A tonic nicotinic brake controls spike timing in striatal spiny projection neurons
Figures
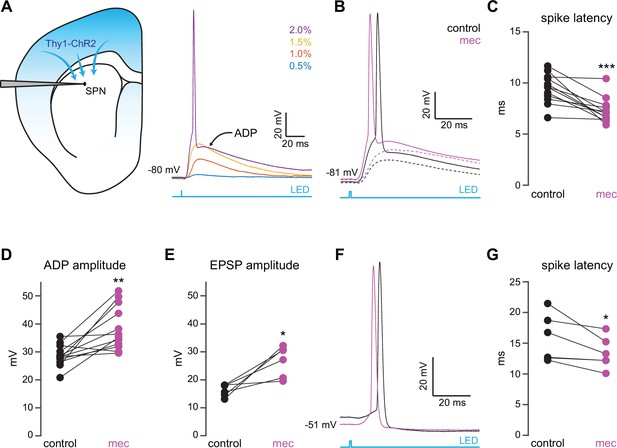
Nicotinic acetylcholine receptor (nAChR)-dependent inhibition and delay of spike latency in spiny projection neurons (SPNs) activated by corticostriatal fibers.
(A) Left: diagram of recording configuration. An SPN is patched in an acute slice from a Thy1-ChR2 mouse. Right: 1-ms-long 470 nm LED pulse of increasing intensity generates excitatory postsynaptic potentials (EPSPs), or an AP followed by an afterdepolarization (ADP) in an SPN. (B) Examples of the effect of 10 µM mecamylamine (mec), an nAChR antagonist, on EPSP amplitude (dashed) or spike latency and ADP amplitude (solid). (C–E) Mecamylamine significantly shortens spike latencies (p=4.8·10–4, n=12 SPNs, signed-rank test [SRT]) (C), as well as ADP (p=2.4·10–3, n=12 SPNs, SRT) (D) and EPSP (p=0.03, n=6 SPNs, SRT) (E) amplitudes. (F) Examples of the effect of mecamylamine on latency of a spike triggered synaptically from a depolarized potential with an LED pulse. (G) Mecamylamine significantly shortens spike latencies in SPNs held at a depolarized potential (p=0.03, n=6 SPNs, SRT). Two-sided Wilcoxon SRT. ***p<0.001, **p<0.01, *p<0.05.
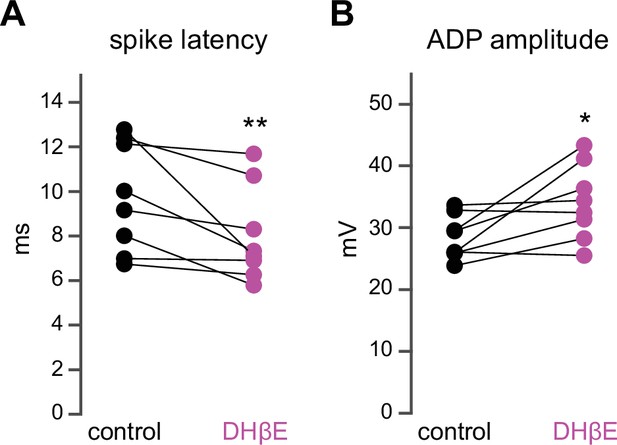
Dihydro-β-erythroidine hydrobromide (DHβE) mimics the effect of mecamylamine on striatal spiny projection neuron (SPN) spike latency and afterdepolarization (ADP) amplitude.
(A–B) DHβE significantly shortens spike latencies (p=7.8·10–3, n=8 SPNs, signed-rank test [SRT]) (A) and ADP amplitudes (p=0.03, n=8 SPNs, SRT) (B).
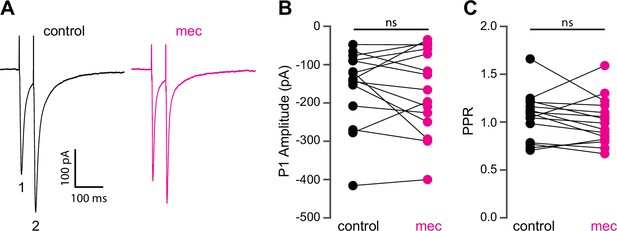
Mecamylamine does not alter release probability of excitatory synaptic inputs.
(A) Example responses of an striatal spiny projection neuron (SPN) to paired pulse stimulation (evoked by two local electrical stimuli separated by 50 ms), before and in the presence of mecamylamine (10 µM). (B) Mecamylamine had no effect on the excitatory postsynaptic current (EPSC) amplitude evoked by the first paired stimulus (P1) (p=0.59, n=15 SPNs, signed-rank test [SRT]). (C) Mecamylamine had no effect on the paired pulse ratio (PPR) (P2/P1, p=0.33, n=15 SPNs, SRT).
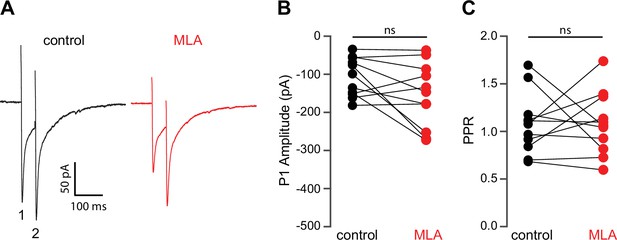
Methyllycaconitine (MLA) does not alter release probablity of excitatory synaptic inputs.
(A) Example responses of a striatal spiny projection neuron (SPN) to paired pulse stimulation (electrical stimuli separated by 50 ms) before and in the presence of MLA (5 µM). (B) MLA had no effect on the excitatory postsynaptic current (EPSC) amplitude evoked by the first paired stimulus (P1) (p=0.1475, n=11, signed-rank test [SRT]). (C) MLA had no effect on the paired pulse ratio (PPR) (P2/P1, p>0.9999, n=11, SRT).
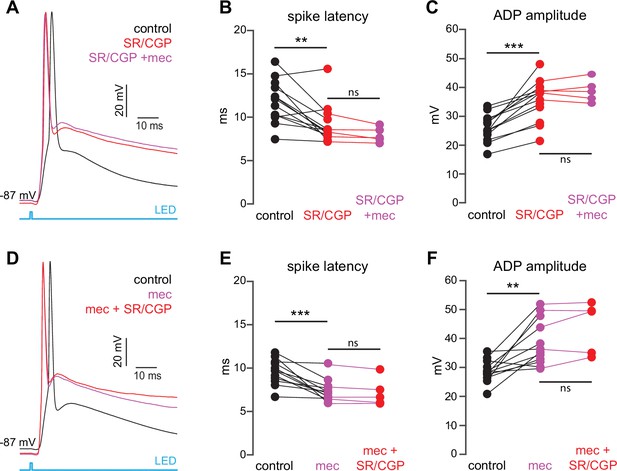
The nicotinic acetylcholine receptor (nAChR)-dependent inhibition of corticostriatal striatal spiny projection neuron (SPN) activation is mediated through and saturates GABAergic inhibition.
(A) Example of the occlusion of the mecamylamine effect on optogenetic synaptic activation of SPNs by receptor (GABAR) antagonists, 10 µM SR-95531 (GABAAR antagonist) and 2 µM CGP-55845 (GABABR antagonist). Distribution of spike latencies (B) and ADP amplitude (C) in response to application of GABAR antagonists followed by mecamylamine, showing that application of GABAR antagonist significantly shortens the action potential (AP) latency (p=4.6·10–3, n=13 SPNs, signed-rank test [SRT]) and enhancement of the ADP amplitude (p=2.4·10–4, n=13 SPNs, SRT). In contrast, the subsequent mecamylamine application fails to further shorten the AP latency (p=0.0625, n=5 SPNs, SRT) or further enhance the ADP amplitude (p=1, n=5, SRT). (D) Example of how the mecamylamine effect saturates the GABAergic inhibition of the optogenetic synaptic activation of SPNs. (E–F) Distribution of spike latencies (E) and ADP amplitude (F) in response to application of mecamylamine followed by GABAR antagonists, showing that the subsequent application of GABAR antagonists fails to further shorten the AP latency (p=0.25, n=5 SPNs, SRT) or further enhance the ADP amplitude (p=0.44, n=5 SPNs, SRT).
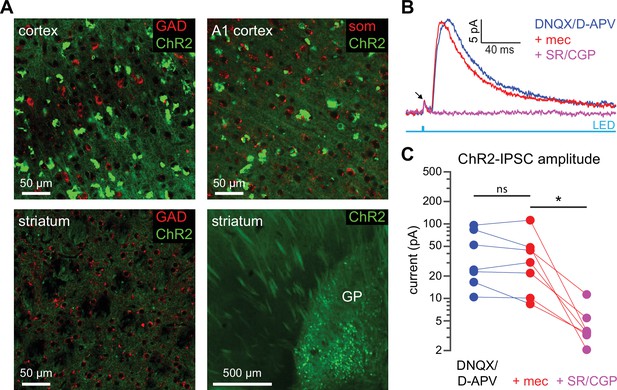
ChR2 expression is not observed in cortical or striatal GABAergic neurons in Thy1-ChR2 mice.
(A) Confocal images from the sensorimotor cortex, auditory (A1) cortex, striatum, and globus pallidus (GP) of a Thy1-ChR2 mouse. Tissue was stained for glutamic acid decarboxylase (GAD) or somatostatin (SOM); colocalization of ChR2 (green) and GAD or SOM was not observed. ChR2 expression was observed in a population of GP neurons (in some mice, EYFP aggregates could be observed in the cortex). (B–C) Example recordings of a striatal spiny projection neuron (SPN) in the presence of the AMPA and NMDA receptor blockers DNQX and D-APV reveal the presence of an optogenetically generated monosynaptic inhibitory postsynaptic current (IPSC) in SPNs from Thy1-ChR2 mice. Arrow indicates compound action current in ChR2-laden axons (Tanimura et al., 2016) (B). This monosynaptic IPSC, however, was insensitive to mecamylamine (glutamate blockers vs. mecamylamine [mec]: p=0.578; glutamate blockers + mec vs. GABA receptor blockers: p=0.0156; n=7, signed-rank test [SRT]) (C).
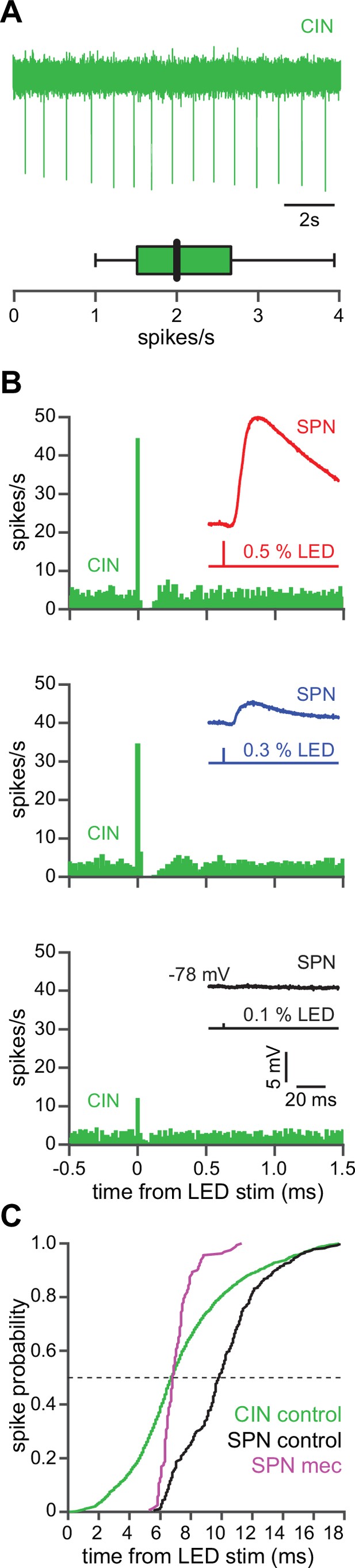
The autonomous firing of cholinergic interneurons (CINs) enables them to respond more rapidly and vigorously than striatal spiny projection neurons (SPNs) to feedforward corticostriatal excitation.
(A) Example of the autonomous discharge of a CIN recorded in cell-attached configuration (top) and the distribution of firing rates (bottom, n=15 CINs) in an acute striatal slice. (B) Peristimulus time histograms (PSTHs) of a CIN in response to optogenetic activation of corticostriatal fibers at various 470 nm LED intensities as compared to the amplitude of the excitatory postsynaptic potentials (EPSPs) evoked in a nearby SPN (insets), demonstrating that CINs are much more sensitive than SPNs to cortical activation. (C) Cumulative distribution of the latency to first spike of CINs recorded in cell-attached mode (green, n=8 CINs) as compared to the latency to first spike in an SPN recorded (from a resting state) in the whole-cell mode (n=12), before (black) and after (magenta) application of mecamylamine.
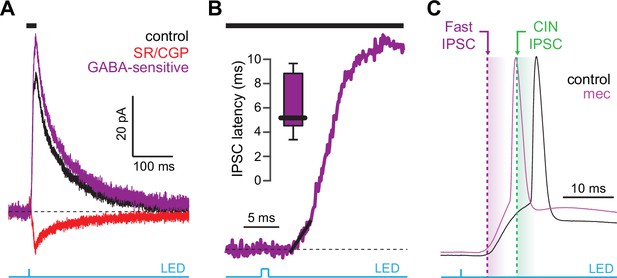
Latency of feedforward GABAergic inhibitory postsynaptic currents (IPSCs) in striatal spiny projection neurons (SPNs) activated synaptically by corticostriatal fibers.
(A) Left: IPSCs recorded in an SPN held at +10 mV before (black) and after (red) application of GABAR antagonists, reveals GABA-sensitive (purple) IPSC. Right: zoom-in of the GABAergic IPSCs enables estimation (fit of diagonal black line) of the latency of the feedforward GABA-sensitive inhibition. (B) Distribution of the latency of feedforward GABAergic inhibition to SPNs in the Thy1-ChR2 mouse (n=7 SPNs). (C) Schematic demonstrating that the feedforward GABAergic inhibition precedes the earliest timing of feedforward cholinergic disynaptic inhibition of SPNs, indicating that phasic activation of CINs cannot explain the nicotinic acetylcholine receptor (nAChR)-dependent delay of spike latency.
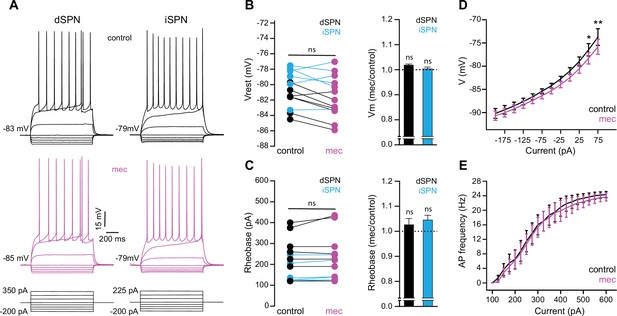
Striatal spiny projection neuron (SPN) intrinsic excitability is not increased by nicotinic acetylcholine receptor (nAChR) blockade.
(A) Example current-voltage (IV) traces of direct pathway SPNs (dSPNs) and indirect pathway SPNs (iSPNs) in the absence and presence of 10 µM mecamylamine. (B) Left: resting membrane potential of dSPNs (black) and iSPNs (blue) in the absence and presence of mecamylamine (all SPNs: p=0.43, n=13; dSPNs: p=0.16, n=7; iSPNs: p=0.87, n=6; signed-rank test [SRT]). Right: percent change in resting membrane potential after mecamylamine application. (C) Left: rheobase current of dSPNs and iSPNs in the absence and presence of mecamylamine (all SPNs: p=0.075, n=13; dSPNs: p=0.5, n=7; iSPNs: p=0.095, n=6; SRT). Right: percent change in rheobase current after mecamylamine application. (D) Voltage responses to subthreshold current injections are not enhanced by mecamylamine (F(11,120)=0.86, p=0.58; n=11; two-way ANOVA), though a post hoc Bonferroni test reveals a significant mecamylamine-induced decrease at 50 pA (p<0.05) and 75 pA (p<0.01) current injections. (E) Action potential (AP) firing frequencies in response to suprathreshold somatic current injections were unaffected by mecamylamine (F(20,231)=0.43, p=0.99; n=12; two-way ANOVA).
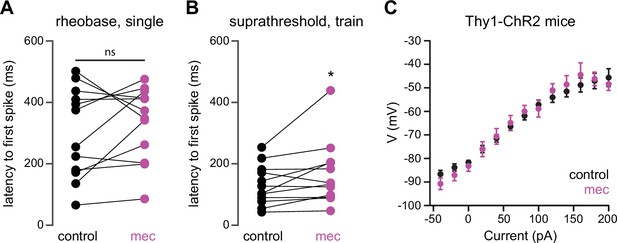
Mecamylamine does not shorten the latency of striatal spiny projection neuron (SPN) action potentials (APs) induced by somatic current injection, or alter somatic excitability of SPNs from Thy1-ChR2 mice.
(A) Latencies of APs induced by rheobase current injection (which induced only a single spike) in a mixed population of SPNs before and in the presence of mecamylamine (p=0.4238, n=12, signed-rank test [SRT]). (B) Latencies of the first AP in a train induced by suprathreshold current injection in a mixed population of SPNs before and in the presence of mecamylamine (p=0 022, n=12, SRT). (C) Current-voltage (IV) curves (mean ± SEM) of a mixed population of SPNs from Thy1-ChR2 mice are not affected by mecamylamine.
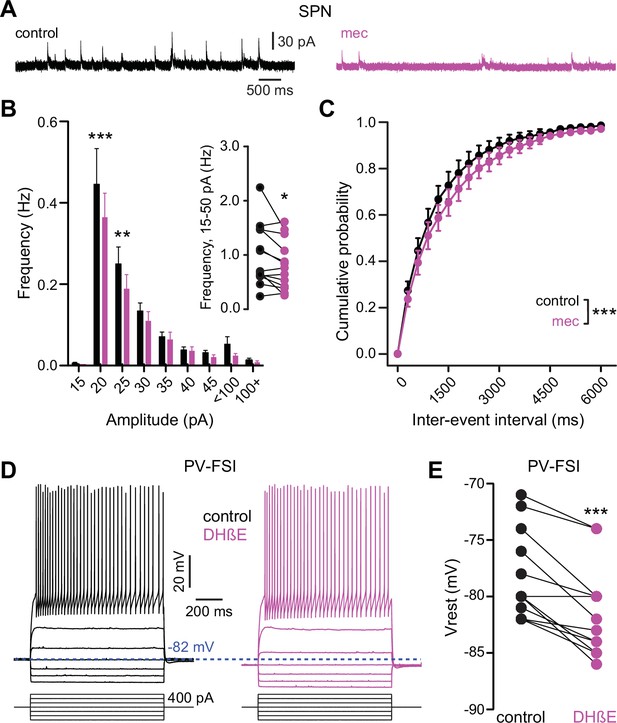
Nicotinic acetylcholine receptor (nAChR) blockade hyperpolarizes parvalbumin-positive fast-spiking interneurons (PV-FSI) resting membrane potential and reduces the frequency of spontaneous inhibitory postsynaptic currents (sIPSCs) in striatal spiny projection neurons (SPNs).
(A) Example recordings of sIPSCs from SPNs voltage-clamped at +10 mV, before and during mecamylamine (10 µM) application. (B) Mecamylamine significantly enhanced sIPSC frequency in SPNs (F8,108=2.607, p=0.012; n=13; two-way ANOVA). A post hoc Bonferroni test revealed that this decrease was limited to low-amplitude sIPSCs (20 pA bin: p<0.001; 25 pA bin: p<0.01). (C) Mecamylamine caused a rightward shift in the cumulative probability distribution of sIPSC interevent intervals in SPNs (F(20,252)=2.10, p=4.7·10–3; n=13; two-way ANOVA). (D) Example traces of a PV-FSI before and after wash-in of 1 µM dihydro-β-erythroidine hydrobromide (DHβE). (E) Resting membrane potentials of PV-FSIs before and after wash-in of DHβE (n=12; p=9.8·10–4; signed-rank test [SRT]).
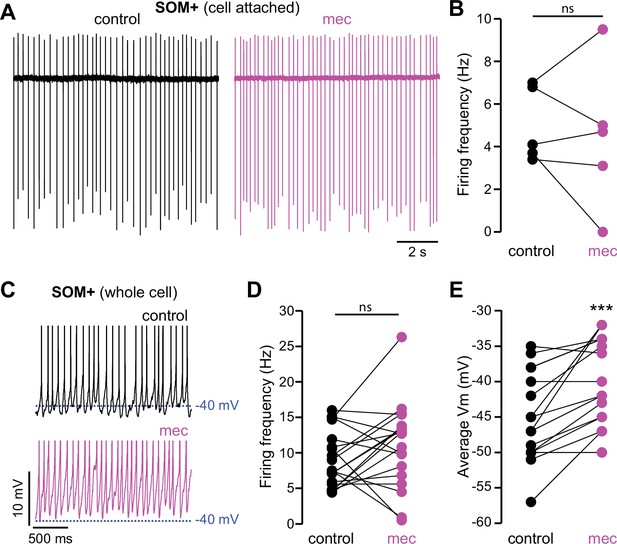
Nicotinic acetylcholine receptor (nAChR) blockade depolarizes somatostatin (SOM)+ interneuron membrane potential.
(A) Example cell-attached recordings of a SOM+ interneuron before and after wash-in of mecamylamine (VC = 0 mV, artificial cerebrospinal fluid [ACSF] in recording electrode). (B) Firing rate calculated from cell-attached recordings before and after mecamylamine (n=5). (C) Example whole-cell recordings of a SOM+ interneuron before and after wash-in of mecamylamine. Action potential (AP) peaks truncated to highlight changes in average membrane potential. (D) Firing rate calculated from whole-cell recordings before and after mecamylamine (n=19, p=0.177, signed-rank test [SRT]). (E) Average membrane potential before and after wash-in of mecamylamine (n=17, p=4·10–4, SRT).
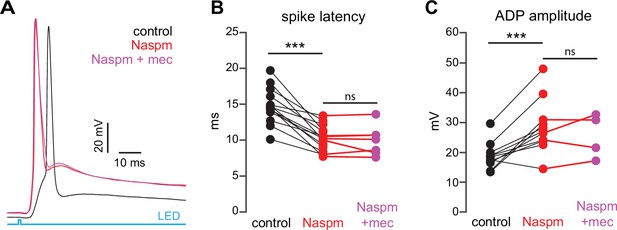
Blockade of Ca2+-permeable AMPA receptors, to prevent cortical activation of parvalbumin-positive fast-spiking interneurons (PV-FSIs) and related GABAergic interneurons (GINs), mimics and occludes the effect of mecamylamine on striatal spiny projection neuron (SPN) spike latency.
(A) Example of the occlusion of the mecamylamine effect on optogenetic synaptic activation of SPNs by the Ca2+-permeable AMPA receptor antagonist 1-naphthyl acetyl spermine (Naspm) (100 µM). Distribution of spike latencies (B) and afterdepolarization (ADP) amplitude (C) in response to application of Naspm followed by mecamylamine show that Naspm significantly shortens the action potential (AP) latency (p=1.22·10–4, n=14 SPNs, signed-rank test [SRT]) and enhancement of the ADP amplitude (p=1.95·10–3, n=11 SPNs, SRT). In contrast, the subsequent mecamylamine application fails to further shorten the AP latency (p=0.6875, n=7 SPNs, SRT) or further enhance the ADP amplitude (p=0.625, n=4, SRT).
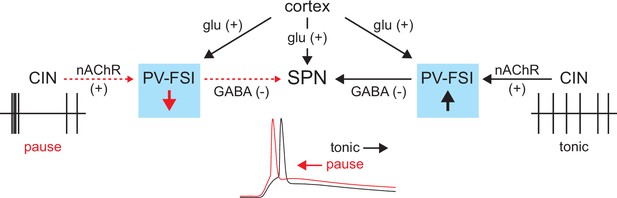
Proposed model of tonic nicotinic acetylcholine receptor (nAChR)-mediated suppression of cortically driven striatal spiny projection neuron (SPN) spike latency.
Example cholinergic interneuron (CIN) and SPN firing patterns shown below; dashed red arrows indicate diminished signaling during CIN pauses; Glu = glutamate.
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Strain, strain background (Mus musculus) | C57BL/6J | Jackson Laboratory | RRID: IMSR_JAX:000664 | |
| Genetic reagent (Mus musculus) | B6.FVB-Tg(Drd2-EGFP/Rpl10a)CP101Htz/J | Jackson Laboratory | RRID: IMSR_JAX:030255 | |
| Genetic reagent (Mus musculus) | B6.Cg-Tg(Drd1a-tdTomato)6Calak/J | Jackson Laboratory | RRID: IMSR_JAX:016204 | |
| Genetic reagent (Mus musculus) | B6.Cg-Tg(Thy1-COP4/EYFP)18Gfng/J | Jackson Laboratory | RRID:IMSR_JAX:007612 | |
| Genetic reagent (Mus musculus) | Pvalb-cre | Jackson Laboratory | RRID: IMSR_JAX:017320 | |
| Genetic reagent (Mus musculus) | Sst-cre | Jackson Laboratory | RRID: IMSR_JAX:018973 | |
| Genetic reagent (Mus musculus) | Ai9 (‘tdTomato’) | Jackson Laboratory | RRID: IMSR_JAX:007909 | |
| Other | AAV5-Ef1a-DIO-EYFP | Addgene | 27056-AAV5 | Adeno-associated virus (AAV) |
| Chemical compound, drug | Mecamylamine hydrochloride | Sigma-Aldrich Tocris | Lot # 019M4108V CAS: 826-39-1 #2843, CAS: 110691-49-1 | |
| Chemical compound, drug | Dihydro-β-erythroidine hydrobromide | Tocris | #2349, CAS: 29734-68-7 | |
| Chemical compound, drug | SR 95531 hydrobromide (Gabazine) | Hello Bio | CAS: 104104-50-9 | |
| Chemical compound, drug | DNQX | TOCRIS | CAS: 2379-57-9 | |
| Chemical compound, drug | D-AP5 | Hello Bio | CAS:79055-68-8 | |
| Chemical compound, drug | Methyllycaconitine citrate | TOCRIS | Lot # 23A/255947 CAS:351344-10-0 | |
| Chemical compound, drug | Naspm trihydrochloride | Alomone Labs | CAS: 1049731-36-3 | |
| Chemical compound, drug | CGP 55845 hydrochloride | Hello Bio | CAS: 149184-22-5 | |
| Chemical compound, drug | XYLAZINE AS HYDROCHLORIDE | EUROVET ANIMAL HEALTH B.V | CAS: 082-91-92341-00 | |
| Chemical compound, drug | CLORKETAM | VETOQUINOL | CAS: 1867-66-9 | |
| Chemical compound, drug | VECTASHIELD Vibrance Antifade Mounting Medium with DAPI | VECTOR LABORATORIES | SKU: H-1800 | |
| Antibody | Recombinant Anti-GAD65+GAD67 antibody [EPR19366] | Abcam | ab183999 | Rabbit monoclonal (1:1000) |
| Antibody | Rabbit polyclonal anti Somatostatin-14 | Peninsula Laboratories | T-4102 | Rabbit polyclonal (1:100) |





