelimbic region of the mPFC in WT (B) and IRSp53-KO mice (C) (3–6 months), indicated by current-firing curves. (n=24 neurons from 3 mice [WT], 18, 3 [WT +mem], 23, 3 [KO], 17, 3 [KO +mem], ***p<0.001, ns, not significant, two-way RM-ANOVA). (D and E) Interspike interval (ISI) histogram (D) and burst firing patterns (E, current-burst firing curve) in layer 5 pyramidal neurons in the mPFC prelimbic region in WT mice (3–6 months) in the presence and absence of memantine treatment. ISIs below the dashed lines (10ms) are classified as burst firing. (n=24, 3 [WT], 18, 3 [WT +mem], **p<0.01, mixed-effects ANOVA). (F and G) ISI histograms (F) and burst firing patterns (G, current-burst firing curve) in layer 5 pyramidal neurons in the mPFC prelimbic region in IRSp53-KO mice (3–6 months) in the presence and absence of memantine treatment. ISIs below the dashed lines (10ms) are classified as burst firing. (n=23, 3 [KO], 17, 3 [KO +mem], *p<0.05, mixed-effects ANOVA). See Supplementary file 2 for statistics. Numerical data used to generate the figure are available in the Figure 7—source data 1.
-
Figure 7—source data 1
Source file for slice electrophysiology data in Figure 7.
The excel file contains the numerical data used to generate Figure 7B–G.
- https://cdn.elifesciences.org/articles/74998/elife-74998-fig7-data1-v2.xlsx
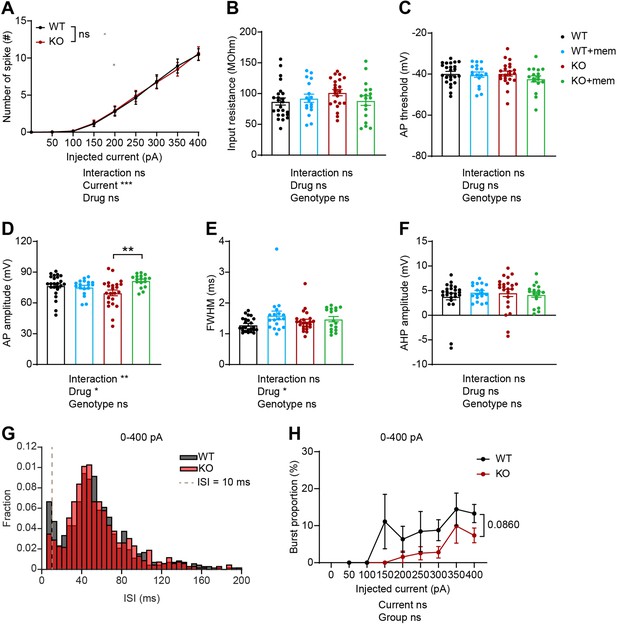
Intrinsic excitability-related parameters in memantine-treated/untreated WT and IRSp53-KO prefrontal neurons.
(A) Baseline intrinsic excitability in layer 5 pyramidal neurons in the prelimbic region of the mPFC in WT and IRsp53-KO mice (3–6 months), indicated by current-firing curve. (n=24 neurons from 3 mice [WT], 23, 3 [KO], ns, not significant, two-way RM-ANOVA). (B–F) Action potential (AP)-related parameters in layer 5 pyramidal neurons in memantine-treated/untreated slices containing the mPFC prelimbic region in WT and IRSp53-KO mice (3–6 months), as indicated by input resistance (B), AP threshold (C), AP amplitude (D), full width at half maximum (FWMH) (E), and afterhyperpolarization (AHP) amplitude (F). (n=24, 3 [WT], 18, 3 [WT +mem], 23, 3 [KO], 17, 3 [KO +mem], *p<0.05, **p<0.01, ns, not significant, two-way RM-ANOVA with Sidak’s multiple comparisons test). (G and H) ISI histograms (G) and burst firing patterns (H, current-burst firing curve) in layer 5 pyramidal neurons in the mPFC prelimbic region in IRSp53-KO mice (3–6 months) in the absence of memantine treatment. ISIs below the dashed lines (10ms) are classified as burst firing. (n=24, 3 [WT], 23, 3 [KO], ns, not significant, mixed-effects ANOVA). See Supplementary file 2 for statistics. Numerical data used to generate the figure are available in the Figure 7—figure supplement 1—source data 1.
-
Figure 7—figure supplement 1—source data 1
Source file for slice electrophysiology data in Figure 7—figure supplement 1.
The excel file contains the numerical data used to generate Figure 7—figure supplement 1A–H.
- https://cdn.elifesciences.org/articles/74998/elife-74998-fig7-figsupp1-data1-v2.xlsx
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Gene (Mus musculus) | IRSp53 (Baiap2) | Kim et al., 2009 | N/A | |
| Software, algorithm | DeepLabCut | Lauer et al., 2022; Mathis et al., 2018 | Ver 2.0, 2.2 | Used for tracking of mice body parts. |
| Software, algorithm | Ethovision XT 13 | Noldus https://www.noldus.com/ | Ver 13 | Used for measuring the locomotion of mice. |
| Software, algorithm | GraphPad Prism 9.0 | GraphPad https://www.graphpad.com/ | Ver 9.0 | Used for all statistics used in the current study. |
| Software, algorithm | MATLAB | MathWorks https://www.mathworks.com/ | Ver 2020a | Used for spike analysis. |
| Software, algorithm | MClust 4.0 | David Redish http://redishlab.neuroscience.umn.edu/MClust/MClust.html | Ver 4.0 | Used for manual single-unit isolation. |
| Software, algorithm | Clampex | Molecular devices | Ver 10.7 | Used for recording electrophysiological signals from cells |
| Software, algorithm | Clampfit | Molecular devices | Ver 10.7 | Used for analysing electrophysiological data |
| Software, algorithm | pCLAMP | Molecular devices | Ver 10 | Used for recording electrophysiological signals from cells |
| Software, algorithm | IntrinsicVIEW | https://github.com/parkgilbong/IntrinsicVIEW; Kim and Kim, 2020 | N/A | Used for intrinsic property analysis |
| Other | Digital lynx SX | Neuralynx https://neuralynx.com/ | Used for in vivo single-unit recording | |
| Other | Multiclamp 700B | Molecular devices | Used for recording electrophysiological signals from cells | |
| Other | Digidata 1550 | Molecular devices | Used for recording electrophysiological signals from cells | |
| Chemical compound, drug | Memantine hydrochloride ≥98% (GC) | Sigma | M9292 | Used for slice electrophysiology experiment |
Additional files
-
Supplementary file 1
Recorded number of neurons from each WT and IRSp53-KO mouse.
- https://cdn.elifesciences.org/articles/74998/elife-74998-supp1-v2.xlsx
-
Supplementary file 2
Statistics for all figures provided.
- https://cdn.elifesciences.org/articles/74998/elife-74998-supp2-v2.xlsx
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/74998/elife-74998-transrepform1-v2.docx





