NFATc1 marks articular cartilage progenitors and negatively determines articular chondrocyte differentiation
Figures
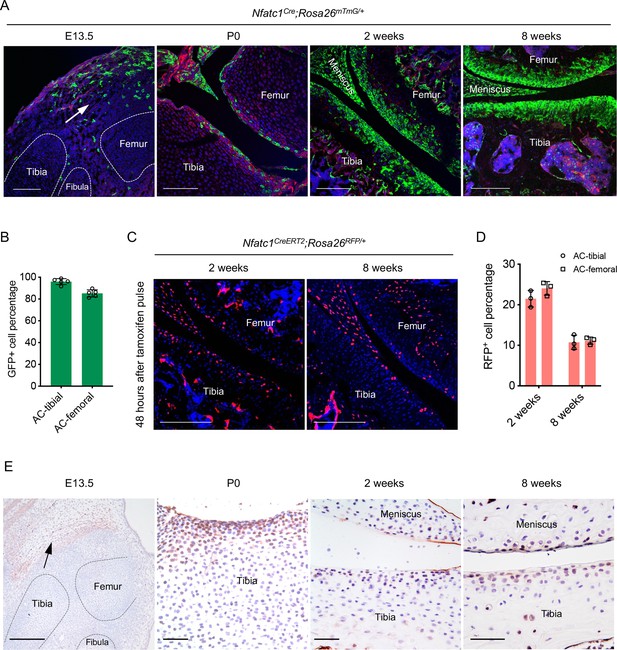
Articular cartilage is derived from NFATc1-expressing progenitors.
(A) Confocal microscopy images showing the distribution of GFP+ cells during articular cartilage development at the knee of Nfatc1Cre;Rosa26mTmG/+ mice (n=5 animals for each age, two knee joints per animal). Arrow indicating the main location of GFP+ cells at the knee at embryonic day 13.5 (E13.5). P0, postnatal day 0. (B) Quantification of GFP+ cells in the articular cartilage of Nfatc1Cre;Rosa26mTmG/+ mouse knee at 8 weeks of age (n=5 animals, one knee joint per animal). AC, articular cartilage. (C) Representative confocal images demonstrating the distribution of RFP+ cells in the articular cartilage at 2 weeks and 8 weeks of age in Nfatc1CreERT2;Rosa26RFP/+ mice 48 hrs after tamoxifen pulse for 5 consecutive days (n=3 mice for each age, two knee joints per animal). (D) Quantification of RFP+ cells in the articular cartilage of Nfatc1CreERT2;Rosa26RFP/+ mouse knee at 2 weeks and 8 weeks of age (n=3 mice for each age, one knee joint per animal). (E) Immunohistochemistry detecting the expression of NFATc1 during mouse articular cartilage development (n=3 mice for each age, two knee joints per animal). Data are mean ± SD of results from five or three animals; scale bars, 200 μm except for the right three images in (E), 50 μm.
-
Figure 1—source data 1
Quantification data for GFP+ or RFP+ cells in articular cartilage.
For Figure 1B Quantification of GFP+ cells in articular cartilage of Nfatc1Cre;Rosa26mTmG/+ mouse knee at 8 weeks of age (%); Figure 1D Quantification of RFP+ cells in articular cartilage of Nfatc1CreERT2;Rosa26RFP/+ mouse knee at 2 weeks and 8 weeks of age after tamoxifen pulse for 5 consecutive days (%).
- https://cdn.elifesciences.org/articles/81569/elife-81569-fig1-data1-v2.xlsx
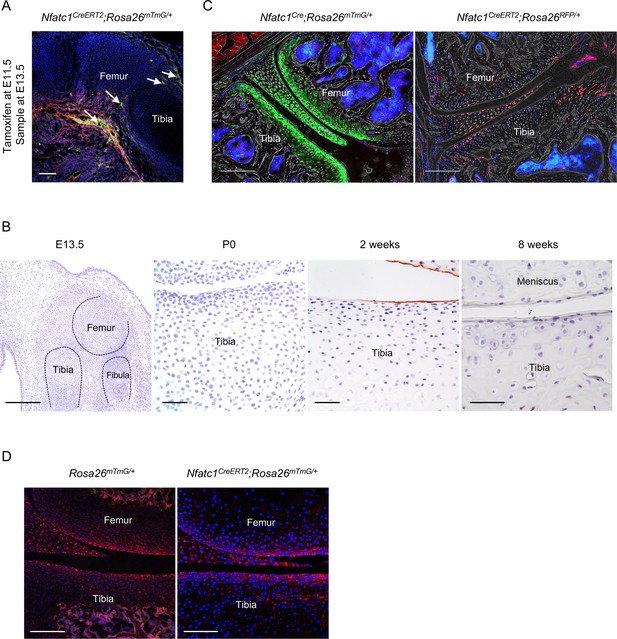
Track NFATc1-expressing progenitors during articular cartilage development.
(A) Representative confocal image showing the distribution of GFP+ cells at the prospective joint site of Nfatc1CreERT2;Rosa26mTmG/+ mice at E13.5 (n=5 mouse embryos). Tamoxifen was administrated to pregnant dams at E11.5 and mouse limbs were sampled at E13.5. Arrows indicating locations of GFP+ cells. (B) Histological images displaying immunostaining of IgG isotype control at different ages of mouse knee development (n=3 mice each age, two knee joints per animal, related to Figure 1E) (C) Representative confocal images showing the distribution of GFP+ or RFP+ cells in articular cartilage of Nfatc1Cre;Rosa26mTmG/+ (n=5, two knee joints for each) or Nfatc1CreERT2;Rosa26RFP/+ (n=3, two knee joints for each) mice respectively at 8 weeks of age. (D) Confocal images showing the absence of GFP+ cells in articular cartilage of Rosa26mTmG/+ control mice and Nfatc1CreERT2;Rosa26mTmG/+ mice without tamoxifen induction (n=3 animals for each). Scale bars, 200 μm except for the right three images in (B), 50 μm.
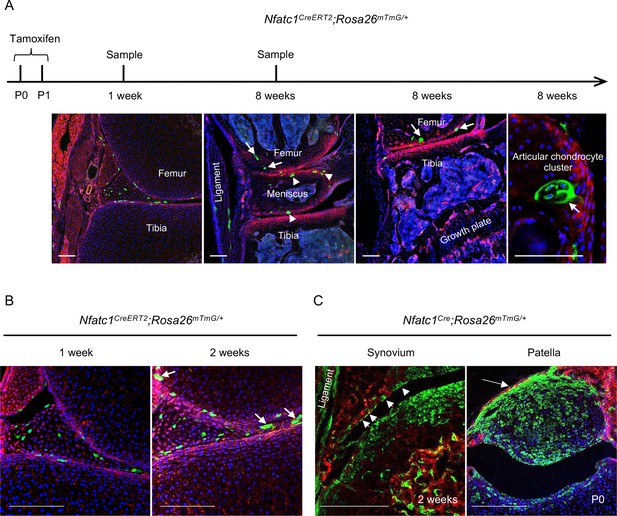
NFATc1-expressing progenitors form cell clusters with articular cartilage development and contribute to the meniscus and articular synovium formation.
(A) Confocal microscopy images showing the distribution of GFP+ cells in the articular cartilage of Nfatc1CreERT2;Rosa26mTmG/+ mouse knee at 1 week and 8 weeks after administering tamoxifen to dams at P0 and P1. The most left image showing GFP+ cells in articular tissues after 1 week of tamoxifen administration. The right three images demonstrating GFP+ cell clusters in the articular cartilage (arrows), meniscus (arrowheads), and ligament. (B) Representative confocal images displaying GFP+ cells or cell clusters (arrows) in the meniscus and articular synovium at 1 week or 2 weeks after tamoxifen administration to dams at P0 and P1. (C) Confocal microscopy images demonstrating that GFP+ cells contribute to the formation of the ligament, synovial lining (left image, arrowheads, 2 weeks of age), and the patella of the knee (right image, arrow, P0) in Nfatc1Cre;Rosa26mTmG/+ mice. All images are representative of five mice at each time point or age, two knee joints per animal. Scale bars, 200 μm.
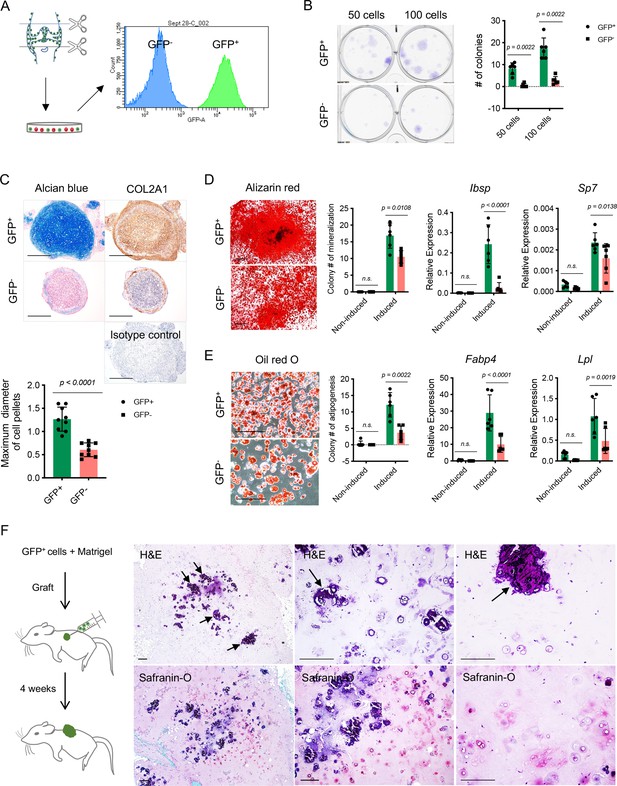
Colony formation and multipotent differentiation of Nfatc1-expressing progenitors.
(A) Schematic diagram showing culturing and sorting GFP+ and GFP- cells from the knee of neonatal Nfatc1Cre;Rosa26mTmG/+ mice. (B) Colony formation assay of GFP+ and GFP- cells with 50 or 100 cells plated in 6-well-plates and cultured for 2 weeks. n=6 with cells from three animals, two replicates for each, nonparametric Mann-Whitney test, experiment repeated twice. (C) Alcian blue staining and immunohistochemistry of COL2A1 showing the chondrogenic potential of GFP+ and GFP- cell pellets after being cultured in the chondrogenic differentiation medium for 3 weeks. Isotype as a negative control for COL2A1 antibody. The maximum diameter of cell pellets reflecting the proliferative capacity of GFP+ and GFP- cells. n=9 with cells from three animals, three replicates for each. (D) Alizarin red staining and gene expression analysis of Ibsp and Sp7 demonstrating the osteogenic potential of GFP+ and GFP- cells after being cultured in the osteogenic differentiation medium for 4 weeks. n=6 with cells from three animals, two replicates for each, nonparametric Mann-Whitney test for colony counting data, two-way ANOVA followed by Sidak’s tests for gene expression data, experiments repeated twice. (E) Oil red O staining and gene expression analysis of Fabp4 and Lpl displaying adipogenesis in GFP+ and GFP- cells after being cultured in the adipogenic differentiation medium for 10 days. n=6 with cells from three animals, two replicates for each, nonparametric Mann-Whitney test for colony counting data, two-way ANOVA followed by Sidak’s tests for gene expression data, experiments repeated twice. (F) Schematic illustration and histology respectively showing transplantation of GFP+ cells along with Matrigel matrix underneath the dorsal skin of severe combined immune-deficient mice and the formation of chondrocytes, chondrocyte clusters, and hypertrophic cartilage-like structure (arrows) 4 weeks later. Images are representative of six animals, with GFP- cells as the control (results shown in Figure 3—figure supplement 1B). All data are mean ± SD. Scale bars, 400 μm (C), 500 μm (D), 200 μm (E, F).
-
Figure 3—source data 1
Data of colony numbers, cell pellet diameters, and qPCR.
For Figure 3B Colony formation assay of GFP+ and GFP- cells with 50 or 100 cells plated in 6-well-plates and cultured for 2 weeks (colony numbers); Figure 3C Maximum diameter of cell pellets (mm); Figure 3D Colony numbers of mineralization, Relative gene expression of Ibsp (2-△Ct) and Relative gene expression of Sp7 (2-△Ct); Figure 3E Colony numbers of adipogenesis, Relative gene expression of Fabp4 (2-△Ct), and Relative gene expression of Lpl (2-△Ct).
- https://cdn.elifesciences.org/articles/81569/elife-81569-fig3-data1-v2.xlsx
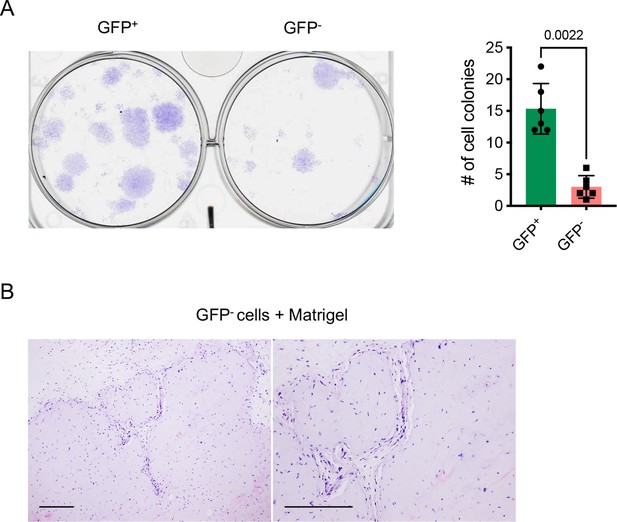
Colony formation assay and in vivo transplantation of GFP- cells.
(A) Colony formation assay of GFP+ and GFP- cells at P5 with 100 cells plated in 6-well-plates and cultured for 2 weeks (n=6 with cells from 3 mice, two replicates for each, nonparametric Mann-Whitney test). (B) Representative histology images demonstrating H&E staining of transplants of GFP- cells with Matrigel matrix underneath the dorsal skin of severe combined immune-deficient mice for 4 weeks. n=6 animals; scale bars, 200 μm.
-
Figure 3—figure supplement 1—source data 1
For Figure 3—figure supplement 1A Colony formation assay of GFP+ and GFP- cells at P5 with 100 cells plated in 6-well-plates and cultured for 2 weeks (colony numbers).
- https://cdn.elifesciences.org/articles/81569/elife-81569-fig3-figsupp1-data1-v2.xlsx
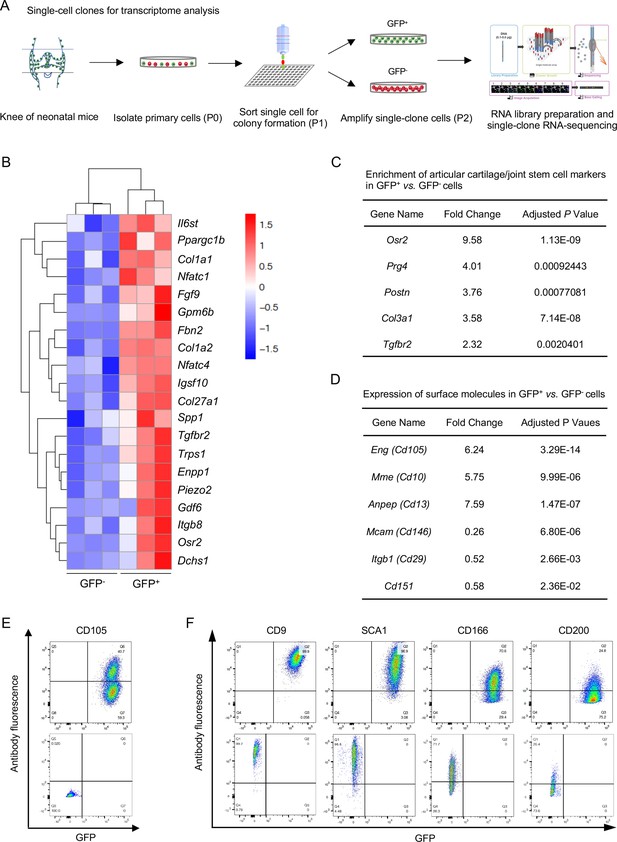
Transcriptional profile of Nfatc1-expressing articular cartilage progenitors.
(A) Schematic diagram showing the process of sorting single-clone cells for RNA-sequencing. (B) Cluster heatmap displaying 20 high-expressing genes associated with articular cartilage development in GFP+ vs. GFP- cells. Color descending from red to blue indicates log10(FPKM +1) from large to small. n=3 with cells from three animals in each group. (C) Transcriptome analysis revealing the enrichment of previously reported articular cartilage progenitor marker genes Osr2, Prg4, Postn, Col3a1, and Tgfbr2 in GFP+ relative to GFP- cells. (D) Transcriptome analysis identifying high expression of Cd105, Cd10, and Cd13 and low expression of Cd146, Cd29, and Cd151 in GFP+ vs. GFP- cells. (E) Flow cytometry verifying the expression of CD105 in GFP+ relative to GFP- cells. (F) Flow cytometry showing the expression of cell surface molecules CD9, SCA1, CD166, and CD200 in GFP+ and GFP- cells. Representative results of cells from three mice, experiment repeated twice.
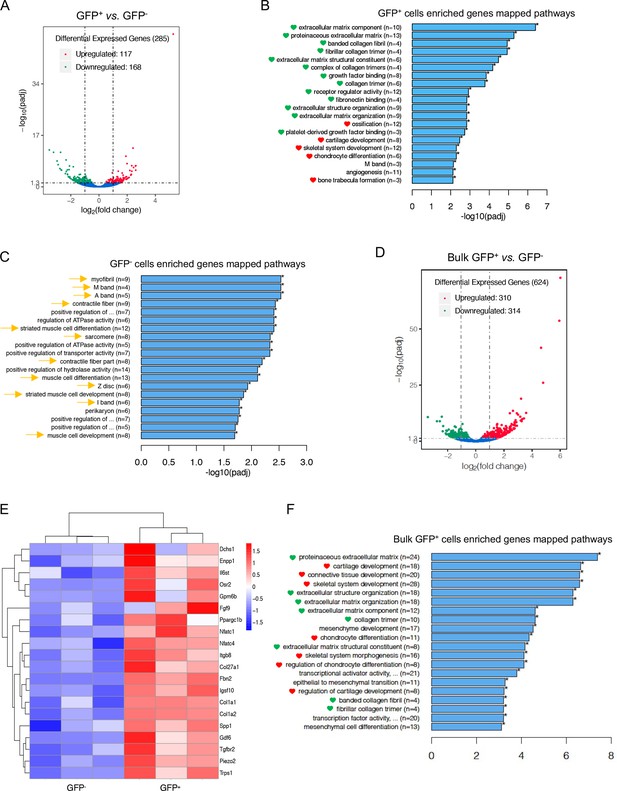
Transcriptional profile of Nfatc1-expressing articular cartilage progenitors.
(A) Volcano diagram from transcriptome analysis of single clone-derived cells showing the distribution of differential expression genes in GFP+ vs. GFP- cells. The threshold of differential expression genes is padj <0.05. (B) Histogram demonstrating top 20 significantly enriched pathways of high-expressing genes in GFP+ relative to GFP- cells by GO enrichment analysis. Green and red hearts referring to pathways related to extracellular matrix biology and skeletal system development, respectively. (C) Histogram demonstrating top 20 significantly enriched pathways of enriched genes in GFP- vs. GFP+ cells by GO enrichment analysis. Yellow arrows indicating pathways related to skeletal muscle development. (D) Volcano diagram from transcriptome analysis of bulk primary GFP+ cells showing the distribution of differential expression genes in GFP+ vs. GFP- cells. padj <0.05 as the threshold of differential expression genes. (E) Cluster heatmap of RNA-sequencing results from bulk primary GFP+ cells verifying the 20 high-expressing genes associated with skeletal and cartilage development in GFP+ vs. GFP- cells. Color descending from red to blue indicates log10(FPKM +1) from large to small. (F) Histogram demonstrating top 20 significantly enriched pathways of high-expressing genes in bulk primary GFP+ cells relative to GFP- cells by GO enrichment analysis. Green and red hearts as indicated in (B). n=3 with cells from three different animals in each group. The n-numbers in (B), (C), and (F) indicating the number of differentially expressed genes concerning the GO term.
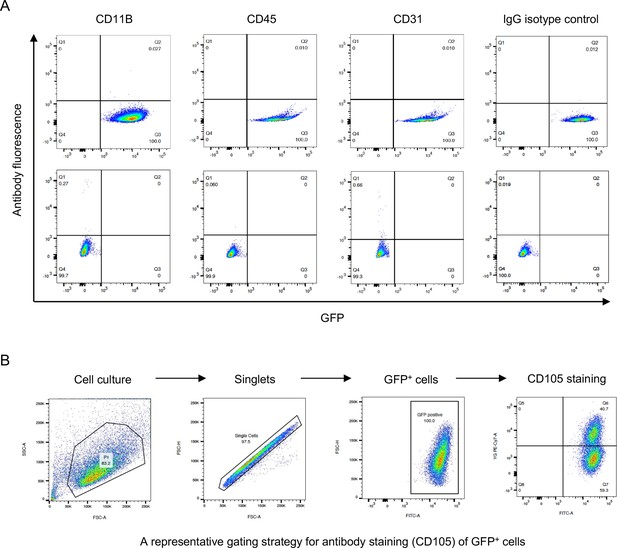
Flow cytometry determining the expression of cell surface markers.
(A) Expression of CD11B, CD45, and CD31 in GFP+ and GFP- cells from neonatal Nfatc1Cre;Rosa26mTmG/+ mouse knee, IgG isotype as the negative control. Representative results of cells from three animals, experiment repeated twice. (B) A representative gating strategy for flow cytometry.
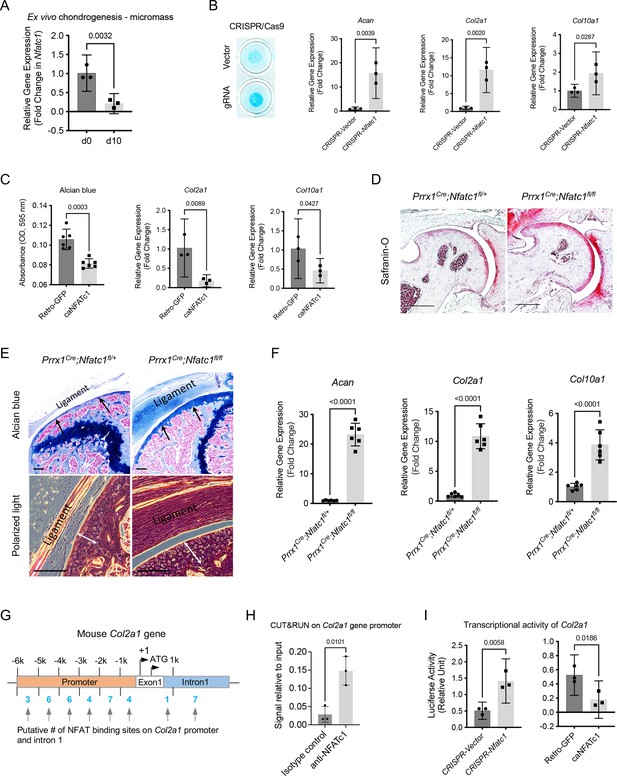
NFATc1 negatively determines articular chondrocyte differentiation through regulating Col2a1 gene transcription.
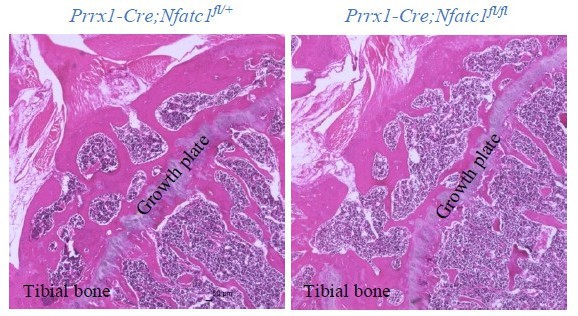
(A) Gene expression analysis displaying the change of Nfatc1 expression in GFP+ cell-micromass cultured in the chondrogenic medium for 10 days. n=3, experiment repeated three times with cells from three mice. (B) Alcian blue staining and gene expression analysis of Acan, Col2a1, and Col10a1 after deleting Nfatc1 by CRISPR/CAS9 technique in ex vivo micromass culture of GFP+ cells from neonatal Nfatc1Cre;Rosa26mTmG/+ mice (day 7, without chondrogenic induction, n=3). Experiment repeated three times with cells from three animals. (C) Quantification of alcian blue staining and gene expression analysis of Col2a1 and Col10a1 showing decreased chondrogenesis after overexpressing NFATc1 in GFP+ cells by infecting a caNFATc1 retrovirus structure. For alcian blue staining, n=6 with cells from three animals, two replicates for each; for gene expression analysis, n=3, experiment repeated twice with cells from two animals. (D) Safranin O staining demonstrating enhanced articular cartilage staining in the hip of Prrx1Cre;Nfatc1fl/fl vs. Prrx1Cre;Nfatc1fl/+ mice at 12 weeks of age. Representative images from five animals in each group were displayed. (E) Representative images of alcian blue staining and polarized light on H&E staining manifesting increased staining (arrows) and thickness of articular cartilage (double arrows) in the knee of Prrx1Cre;Nfatc1fl/fl relative to Prrx1Cre;Nfatc1fl/+ mice at 16 weeks of age. n=5 animals for each group. (F) Quantitative PCR determining the expression of Acan, Col2a1, and Col10a1 genes in articular cartilage of Prrx1Cre;Nfatc1fl/fl relative to Prrx1Cre;Nfatc1fl/+ mice at 8 weeks of age. n=6 animals for each group. (G) Computational screening of NFAT binding sites on mouse Col2a1 promoter and intron 1 sequences by PROMO software recognizing 38 putative NFAT binding sites across –6 k bp of the promoter and intron 1. The transcriptional starting site is counted as +1. Location is given in bp relative to the transcriptional starting site. (H) Cleavage under targets and release using nuclease (CUT&RUN)-qPCR showing the binding of NFATc1 to the promoter region of mouse Col2a1 (chr15: 98004609–98004620, mm10). (I) Luciferase assay of transcriptional activity of Col2a1 after deleting or overexpressing NFATc1 in ATDC5 cells. n=3, experiment repeated three times. All data shown as mean ± SD, two-tailed Student’s t-test performed. Scale bar, 200 μm (D), 100 μm (E).
-
Figure 5—source data 1
Data of qPCR and luciferase assay.
For Figure 5A Relative gene expression of Nfatc1 (2-△△Ct); Figure 5B Relative gene expression of Acan, Col2a1, and Col10a1 (2-△△Ct); Figure 5C Quantification of alcian blue staining, Relative gene expression of Col2a1 and Col10a1 (2-△△Ct); Figure 5F Relative gene expression of Acan, Col2a1, and Col10a1 (2-△△Ct); Figure 5H CUT&RUN signal relative to input (Col2a1); Figure 5I Luciferase assay of transcriptional activity of Col2a1 after deleting or overexpressing NFATc1 in ATDC5 cells (relative unit).
- https://cdn.elifesciences.org/articles/81569/elife-81569-fig5-data1-v2.xlsx
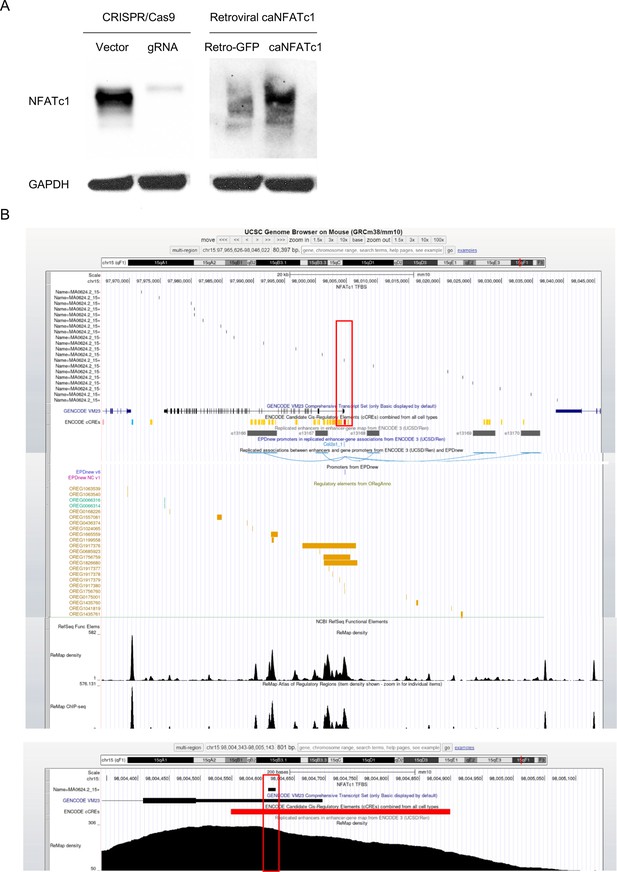
Western blot determining NFATc1 expression and NFATc1 binding site at the Col2a1 gene promoter by FIMO analysis.
(A) Western blot determining the deletion or overexpression of NFATc1 by lentiviral CRISPR/CAS9 or retroviral caNFATc1, respectively. The empty vector without a gRNA sequence was used as a control for CRISPR/CAS9 lentivirus production and infection. The pMSCV-GFP vector (Retro-GFP) was utilized as a control for caNFATc1 retrovirus production and infection. (B) FIMO output visualized in the UCSC Genome Browser showing one of the 20 NFATc1 motif occurrences localized in the Col2a1 promoter region (marked with red rectangles). The lower image with the red rectangle is a zoom-in of the upper one.
-
Figure 5—figure supplement 1—source data 1
Raw uncropped blots.
- https://cdn.elifesciences.org/articles/81569/elife-81569-fig5-figsupp1-data1-v2.zip
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Genetic reagent (Mus. musculus) | Nfatc1tm1.1(cre)Bz; Nfatc1Cre | PMID:23178125 | RRID:MGI:5471107 | Dr. Bin Zhou (Albert Einstein College of Medicine) |
| Genetic reagent (M. musculus) | Nfatc1tm1.1(cre/ERT2)Bzsh; Nfatc1CreERT2 | PMID:24994653 | RRID:MGI:5637438 | Dr. Bin Zhou (Shanghai Institutes for Biological Sciences) |
| Genetic reagent (M. musculus) | B6(Cg)-Nfatc1tm3Glm/AoaJ; Nfatc1fl | The Jackson Laboratory | Strain #:022786 RRID:IMSR_JAX:022786 | |
| Genetic reagent (M. musculus) | B6.Cg-Tg(Prrx1-cre)1Cjt/J; Prrx1Cre | The Jackson Laboratory | Strain #:005584 RRID:IMSR_JAX:005584 | |
| Genetic reagent (M. musculus) | B6.129(Cg)- Gt(ROSA)26 Sortm4(ACTB-tdTomato,-EGFP)Luo; Rosa26mTmG; mTmG | The Jackson Laboratory | Strain #:007676 RRID:IMSR_JAX:007676 | |
| Genetic reagent (M. musculus) | B6.Cg- Gt(ROSA)26Sortm9(CAG-tdTomato)Hze; Rosa26RFP; Ai9 | The Jackson Laboratory | Strain #:007909 RRID:IMSR_JAX:007909 | |
| Genetic reagent (M. musculus) | CB17.Cg-PrkdcscidLystbg-J/Crl; SCID/Beige mice | Beijing Vital River Laboratory Animal Technology Co., Ltd | Strain #:405 | |
| Antibody | anti-Collagen Type II (mouse monoclonal) | Merck (Millipore) | Cat#:MAB8887 RRID:AB_2260779 | (1:500) |
| Antibody | anti-NFATc1 antibody (mouse monoclonal) | Santa Cruz Biotechnology | Cat#:sc-7294 RRID:AB_2152503 | IHC (1:200); Western blotting (1:1000) |
| Antibody | Allophycocyanin (APC) anti-mouse CD45 (rat monoclonal) | Thermo Fisher Scientific | Cat#:MCD4505 RRID:AB_10376146 | (1:100) |
| Antibody | APC anti-mouse CD166 (rat monoclonal) | Thermo Fisher Scientific | Cat#:17-1661-82 RRID:AB_2573170 | (1:100) |
| Antibody | APC anti-mouse CD31 (rat monoclonal) | BioLegend | Cat#:102410 RRID:AB_312905 | (1:100) |
| Antibody | APC anti-mouse CD200 (rat monoclonal) | BioLegend | Cat#:123810 RRID:AB_10900447 | (1:100) |
| Antibody | APC anti-mouse SCA1 (rat monoclonal) | BioLegend | Cat#:122511 RRID:AB_756196 | (1:100) |
| Antibody | APC anti-mouse CD105 (rat monoclonal) | BioLegend | Cat#:120414 RRID:AB_2277914 | (1:100) |
| Antibody | APC Rat IgG2a, κ isotype control antibody (rat monoclonal) | BioLegend | Cat#:400512 RRID:AB_2814702 | (1:100) |
| Antibody | APC Rat IgG2b, κ isotype control antibody (rat monoclonal) | BioLegend | Cat#:400612 RRID:AB_326556 | (1:100) |
| Antibody | APC Rat IgG1, κ isotype control (rat monoclonal) | BioLegend | Cat#:400412 RRID:AB_326518 | (1:100) |
| Antibody | Alexa Fluor 647 anti-mouse CD9 (rat monoclonal) | BioLegend | Cat#:124810 RRID:AB_2076037 | (1:200) |
| Antibody | Alexa Fluor 647 Rat IgG2a, κ isotype control antibody (rat monoclonal) | BioLegend | Cat#:400526 RRID:AB_2864284 | (1:200) |
| Antibody | Go-ChIP-Grade purified anti-NFATc1 antibody (mouse monoclonal) | BioLegend | Cat#:649608 RRID:AB_2721596 | (10 μg/ml) |
| Antibody | Purified mouse IgG1, κ isotype ctrl antibody (mouse monoclonal) | BioLegend | Cat#:400102 RRID:AB_2891079 | CUT&RUN (10 μg/ml); IHC (1:500) |
| Antibody | anti-GAPDH (rabbit monoclonal) | Cell Signaling Technology | Cat#:2118 RRID:AB_561053 | (1:2000) |
| Chemical compound, drug | Tamoxifen | Sigma-Aldrich | Cat#:T5648 | Adult mice (1 mg/10 g); 2-week-old or dam mice (0.5 mg/10 g) |
| Cell line (M. musculus) | ATDC5 cells (mouse 129 teratocarcinoma-derived osteochondral progenitors) | Merck (Sigma-Aldrich) | Cat#:99072806 RRID:CVCL_3894 | Authenticated by chondrogenic differentiation; tested negative for mycoplasma |
| Sequence-based reagent | sgRNA targeting mouse Nfatc1 exon 3 | This paper | TACGAGCTTCGGATCGAGGT | |
| Recombinant DNA reagent | pMSCV-caNFATc1; pMSCV-GFP | PMID:18243104 | ||
| Recombinant DNA reagent | pGL2B-COL2-6.5E309 | Other | Dr. Mary Goldring | |
| Transfected construct (mammalian) | gag/pol | Addgene | Cat#:14887 RRID:Addgene_14887 | |
| Transfected construct (mammalian) | VSV.G | Addgene | Cat#:14888 RRID:Addgene_14888 | |
| Transfected construct (mammalian) | pCMV-dR8.2 dvpr | Addgene | Cat#:8455 RRID:Addgene_8455 | |
| Transfected construct (mammalian) | pCMV-VSV-G | Addgene | Cat#:8454 RRID:Addgene_8454 | |
| Commercial assay or kit | M.O.M. Immunodetection Kit | Vector Laboratories | Cat#:BMK-2202 RRID:AB_2336833 | |
| Commercial assay or kit | CUT&RUN Assay Kit | Vazyme | Cat#:S702 | |
| Commercial assay or kit | Dual-Luciferase Reporter Assay System | Promega | Cat#:E1910 | |
| Software | FlowJo | BD Biosciences | RRID:SCR_008520 | Version 10.8.1 |
| Software | GraphPad Prism | GraphPad Prism (https://graphpad.com) | RRID:SCR_015807 | Version 9.2.1 |
Additional files
-
Supplementary file 1
Expression of cell surface markers in GFP+ and GFP- cells.
- https://cdn.elifesciences.org/articles/81569/elife-81569-supp1-v2.xlsx
-
Supplementary file 2
Primer sequences for quantitative PCR.
- https://cdn.elifesciences.org/articles/81569/elife-81569-supp2-v2.xlsx
-
MDAR checklist
- https://cdn.elifesciences.org/articles/81569/elife-81569-mdarchecklist1-v2.pdf