A rapid review of COVID-19’s global impact on breast cancer screening participation rates and volumes from January to December 2020
Figures
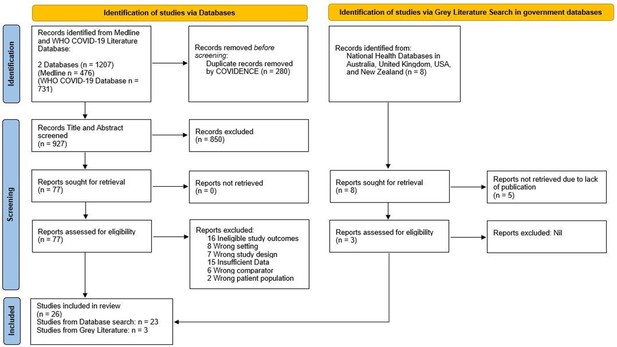
PRISMA Flow Diagram for Record Identification, Screening and Inclusion for Analysis (Page et al., 2021).
Tables
Descriptive characteristics of included cross-sectional studies (n = 26).
| Study | Publication type | Study design | Country | Region (If not national) | Total Female Population of Study Area | Sample size | Study screening data source | Screening (National/Regional) | Screening age range | Screening type | Screening time comparison | Types of Restrictions present over study period† | COVID-19 7 day new infection rate in region of focus (per 100000)* | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| International Travel Limits | Internal Movement Controls | Stay at home requirement | Public transport closure | Ban on gatherings of >10 people | Public events ban | Workplace closure | School closure | Minimum infection rate in study period | Maximum infection rate in study period | ||||||||||||
| Europe (n=7) | |||||||||||||||||||||
| Campbell et al., 2021 | Peer- reviewed | Cross sectional | Scotland (UK) | 2728000 | Not specified | NHS Scotland | National | 50–70 | Digital Mammography | Aug – Dec 2019 vs Aug -Dec 2020 | Yes | Yes | No | No | Yes | Yes | Yes | No | 10.14 | 212.67 | |
| Jidkova et al., 2022 | Peer- reviewed | Cross sectional | Belgium | Flanders | 3382265 | Not specified | Flanders Online Screening Database | Regional | 50–69 | Digital Mammography | Jul – Nov 2019 vs Jul – Nov 2020 | Yes | Yes | Yes | No | Yes | Yes | Yes | Yes | 3.58 | 580.63 |
| Knoll et al., 2022 | Preprint | Cross sectional | Austria | Innsbruck | 567300 | 596 | Database from gynecological oncological center in Austria, Tyrol | Local | 45–69 years invited for screening. Women aged 40–44 years and 70–75 years may opt in | Digital Mammography | Mar – Dec 2019 vs Mar – Dec 2020 | Yes | Yes | Yes | No | Yes | Yes | Yes | Yes | no data | no data |
| Eijkelboom et al., 2021 | Peer- reviewed | Cross sectional | Netherlands | 8701000 | 3371 | Netherlands Cancer Registry | National | 50–75 | Digital Mammography | Jan – Feb 2020 vs Jul – Aug 2020 | Yes | No | No | No | Yes | No | Yes | No | 0.32 | 67.25 | |
| Losurdo et al., 2022 | Peer- reviewed | Cross sectional | Italy | Friuli Venezia Giulia | 624418 | 58643 | “Data-Breast” database of the “Eusoma certified SSD Breast Unit of Trieste and from the Surgical Department of DAI Chirurgia Generale—ASUGI. | Regional | 50–69 | Digital Mammography | Oct – Dec 2019 vs Oct – Dec 2020 | Yes‡ | Yes‡ | Yes‡ | No‡ | Yes‡ | Yes‡ | Yes‡ | Yes‡ | 19.2 | 497.6 |
| Toss et al., 2021 | Peer- reviewed | Cross sectional | Italy | Northern Italy, Emilia Romagna | 2291000 | 24994 | Emilia Romagna National Healthcare System | Regional | 45–79 | Digital Mammography | 2019 vs 2020 | Yes‡ | Yes‡ | Yes‡ | Yes‡ | Yes‡ | Yes‡ | Yes‡ | Yes‡ | 4.00 | 390.9 |
| NHS England, 2021 | Government paper | Cross sectional | England (UK) | 33940000 | 2230000 | NHS England | National | 50–71 | Digital Mammography | 2019 vs 2020 | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | 0.00 | 92.36 | |
| Oceania (n=2) | |||||||||||||||||||||
| BreastScreen Australia, 2020 | Government Paper | Cross sectional | Australia | 12780000 | Not specified | BreastScreen Australia | National | 50–74 | Digital Mammography | May – Sep 2018 vs May – Sep 2020 | Yes | Yes | Yes | No | Yes | Yes | Yes | Yes | 0.18 | 13.31 | |
| BreastScreen Aoteroa, 2022 | Government Paper | Cross sectional | New Zealand | 2497000 | Not specified | BreastScreen Aotearoa | National | 45–69 | Digital Mammography | May – Dec 2018 vs May - Dec 2020 | Yes | Yes | Yes | No | Yes | Yes | Yes | Yes | 0 | 1.06 | |
| Asia (n=1) | |||||||||||||||||||||
| Shen et al., 2022 | Peer- reviewed | Cross sectional | China | Taiwan | 11981657 | 699911 | Taiwan National Infectious Disease Statistics system | Regional | 40–69 | Digital Mammography | Jan – Apr 2019 vs Jan – Apr 2020 | Yes | No | No | No | No | No | No | Yes | no data | no data |
| Americas (n=16) | |||||||||||||||||||||
| Bessa, 2021 | Peer- reviewed | Cross sectional | Brazil | 106500000 | (2019: 20636636; 2020: 21140958) | Brazilian Unified Health System (SUS) | National | 50–69 | Digital Mammography | 2019 vs 2020 | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | 0.00 | 149.68 | |
| Ribeiro et al., 2022 | Peer- reviewed | Cross sectional | Brazil | 106500000 | 5996798 | Brazilian National Health Service (SUS) Outpatient Information System (SIA/SUS), SUS Hospital Information System (SIH/SUS), High Complexity Procedure Authorizations database (APAC), Cancer Information System (ISCAN). | National | 50–69 | Digital Mammography | Jul – Dec 2019 vs Jul – Dec 2020 | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | 53.72 | 149.68 | |
| Chiarelli et al., 2021 | Peer- reviewed | Cross sectional | Canada | Ontario | 7371000 | 426967 | Ontario Breast Screening Program (OBSP) | Regional | 50–74 | Digital Mammography, MRI (High risk) | Jul - Dec 2019 vs Jul - Dec 2020 | Yes | Yes | Yes | No | Yes | Yes | Yes | Yes | 3.99 | 117.01 |
| Walker et al., 2021 | Peer- reviewed | Cross sectional | Canada | Ontario | 7371000 | 605889 (2019) 284242 (2020) | Ontario Breast Screening Program (OBSP) | Regional | 50–74 | Digital Mammography | Modelled 2019 data vs Dec 2020 | Yes | Yes | Yes | No | Yes | Yes | Yes | Yes | 75.74 | 117.01 |
| Doubova et al., 2021 | Peer- reviewed | Cross sectional | Mexico | 64570000 | 1431216 | Mexican Institute of Social Security (IMSS) | National | 40 - unspecified | Digital Mammography | Jan 2019 – Mar 2020 vs Apr – Dec 2020 | Yes | Yes | Yes | No | No | Yes | Yes | Yes | 2.60 | 61.12 | |
| Chen et al., 2021 | Peer- reviewed | Cross sectional | USA | 167500000 | Not specified | HealthCore Integrated Research Database | National | 50–79 years | Digital Mammography | Jul 2019 vs Jul 2020 | Yes | Yes | Yes | No | Yes | Yes | Yes | Yes | 119.03 | 142.00 | |
| Amornsiripanitch et al., 2021 | Peer- reviewed | Cross sectional | USA | Massachusetts | 3537000 | 32387 | Electronic medical record (Epic, Verona, WI) - Massachusetts. One tertiary care academic center, a community hospital, a specialized cancer center, three outpatient imaging centers, one urban healthcare center, and one mobile mammography van | Regional | 40 - unspecified years | Digital Mammography | Jun – Aug 2019 vs Jun – Aug 2020 | Yes | Yes | No | No | Yes | Yes | No | No | 17.06 | 53.09 |
| Becker et al., 2021 | Peer- reviewed | Cross sectional | USA | Michigan | 5062000 | 7250080 | Women enrolled in Health Managed Organization (HMO) Blue Cross Blue Shield (BCBS) in Michigan | Regional | 40–74 | Digital Mammography | Dec 2019 vs Dec 2020 | Yes | Yes | Yes | No | Yes | Yes | Yes | Yes | 147.56 | 328.94 |
| DeGroff et al., 2021 | Peer- reviewed | Cross sectional | USA | 167500000 | 630264 | Breast and Cervical Cancer Early Detection Program (NBCCEDP) Database, which provides cancer screening services to women with low income and inadequate health insurance | National | 40–74 | Digital Mammography | Jun 2019 vs Jun 2020 | Yes | Yes | Yes | No | Yes | Yes | Yes | Yes | 45.46 | 103.84 | |
| Dennis et al., 2021 | Peer- reviewed | Cross sectional | USA | 167500000 | 475083 | Behavioral Risk Factor Surveillance System (BRFSS) survey database | National | 40–74 | Digital Mammography | 2014–2019 vs 2020 | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | 0.00 | 460.68 | |
| Fedewa et al., 2021 | Peer- reviewed | Cross sectional | USA | 167500000 | 2019:142003 2020:150630 | Data from 32 CHCs of the American Cancer Society's Community Health Advocates Implementing Nationwide Grants for Empowerment and Equity (CHANGE) grant program to increase BCSRs and follow-up care | National | 50–74 | Digital Mammography | 2019 vs 2020 | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | 0.00 | 460.68 | |
| Lehman et al., 2022 | Preprint | Cross sectional | USA | 167500000 | 29276 | Screening database over 5 facilities | National | Unspecified | Digital Mammography | 2019 vs 2020 | Yes | Yes | Yes | No | Yes | Yes | Yes | Yes | 0.00 | 460.68 | |
| London et al., 2022 | Peer- reviewed | Cross sectional | USA | 167500000 | 34000000 (full study including colorectal cancers) | TriNetX Research Network | National | Unspecified | Digital Mammography | Jul – Dec 2019 vs Jul – Dec 2020 | Yes | Yes | Yes | No | Yes | Yes | Yes | Yes | 74.54 | 460.68 | |
| Miller et al., 2021 | Peer- reviewed | Cross sectional | USA | Virginia | 2757460 | Not specified | Instituition Database, University of Virginia | Regional | Unspecified (45 - 70) | Digital Mammography | Jan – Nov 2019 vs Jan - Nov 2020 | Yes | Yes | Yes | No | Yes | Yes | Yes | Yes | no data | no data |
| Sprague et al., 2021 | Peer- reviewed | Cross sectional | USA | 167500000 | 461083 | 62 radiology facilities of Breast Cancer Surveillance Consortium | National | 40–79 | Digital Mammography | Jan-Jul 2019 vs Jan-Jul 2020 | Yes | Yes | Yes | No | Yes | Yes | Yes | Yes | 0.00 | 142.00 | |
| Nyante et al., 2021 | Peer- reviewed | Cross sectional | USA | North Carolina | 5099371 | 42412 | 7 academic and community breast imaging facilities in North Carolina | Regional | 40–79 | Digital Mammography | Modelled Sep 2019 data vs Sep 2020 | Yes | No | No | No | No | Yes | Yes | Yes | 80.27 | 91.26 |
-
England’s and Scotland’s NHS systems are devolved and, therefore, are separate national entities. However, they hold similar screening criterion where breast screening policy in the NHS (across the UK) is that all women aged 50–70 y + 364 d are invited for breast screening once every 3 y.
-
*
These infection rates were region-specific and analogous to the region the study involved. If study period was ≤1 mo, only infection data from the first and last week of the period will be collected. If study period was over the whole year of 2020, the earliest available public health data was used (e.g., study period started from January 2020 but data was only available in March, March data used as first interval of analysis). It should be noted that there is reporting bias here as testing rates may differ between countries. These infection incidence rates were based on national/regional data depending on whether the study population originated from an entire nation or a limited region within a nation. (Dipartimento della Protezione Civile, 2023a; Dipartimento della Protezione Civile, 2023b; Government of Ontario, 2023; Government of the Netherlands, 2023; Cooper et al., 2023; IARC, 2022; Mathieu, 2022; MDHHS, 2023; Medicaid.gov, 2022; NCDHHS, 2023; NHS England, 2021; OECD, 2021a; PAHO, 2020; Sciensano, the Belgian Institute for Health, 2023; SPICe, 2023; State of Michigan, 2020; State of North Carolina, 2020; The Scottish Government, 2022; UK Government, 2023; WHO, 2023; WHO, 2022; WHO, 2022; Worldometer, 2022; WHO, 2021; Yucatan Times, 2021).
-
†
Types of restrictions will include restrictions that were withdrawn at any point of the study period. Restrictions present were classified as per non-pharmacological interventions mentioned by the paper Li et al., 2021 ‘The Temporal Association of introducing and lifting non-pharmaceutical interventions with the time-varying reproduction number (R) of SARS-COV-2: A modelling study across 131 countries’, The Lancet Infectious Diseases, if restrictions were introduced/withdrawn during the study period, it will still be indicated as a 'Yes', Data from Oxford COVID-19 policy tracker, devolved state-wide healthcare organization websites in Canada, the USA, and UK was used to assess this.
-
‡
Data was unavailable for regions in this country, national restrictions were assessed instead.
Breast cancer screening volumes change among 106,484,908 subjects from eight countries.
| Percentage change in volume of breast cancer screening (N = 17) | |||||||
|---|---|---|---|---|---|---|---|
| Study | Country | Region | National/regional (scope of study population*) | Type of breast screening program employed within the study population | Sample size | Screening timeframe comparison | Volume change relative to non-COVID-19 period (%) |
| Europe (n = 2) | |||||||
| Losurdo et al., 2022 | Italy | Friuli Venezia Giulia | Regional | Population-based screening present in country | 58,643 | Oct–Dec 2019 vs. Oct–Dec 2020 | 11.90 |
| NHS England, 2021 | UK | England | National | Population-based screening present in country | 3,420,000 | Monthly average 2019 vs. monthly average 2020 | 15.80 |
| Oceania (n = 1) | |||||||
| BreastScreen Australia, 2020 | Australia | NA | National | Population-based screening present in country | 802,146 | May–Sep 2018 vs. May–Sep 2020 | 12.88 |
| Asia (n = 1) | |||||||
| Shen et al., 2022 | China | Taiwan | Regional | Population-based screening present in country | 699,911 | Jan–Apr 2019 vs. Jan–Apr 2020 | 22.07 |
| America (n = 13) | |||||||
| Bessa, 2021 | Brazil | NA | National | Population-based screening present in country† | (2019: 20,636,636; 2020: 21,140,958) | 2019 vs. 2020 | 42.72 |
| Ribeiro et al., 2022 | Brazil | NA | National | Population-based screening present in country but private sector databases included Brazilian National Health Service (SUS), Outpatient Information System (SIA/SUS), SUS Hospital Information System (SIH/SUS), High Complexity Procedure Authorizations database (APAC), Cancer Information System (ISCAN) | 5,996,798 | Jul–Dec 2019 vs. Jul–Dec 2020 | 41.49 |
| Doubova et al., 2021 | Mexico | NA | National | Population-based screening present in country ‡ | 1,431,216 | Jan 2019–Mar 2020 vs. Apr–Dec 2020 | 61.30 |
| Chiarelli et al., 2021 | Canada | Ontario | Regional | Population-based screening present in country | 426,967 | Jul–Dec 2019 vs. Jul–Dec 2020 | 31.30 |
| Walker et al., 2021 | Canada | Ontario | Regional | Population-based screening present in country | 890,131 | Modeled 2019 data vs. Dec 2020 | 22.80 |
| Lehman et al., 2022 | USA | NA | National | Privatized system with mix of national and private insurance usage | 29,276 | 2019 vs. 2020 | 36.50 |
| Miller et al., 2021 | USA | North Carolina | Regional | Privatized system with mix of national and private insurance usage | 8,536,000 | Jan–Nov 2019 vs. Jan–Nov 2020 | 9.80 |
| Amornsiripanitch et al., 2021 | USA | Massachusetts | Regional | Privatized system with mix of national and private insurance usage | 32,387 | Jun–Aug 2019 vs. Jun–Aug 2020 | 10.50 |
| London et al., 2022 | USA | NA | National | Privatized system with mix of national and private insurance usage | 34,000,000 | Dec 2019 vs. Dec 2020 | 20.00 |
| DeGroff et al., 2021 | USA | NA | National | The National Breast and Cervical Cancer Early Detection Program (NBCCEDP) that provides cancer screening services to women with low income and inadequate health insurance | 630,264 | Jun 2019 vs. Jun 2020 | 39.00 |
| Becker et al., 2021 | USA | Michigan | Regional | Health Managed Care Organization (HMO)-based screening (database covers HMO data from Michigan) | 7,250,080 | Dec 2019 vs. Dec 2020 | 18.10 |
| Sprague et al., 2021 | USA | NA | National | Privatized system with mix of national and private insurance usage | 461,083 | Jul 2019 vs. Jul 2020 | 10.30 (–20.40 to 6.60) |
| Nyante et al., 2021 | USA | North Carolina | Regional | Privatized system with mix of national and private insurance usage | 42,412 | Modeled Sep 2019 data vs. Sep 2020 | 9.00 |
-
NA indicates not applicable. For studies conducted in the USA, ACS guidelines were used as the data collection comparator starting point where March–June 2020 was considered to be a suspension in screening.
-
*
This column highlights the origin of the study population in which whether it was drawn from a specific region within a nation, or if the study population was drawn from the entire country.
-
†
The study population from this specific study (Bessa, 2021) was solely drawn from a national population-based screening database in Brazil. It should be noted that Brazil has a lower proportion of population-based breast screening coverage relative to other countries; having a coverage of 24% in the eligible population (Unger-Saldaña et al., 2020).
-
‡
It should be noted that Mexico has a lower proportion of population-based breast screening coverage relative to other countries due to recent introduction; having ~20% coverage of the eligible population (OECD, 2021b; PAHO, 2020).
Breast cancer screening participation uptake rates change from nine studies from five countries.
| Percentage change in participation uptake rate of breast cancer screening (N = 9) | |||||||
|---|---|---|---|---|---|---|---|
| Study | Country | Region | National/regional (scope of study population)* | Type of breast screening program employed within the study population | Sample size | Screening timeframe comparison | Participation rate change relative to non-COVID-19 period |
| Europe (n = 3) | |||||||
| NHS England, 2021 | UK | England | National | Population-based screening available in country | 3,420,000 | 2019 vs. 2020 | 11.80% |
| Campbell et al., 2021 | UK | Scotland | National | Population-based screening available in country | NA | Aug–Dec 2019 vs. Aug–Dec 2020 ‡ | +10.96% (Aug 2020) |
| +2–8% (Sep 2020–Mar 2021 vs. Sep 2019– Mar 2020)‡ | |||||||
| Jidkova et al., 2022 | Belgium | Flanders | Regional | Population-based screening available in country | NA | Jul–Dec 2019 vs. Jul–Dec 2020 | 1.0% (–1.3; –0.7) |
| Oceania (n = 1) | |||||||
| BreastScreen Aoteroa, 2022 | New Zealand | NA | National | Population-based screening available in country | NA | Dec 2018/2019 vs. May–Dec 2020 | 6.70% |
| Americas (n = 5) | |||||||
| Bessa, 2021 | Brazil | NA | National | Population-based screening available in country† | (2019: 20,636,636; 2020: 21,140,958) | 2019 vs. 2020 | 43.54% |
| Dennis et al., 2021 | USA | NA | National | Privatized system with mix of national and private insurance usage | 475,083 (age: 50–74) 117,498 (age: 40–49) | 2014–2019 vs. 2020 | 5.30% (50–79) |
| 7.20% (40–49) | |||||||
| Fedewa et al., 2021 | USA | NA | National | Privatized system with mix of national and private insurance usage | 434,840 | 2019 vs. 2020 | 8.00% |
| Amornsiripanitch et al., 2021 | USA | Massachusetts | Regional | Privatized system with mix of national and private insurance usage | 32,387 | Jun–Aug 2019 vs. Jun–Aug 2020 | 14.80% |
| Chen et al., 2021 | USA | NA | National | Privatized system with mix of national and private insurance usage | NA | Jul 2019 vs. Jul 2020 | 3.33% |
-
NA indicates not applicable For studies conducted in the USA, ACS guidelines were used as the data collection comparator starting point where Mar-Jun 2020 was considered to be a suspension in screening.
-
*
This column highlights the origin of the study population in which whether it was drawn from a specific region within a nation, or if the study population was drawn from the entire country.
-
†
The study population from this specific study (Bessa, 2021) was solely drawn from a national population-based screening database in Brazil. It should be noted that Brazil has a lower proportion of population-based breast screening coverage relative to other countries; having a coverage of 24% in the eligible population (Unger-Saldaña et al., 2020).
-
‡
It should be noted that this study presented a range of values (2–8%) comparing the uptake rate from Sep 2020 to Mar 2021 vs. Sep 2019 to Mar 2020. As the timeframe of Jan–Mar 2021 was not within the scope of the study, we used the point estimate of the uptake rate in Aug 2020 vs. Aug 2019 as our last available data point instead.
Summary of results of appraisal of all included studies with Joanna Briggs Institute (JBI) Critical Appraisal Tool for cross-sectional studies.
| JBI Critical Appraisal Tool for cross-sectional studies appraisal table | ||||||||
|---|---|---|---|---|---|---|---|---|
| Study | Were the criteria for inclusion in the sample clearly defined? | Were the study subjects and the setting described in detail? | Was the exposure measured in a valid and reliable way? | Were objective, standard criteria used for measurement of the condition? | Were confounding factors identified? | Were strategies to deal with confounding factors stated? | Were the outcomes measured in a valid and reliable way? | Was appropriate statistical analysis used? |
| Amornsiripanitch et al., 2021 | Y | Y | Y | Y | Y | N | Unclear | Unclear |
| Becker et al., 2021 | Y | Y | Y | N | Y | N | N | Unclear |
| Bessa, 2021 | Y | Unclear | Y | Y | N | N | N | Unclear |
| Campbell et al., 2021 | Y | Unclear | Y | Y | Unclear | N | Y | Unclear |
| Chen et al., 2021 | Y | Unclear | Y | Y | Y | N | Y | Unclear |
| Chiarelli et al., 2021 | Y | Y | Y | Y | Unclear | N | Y | Unclear |
| DeGroff et al., 2021 | Y | Y | Y | Y | Y | N | Y | Unclear |
| Dennis et al., 2021 | Y | Y | Y | Y | N | N | Y | Unclear |
| Doubova et al., 2021 | Y | Unclear | Y | Y | N | N | N | Y |
| Jidkova et al., 2022 | Y | Y | Y | Y | Unclear | N | Y | Unclear |
| Knoll et al., 2022 | Y | Y | Y | Y | N | N | Y | Unclear |
| Fedewa et al., 2021 | Y | Y | Y | Y | N | N | Y | Unclear |
| BreastScreen Australia, 2020 | Y | Y | Y | Y | N | N | Y | N |
| Eijkelboom et al., 2021 | Y | Y | Y | Y | Y | Y | Y | Unclear |
| Lehman et al., 2022 | N | N | Y | Y | Y | N | Y | Unclear |
| London et al., 2022 | N | N | Y | Y | N | N | N | Unclear |
| Losurdo et al., 2022 | Y | Y | Y | Y | N | N | Y | Unclear |
| Walker et al., 2021 | Y | Y | Y | Y | Unclear | N | Y | Unclear |
| Toss et al., 2021 | Y | Y | Y | Y | N | N | Y | Unclear |
| Shen et al., 2022 | Y | Y | Y | Y | Unclear | N | Y | Unclear |
| Ribeiro et al., 2022 | Y | Y | Y | Y | N | N | Y | Unclear |
| Miller et al., 2021 | Y | Unclear | Y | Y | N | N | Y | Y |
| Sprague et al., 2021 | Y | Y | Y | Y | Unclear | Y | Y | Y |
| Nyante et al., 2021 | Y | Y | Y | Y | Y | Y | Y | Y |
| NHS England, 2021 | Y | Y | Y | Y | N | N | Y | N |
| BreastScreen Aoteroa, 2022 | Y | Y | Y | Y | N | N | Y | N |
-
Green = yes; yellow = unclear; orange = no.
Search strategies for rapid review of breast cancer participation and volume during COVID.
| Search string for Ovid MEDLINE(R) and In-Process, In-Data-Review & Other Non-Indexed | ||
|---|---|---|
| Citations | ||
| Search number | Search domain | Search string in: [mp = title, book title, abstract, original title, name of substance word, subject heading word, floating subheading word, keyword heading word, organism supplementary concept word, protocol supplementary concept word, rare disease supplementary concept word, unique identifier, synonyms] |
| #1 | COVID-19 | (COVID-19 OR 2019 novel coronavirus disease OR 2019 novel coronavirus infection OR 2019 ncov disease OR 2019 ncov infection OR 2019-ncov disease OR 2019-ncov diseases OR 2019-ncov infection OR 2019-ncov infections OR covid 19 OR covid 19 pandemic OR covid 19 virus disease OR covid 19 virus infection OR covid-19 OR covid-19 pandemic OR covid-19 pandemics OR covid-19 virus disease OR covid-19 virus diseases OR covid-19 virus infection OR covid-19 virus infections OR covid19 OR coronavirus disease 19 OR coronavirus disease 2019 OR coronavirus disease-19 OR disease 2019, coronavirus OR sars cov 2 infection OR sars coronavirus 2 infection OR sars-cov-2 infection OR sars-cov-2 infections OR severe acute respiratory syndrome coronavirus 2 infection OR disease, 2019-ncov OR disease, covid-19 virus OR infection, 2019-ncov OR infection, covid-19 virus OR infection, sars-cov-2 OR pandemic, covid-19 OR virus disease, covid-19 OR virus infection, covid-19 OR Coronavirus, 2019 Novel OR ncov OR covid* OR coronavirus* OR SARS* OR severe acute respiratory syndrome OR coronavirus pandemic OR coronavirus disease pandemic) |
| #2 | Breast cancer | (Breast Neoplasms OR Breast Carcinoma In Situ OR Carcinoma, Ductal, Breast OR Carcinoma, Lobular OR breast cancer OR breast carcinoma* OR breast malignant neoplasm* OR breast malignant tumo?r* OR breast neoplasm* OR breast tumo?r* OR cancer of breast? OR cancer of the breast? OR mammary carcinoma* OR mammary neoplasm* OR malignant neoplasm? of breast OR malignant tumo?r? of breast OR mammary cancer* OR neoplasm?, breast OR tumo?r, breast OR tumo?rs, breast OR cancer?, breast OR cancer?, mammary OR carcinoma?, human mammary OR carcinoma?, breast OR neoplasm?, human mammary OR breast carcinoma in situ OR lobular carcinoma in situ OR lcis, lobular carcinoma in situ OR mammary ductal carcinoma? OR carcinoma, ductal, breast OR carcinoma, infiltrating duct OR carcinoma, invasive ductal, breast OR carcinoma, mammary ductal OR carcinomas, infiltrating duct OR carcinomas, mammary ductal OR invasive ductal carcinoma, breast OR lobular carcinoma? OR carcinoma?, lobular OR breast* OR breast tumo?r OR breast tumo?rs OR breast malignant tumo?rs OR breast malignan* OR mammary malignan* OR malignant tumo?rs of breast OR neoplasm? of breast OR breast neoplasm OR lcis) |
| #3 | Mass screening | (Mass Screening OR Mass Chest X-ray OR Early Diagnosis OR Early Detection of Cancer OR Mammography OR screening* OR Ultrasonography, Mammary OR Ultrasonography OR mass chest x ray OR mass chest x-ray* OR mass chest xray* OR x-ray, mass chest OR x-rays, mass chest OR xray, mass chest OR xrays, mass chest OR disease early detection OR early detection of disease OR early diagnosis OR diagnosis, early OR cancer early detection OR cancer early diagnosis OR early detection of cancer OR early diagnosis of cancer OR digital breast tomosyntheses OR digital breast tomosynthesis OR x ray breast tomosynthesis OR x-ray breast tomosyntheses OR x-ray breast tomosynthesis OR breast tomosyntheses, digital OR breast tomosyntheses, x-ray OR breast tomosynthesis, digital OR breast tomosynthesis, x-ray OR breast tissue imaging OR mastography OR mass breast xray OR mass breast x-ray OR chest xray OR chest x-ray OR mammogra* OR program* OR ultrasonic* OR echograph* OR echotomograph* OR sonography* OR ultrasonograph* OR ultrasound* OR exam*) |
| #4 | Search string | 1 AND 2 AND 3 |
| #5 | Final search string | Limit 4 to English language |
| Search string for WHO COVID-19 Literature Database (updated to March 12, 2022) | ||
| Search number | Search concept | Title, abstract, subject |
| #1 | Breast cancer | ((Breast Neoplasms) OR (Breast Carcinoma In Situ) OR (Carcinoma, Ductal, Breast) OR (Carcinoma, Lobular) OR (breast cancer*) OR (breast carcinoma*) OR (breast malignant neoplasm*) OR (breast malignant tumo?r*) OR (breast neoplasm*) OR (breast tumo?r*) OR (cancer of breast?) OR (cancer of the breast?) OR (mammary carcinoma*) OR (mammary neoplasm*) OR (malignant neoplasm? of breast) OR (malignant tumo?r? of breast) OR (mammary cancer*) OR (breast carcinoma in situ) OR (lobular carcinoma in situ) OR (mammary ductal carcinoma*) OR (breast ductal carcinoma*) OR (infiltrating duct carcinoma*) OR (invasive ductal carcinoma) OR (mammary ductal carcinoma*) OR (invasive ductal breast carcinoma) OR (lobular carcinoma*) OR (breast tumo?r*) OR (breast malignant tumo?r*) OR (breast malignan*) OR (mammary malignan*) OR (malignant tumo?rs of breast*) OR (neoplasm? of breast) OR (lcis*)) |
| #2 | Screening | ((Mass Screening) OR (Mass Chest X-ray) OR (Early Diagnosis) OR (Early Detection of Cancer) OR (Mammography) OR (Ultrasonography, Mammary) OR (Ultrasonography) OR (national screening) OR (screening*) OR (mass chest x ray) OR (mass chest x-ray*) OR (mass chest xray*) OR (mass chest x-ray*) OR (disease early detection) OR (early detection of disease) OR (early diagnosis) OR (diagnosis, early) OR (cancer early detection) OR (cancer early diagnosis) OR (early detection of cancer) OR (early diagnosis of cancer) OR (digital breast tomosyntheses) OR (digital breast tomosynthesis) OR (x ray breast tomosynthesis) OR (x-ray breast tomosyntheses) OR (breast tomosynthesis*) OR (breast tissue imaging) OR (mastography) OR (mass breast xray) OR (mass breast x-ray) OR (chest xray) OR (chest x-ray) OR (mammogra*) OR (program*) OR (ultrasonic*) OR (echograph*) OR (ultrasonographic*) OR (sonography*) OR (echotomograph*) OR (ultrasound*) OR (exam*)) |
| #3 | Final search string | #1 AND #2 |
| English-language filter | ||
Additional files
-
MDAR checklist
- https://cdn.elifesciences.org/articles/85680/elife-85680-mdarchecklist1-v2.pdf
-
Supplementary file 1
Full descriptive characteristics and data from included cross-sectional studies (n=26).
- https://cdn.elifesciences.org/articles/85680/elife-85680-supp1-v2.xlsx





