Insulin sensitivity in mesolimbic pathways predicts and improves with weight loss in older dieters
Figures
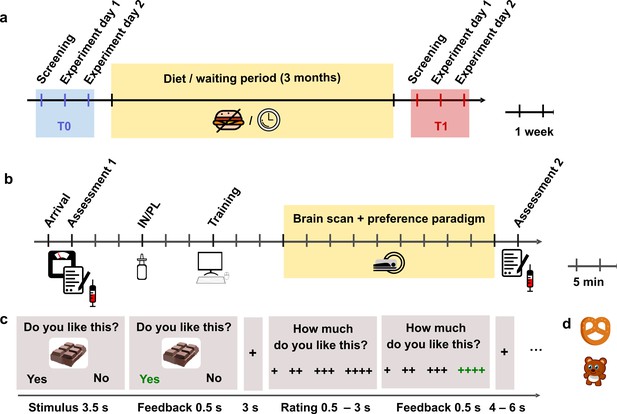
Outline of the study design and experimental task.
(a) Timeline of the longitudinal design. Each participant attended four fMRI sessions, two each before and after the 3-month intervention interval. (b) Protocol of the experimental MRI sessions. (c) Timing of the fMRI paradigm. Example of a high sugar food trial. (d) Examples of low-sweet food and non-food items.
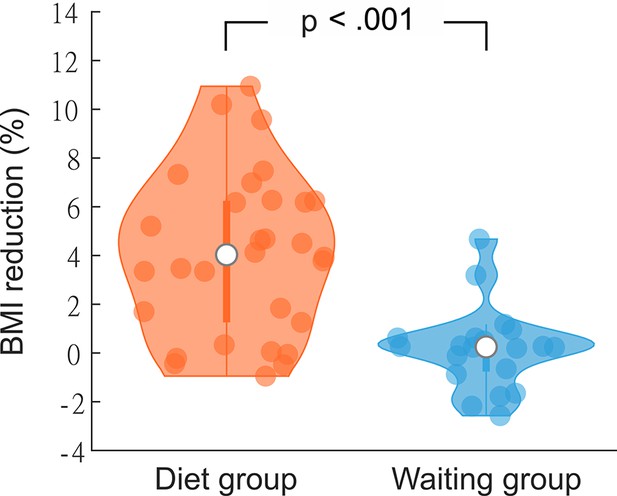
Percentage body mass index (BMI) change after 3 months in the diet (N = 30) and the waiting (N = 20) group.
Violin plots show individual data, median, interquartile range, and 1.5× interquartile range.
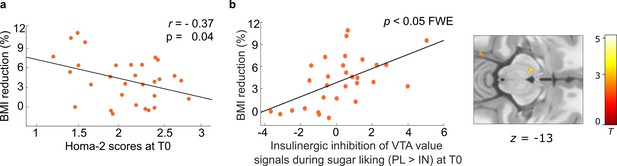
Predictors of subsequent weight loss in dieters.
(a) Higher peripheral insulin sensitivity measured via Homa-2 scores at baseline (T0) was related to higher percentage body mass index (BMI) reduction in dieters (N = 30) at follow-up (T1). (b) Correlation between insulin effects in the ventral tegmental area (VTA) at baseline and subsequent weight loss in dieters. Individual blood oxygenation level-dependent (BOLD) signals were extracted from the peak voxel in the VTA resulting from the parametric contrast PLHS>LS > INHS>LS within dieters, p < 0.05 FWE corrected for bilateral VTA mask.
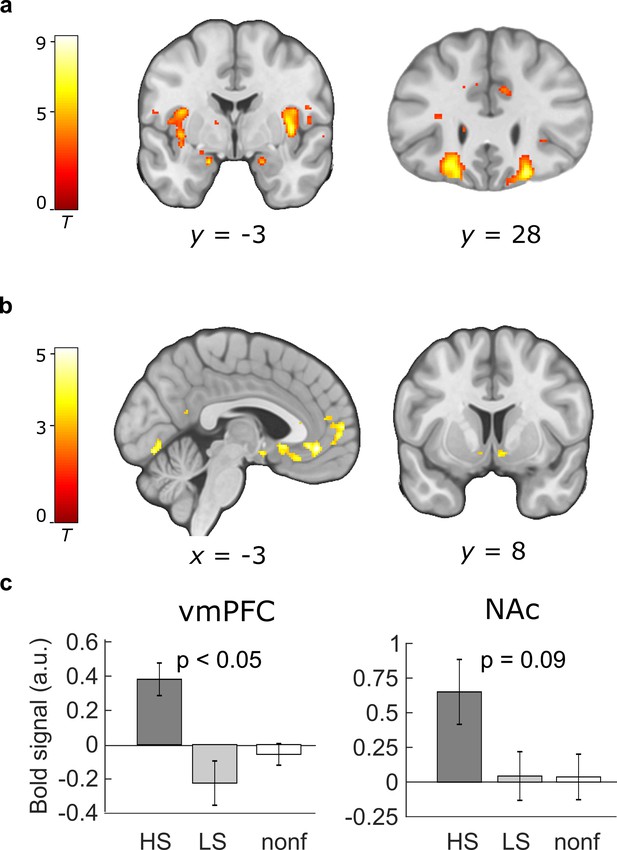
Paradigm-induced activation patterns during baseline placebo.
(a) Categorical effect of food stimulus presentation. Greater activity in the insula, amygdala, and orbitofrontal cortex was observed in the food compared to the non-food condition across both groups (N = 50; included contrast images: food > non-food). (b) Neural representation of preference values (parametric analysis). Regions in which the correlation with preference values was significant across participants included the ventromedial prefrontal cortex (vmPFC) and the bilateral nucleus accumbens (NAc) (included contrast images: all food × liking). (c) Sugar-specific blood oxygenation level-dependent (BOLD) signals in the vmPFC and right NAc. Bar plots show means and standard error of the mean (SEM) of contrast estimates extracted from peak voxels from the comparison HS > LS (included contrast images: HS × liking > LS × liking). All peaks and displayed p values are p < 0.05 FWE corrected. Activations are overlaid on a custom template (display threshold p < 0.005 uncorrected).
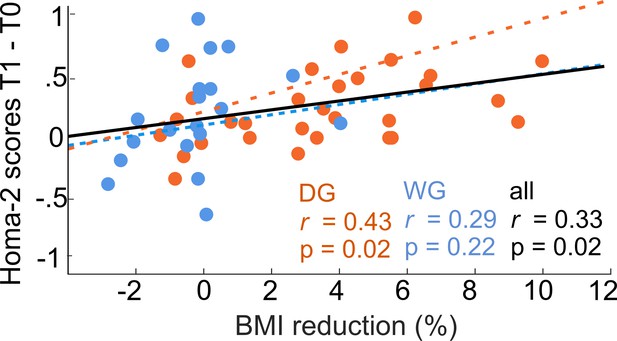
Weight loss is related to improvements in peripheral insulin sensitivity.
Within dieters (N = 30) and across participants (N = 50) a higher percentage of body mass index (BMI) changes was correlated with an increase of insulin sensitivity as measured via the Homa-2 scores.
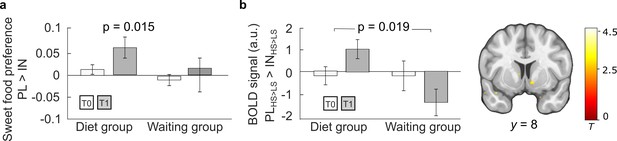
Central insulin effects on behavior and brain activity before and after 3 months.
(a) Behavioral insulin effects on sweet food preference. While there was no insulin effect observed at baseline T0 in both groups, the percentage of preferred sweet food items decreased significantly under insulin compared to placebo at follow-up in dieters. (b) General linear modeling of sweet versus non-sweet value signals under insulin compared to placebo revealed a significantly stronger signal decrease in the diet group (N = 30) compared to the waiting group (N = 20) at follow-up (T1) compared to baseline (T0) in the right nucleus accumbens (NAc) included contrast images: T1 [PLHS>LS > INHS>LS] > T0 [PLHS>LS > INHS>LS]. Bar plots show group means and standard error of the mean (SEM) of mean contrast estimates extracted from significant peak voxel. p < 0.05 FWE corrected for bilateral NAc mask. Activations are overlaid on a custom template (display threshold p < 0.005 uncorrected).
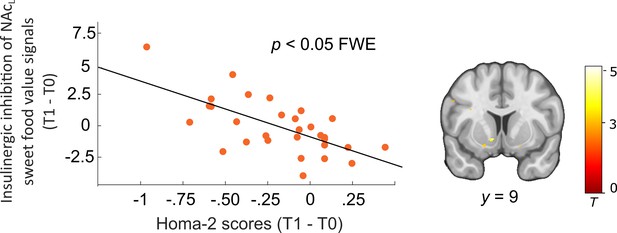
Interaction between peripheral and central insulin changes in dieters (N = 30).
Improved (T1 > T0) insulinergic inhibition of sweet food value signals in the left nucleus accumbens (NAc) correlated with improved peripheral insulin sensitivity. Individual blood oxygenation level-dependent (BOLD) signals were extracted from the peak voxel in the right NAc resulting from simple regression analysis including the parametric contrast HS > LSPL>IN_post > HS > LSPL>IN_pre and HOMA-2 changes (post > pre) as covariate of interest. p < 0.05 FWE corrected for bilateral NAc mask. Activations are overlaid on a custom template (display threshold p < 0.005 uncorrected).
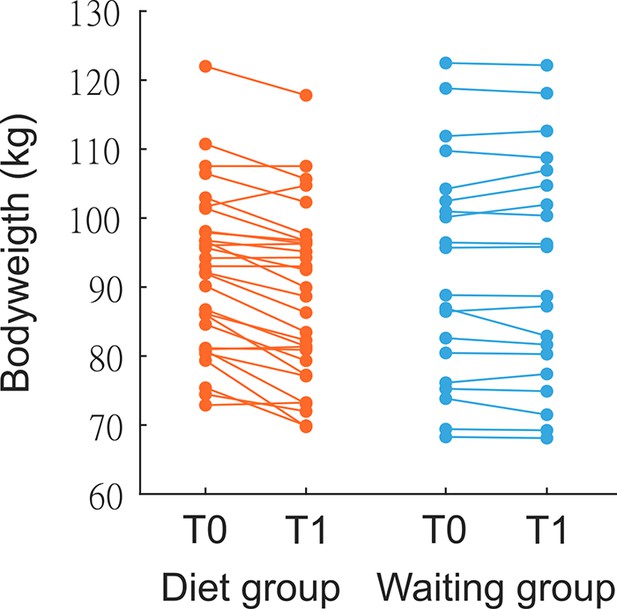
Individual weight change in dieters and controls.
Weight change in kg after 3-month dietary intervention/waiting phase.
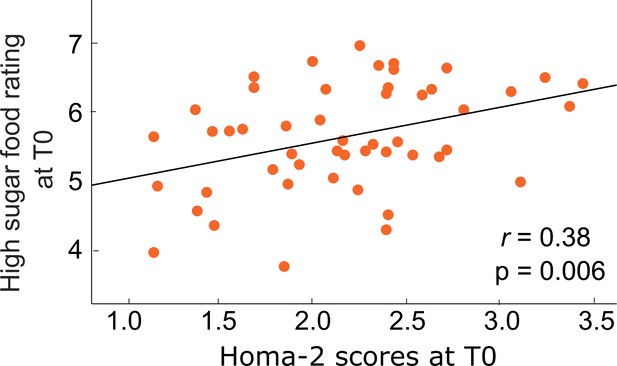
Correlation between peripheral insulin sensitivity and sweet food liking.
Lower insulin sensitivity as measured via the HOMA-2 score was related to higher sugar liking. No correlation with insulin sensitivity was found for low sugar liking (p > 0.17).
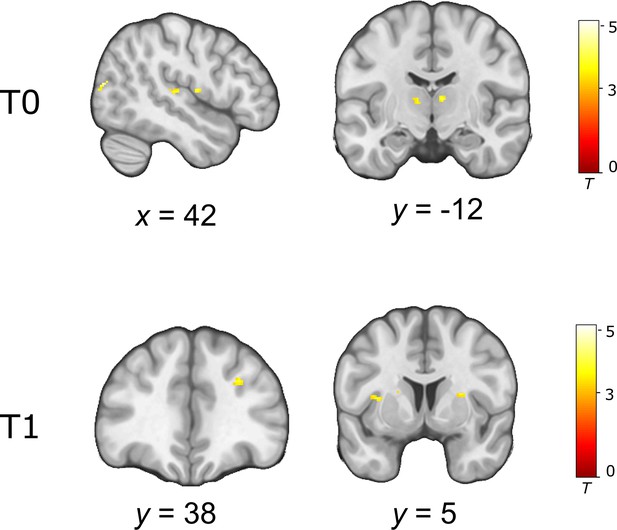
Uncorrected whole-brain response to insulin (HS > LS) at T0 and T1 across all participants.
Activations are overlaid on a custom template (display threshold p < 0.001, k = 10).
Tables
Sample characteristics at baseline and follow-up.
| DG (N = 30) | WG (N = 20) | ||||||
|---|---|---|---|---|---|---|---|
| T0 | T1 | p time | T0 | T1 | p time | p time × group | |
| BMI (kg/m²) | 32.1 (0.7) | 30.8 (0.6) | *** | 32.8 (1.1) | 32.8 (1.2) | N.S. | *** |
| Waist (cm) | 103.7 (1.8) | 98.1 (1.9) | *** | 103.1 (2.7) | 99.7 (2.8) | N.S. | N.S. |
| Bodyfat | 37.4 (1.3) | 36.7 (1.4) | N.S. | 39.5 (1.6) | 39.0 (1.7) | N.S. | N.S. |
| Blood | |||||||
| HOMA-2 | 2.1 (0.1) | 1.9 (0.1) | * | 2.3 (0.2) | 2.3 (0.1) | N.S. | N.S. |
| Glucose (mmol/l) | 5.5 (0.1) | 5.4 (0.1) | N.S.1 | 5.7 (0.1) | 5.8 (0.1) | N.S.1 | N.S.² |
| Insulin (pmol/l) | 78.8 (4.0) | 70.3 (4.4) | + | 95.5 (8.7) | 94.2 (7.7) | N.S. | N.S. |
| C-peptide (nmol/l) | 0.9 (0.03) | 0.8 (0.04) | * | 1.1 (0.06) | 1.0 (0.05) | N.S. | N.S. |
| HbA1C | 5.4 (0.03) | 5.4 (0.04) | N.S. | 5.5 (0.07) | 5.5 (0.07) | N.S. | N.S. |
-
***p < 0.001, *p < 0.05, +p < 0.10, s.e.m. in parantheses
-
DG, diet group; WG, waiting group; PL, placebo; IN, insulin; T0, baseline; T1, follow-up; 1 Wilcoxon-rank Test; ² Mann-Whitney-U-Test.
-
BMI, body mass index; HOMA-2, c-peptide-based Homeostatic Model Assessment for Insulin Resistance; N.S, not significant.
Fasting duration before each study day and hunger ratings before each MRI scan.
| WG | T0 | p | T1 | p | p | ||
|---|---|---|---|---|---|---|---|
| PL | IN | (session) | PL | IN | (session) | (session × time) | |
| Fasting time (hr) | 12.4 (0.3) | 12.8 (0.4) | N.S. | 12.5 (0.3) | 12.6 (0.4) | N.S. | N.S. |
| Hunger rating | 2.5 (0.6) | 2.2 (0.5) | N.S. | 2.8 (0.5) | 3.1 (0.7) | N.S. | N.S. |
| DG | T0 | p | T1 | p | p | ||
| PL | IN | (session) | PL | IN | (session) | (session × time) | |
| Fasting time (hr) | 12.3 (0.3) | 12.2 (0.3) | N.S. | 12.4 (0.3) | 12.3 (0.3) | N.S. | N.S. |
| Hunger rating | 2.2 (0.4) | 2.3 (0.4) | N.S. | 2.9 (0.5) | 2.8 (0.5) | N.S. | N.S. |
-
Neither did the fasting times between the last food intake and the beginning of the study day differ between sessions (PL/IN, T0/T1) or groups, nor was there a group x session effect. Before entering the scanner, participants rated their current feelings of hunger on a scale from 0 (“not hungry at all”) to 10 (“extremely hungry”). Values did not differ between sessions or groups and there was no group x session interaction. Values indicate means with s.e.m. in parentheses. DG: diet group, WG: waiting group, PL: placebo, IN = insulin, T0: baseline, T1: follow-up. N.S. not significant. 1 Because at least one measure of this variable (across all time-points) was not normally distributed, a Wilcoxon-Rank-Testing was applied.
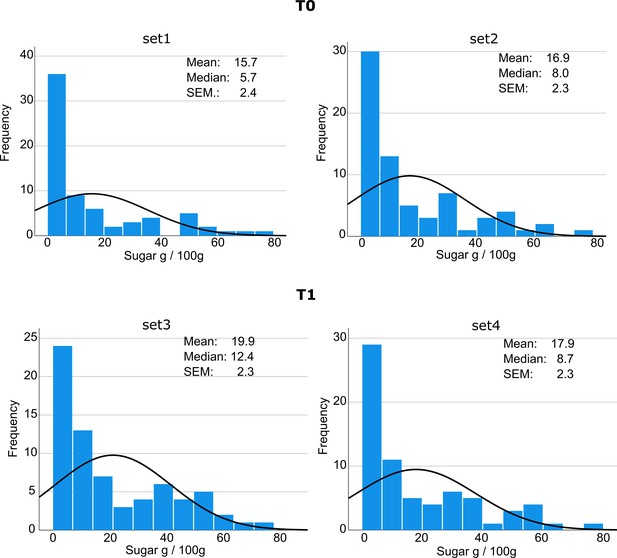
Stimuli characteristics.
| Set 1 | Set 2 | Set 3 | Set 4 | p | |
|---|---|---|---|---|---|
| Parametric liking score All food items | 2.86 (0.07) | 2.90 (0.07) | 2.98 (0.06) | 2.96 (0.06) | N.S. |
| HS food items | 3.00 (0.09) | 2.80 (0.10) | 3.03 (0.08) | 2.87 (0.08) | N.S. |
| LS food items | 2.70 (0.09) | 3.00 (0.10) | 2.91 (0.10) | 3.04 (0.10) | N.S. |
| Picture saliency All food items | 0.19 (0.01) | 0.21 (0.01) | 0.18 (0.01) | 0.20 (0.01) | N.S. |
| HS food items | 0.19 (0.01) | 0.21 (0.01) | 0.18 (0.01) | 0.20 (0.01) | N.S. |
| LS food items | 0.20 (0.01) | 0.20 (0.01) | 0.21 (0.01) | 0.20 (0.01) | N.S. |
-
In a validation study, an independent sample of 16 participants rated the preference of items on a scale from 1 (~ “I do not like this at all”) to 4 (~ “I like this very much”). Saliency is calculated based on the Image Signature algorithm, as described by Hou et al., 2012. One-way ANOVAs showed that the four sets did not differ in regard to the parametric liking (all P > 0.07) and saliency (all P > 0.43) scores across food items and for HS and LS food items separately. Values indicate means with s.e.m. in parentheses. N.S. not significant.
Regression models for the prediction of dietary success.
| Dependent variable | Model (R² adjusted) | Predictor variable (standardized β-coefficients) | ||
|---|---|---|---|---|
| Insulin sensitivity (HOMA-2) | Insulin effects on VTA signal | BMI | ||
| Model 1 | ||||
| BMI change (%) | 0.39*** | −0.35*c | 0.54***c | |
| Model 2 | ||||
| BMI change (%) | 0.37** | −0.38* | 0.51**c | 0.07 |
-
*
**p < 0.001, **p < 0.01, *p < 0.05, csignificant after Bonferroni correction.
Pre–post blood values at baseline and follow-up.
| DG_T0 | PL | p | IN | p | p | ||
|---|---|---|---|---|---|---|---|
| Pre | Post | Pre | Post | (interaction) | |||
| Insulin (pmol/l) | 78.8 (4.0) | 62.8 (4.6) | 0.003 | 80.6 (4.5) | 72.9 (6.0) | 0.16 | N.S. |
| Glucose (mmol/l) | 5.5 (0.1) | 5.6 (0.1) | N.S. | 5.4 (0.1) | 5.4 (0.1) | N.S. | N.S. |
| DG_T1 | |||||||
| Insulin (pmol/l) | 70.3 (4.4) | 57.7 (3.8) | 0.001 | 72.9 (4.9) | 62.6 (4.9) | 0.036 | N.S. |
| Glucose (mmol/l) | 5.4 (0.1) | 5.4 (0.1) | N.S.1 | 5.5 (0.1) | 5.3 (0.1) | 0.02 | N.S.1 |
| WG_T0 | PL | p | IN | p | p | ||
| Pre | Post | Pre | Post | (interaction) | |||
| Insulin (pmol/l) | 97. 9 (8.6) | 70.2 (8.1) | 0.001 | 88.6 (7.4) | 70.2 (6.5) | 0.001 | N.S. |
| Glucose (mmol/l) | 5.7 (0.1) | 5.7 (0.1) | N.S. | 5.7 (0.1) | 5.6 (0.1) | N.S. | N.S. |
| WG_T1 | |||||||
| Insulin (pmol/l) | 97.3 (7.9) | 65.3 (6.0) | 0.001 | 103.0 (8.9) | 80.5 (7.8) | 0.001 | N.S. |
| Glucose (mmol/l) | 5.8 (0.1) | 5.7 (0.1) | N.S. | 5.9 (0.1) | 5.7 (0.1) | 0.02 | N.S. |
-
Blood samples were sampled after arrival and after completion of the scanning sessions (see Figure 1b). There was a significant insulin level x session interaction across participants at T0 (F(1,49) = 4.1; P = .047, rmANOVA) driven by a stronger insulin decrease in the placebo session. This effect was not significant within single groups, in interaction with groups, nor were there any significant session effects at T1 (all P > .30). Values indicate means with s.e.m. in parentheses. DG: diet group, WG: waiting group, PL: placebo, IN = insulin, T0: baseline, T1: follow-up. N.S. not significant. 1 Because that at least one measure of this variable (across all time-points) was not normally distributed, a Wilcoxon-Rank-Testing was applied.