Midbrain encodes sound detection behavior without auditory cortex
Figures
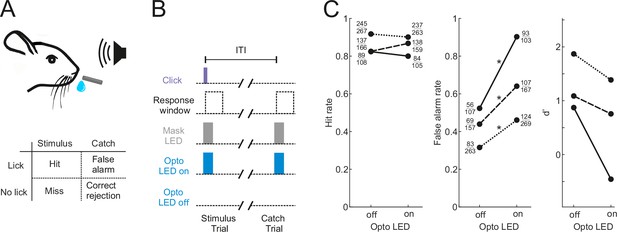
Optogenetic inactivation of the auditory cortex impairs sound detection performance in head-fixed mice.
(A) Schematic of the click detection task. (B) Trial structure for experiments involving optogenetic manipulation. Stimulus trials (click) and catch trials (no click) were randomly interleaved and consecutive trials were separated by a randomly varying inter-trial interval (ITI). LEDs placed over each auditory cortex were switched on randomly in half of the stimulus and catch trials to photoactivate the opsin. A separate set of LEDs (Mask LEDs) placed directly in front of the mouse’s eyes were switched on in all Opto-on and Opto-off trials to prevent mice from visually registering the light from the photoactivation LEDs. (C) Detection performance in trials during which light was shone on the auditory cortex for optogenetic silencing (Opto LED – on) vs control trials (Opto LED - off). Different line styles indicate different mice (n=3). Numbers next to data points indicate the numbers of hit and false alarm trials over a total number of stimulus and catch trials, respectively. *p<0.001, two-sided Chi-squared proportion test.
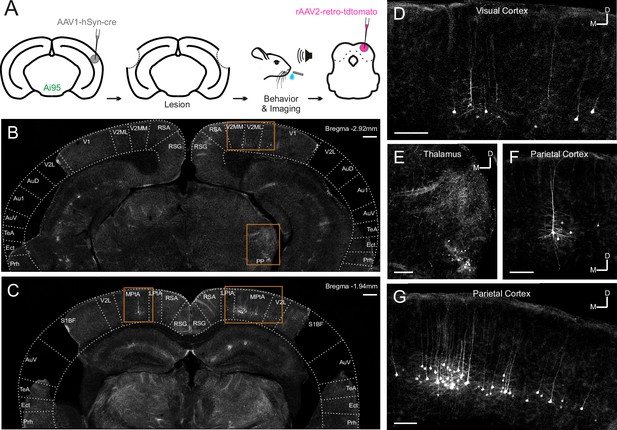
Retrograde viral tracing of inferior colliculus (IC)-projecting neurons in bilaterally lesioned mice.
(A) Timeline of experimental procedures. AAV1.hSyn.cre.WPRE was injected into the right auditory cortex of GCaMP6f-reporter (Ai95D) mice. This causes transsynaptic delivery of the virus to the IC and expression of GCaMP6f in corticorecipient IC neurons. Several weeks later, the mice underwent bilateral lesioning of the auditory cortex either by aspiration or by thermocoagulation (see Figure 2—figure supplement 2 for histological sections from a mouse that underwent thermocoagulation) and were implanted with a glass window over the right auditory cortex. Following recovery from this procedure, water access was restricted and, 2–3 days later, behavioral training and imaging commenced. After data collection had been completed, rAAV2-retro-tdTomato was injected in the dorsal IC in order to label corticocollicular neurons that had remained intact. (B, C) Coronal sections showing lesion extent at different rostrocaudal positions for one example mouse. Area borders were drawn onto the images according to Paxinos et al., 2001. No retrogradely-labeled neurons were found near the lesion borders, suggesting that the auditory cortex had been completely removed. Corticocollicular projections from non-temporal regions as well as thalamocollicular projections remained intact. Scale bars, 200 µm. (D) High magnification image (location shown by the upper rectangle in (B)) showing corticocollicular neurons in the visual cortex. Scale bar, 100 µm. (E) High magnification image (location shown by the lower rectangle in (B)) showing thalamocollicular neurons in the peripeduncular nucleus of the thalamus (PP). Scale bar, 100 µm. (F, G) High magnification images (locations shown by the left and right rectangles in (C), respectively) showing corticocollicular neurons in the parietal cortex. Scale bars,100 µm. Cortical area abbreviations: Au1, primary auditory; AuD, secondary auditory, dorsal; AuV, secondary auditory, ventral; Ect, ectorhinal; LPta, lateral parietal association; MPta, medial parietal association; Prh, perirhinal; RSG, retrosplenial granular; RSA, retrosplenial agranular; S1BF, primary somatosensory, barrel field; TeA, temporal association; V1, primary visual; V2L, secondary visual, lateral; V2ML, secondary visual, mediolateral; V2MM, secondary visual, mediomedial.
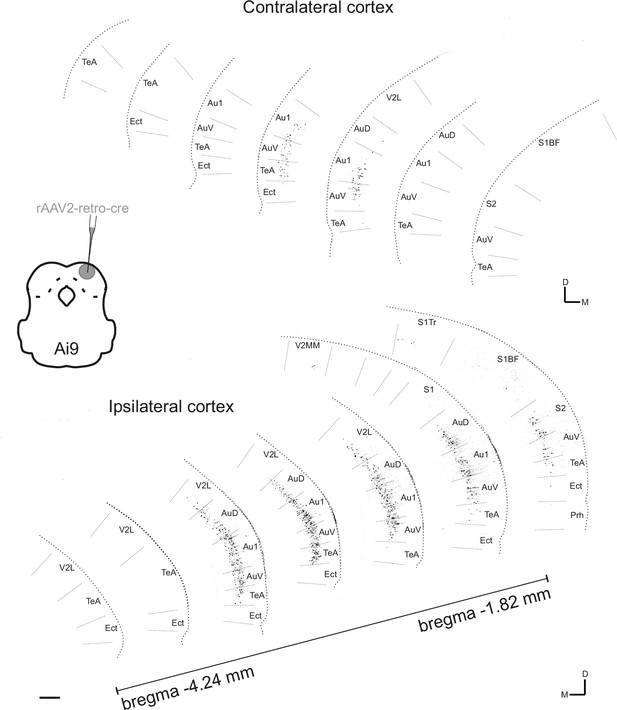
Contra- and ipsilateral corticocollicular neurons along the rostrocaudal axis.
Seven coronal sections are shown from each hemisphere covering approximately 2.5 mm of the rostrocaudal axis. Corticocollicular neurons were labeled by injecting a total of 150 nL of rAAV2-CAG-cre into the dorsal inferior colliculus (IC) (at three sites and several depths from 100 µm - 400 µm below the brain surface) of a tdTomato reporter mouse (Ai9). Data were obtained using whole-brain laser scanning two-photon tomography. The resulting images were grayscale inverted and thresholded to remove all background labeling so that they could be more easily arranged into a common figure. Area borders were drawn onto the images according to Paxinos et al., 2001. Cortical area abbreviations: Au1, primary auditory; AuD, secondary auditory, dorsal; AuV, secondary auditory, ventral; Ect, ectorhinal; Prh, perirhinal; S1, primary somatosensory; S1BF, primary somatosensory, barrel field; S1Tr, primary somatosensory, trunk region; S2, secondary somatosensory; TeA, temporal association; V2L, secondary visual, lateral. Scale bar, 200 µm.
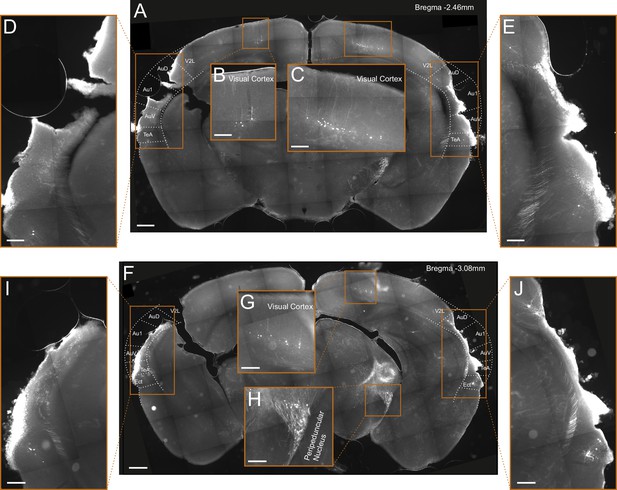
Lesioning by thermocoagulation.
(A) Coronal section showing lesion extent in a mouse that had undergone lesioning by thermocoagulation. After data collection had been completed, rAAV2-retro-tdTomato was injected along the dorsal inferior colliculus (IC) in order to label corticocollicular neurons that had remained intact. Area borders were drawn onto the images according to Paxinos et al., 2001. Scale bar, 500 µm. (B, C) Higher magnification images showing tdTomato-labeled corticocollicular neurons in the left and right visual cortex. Scale bars, 200 µm. (D, E) Higher magnification images of the temporal regions surrounding the lesion sites, show a very small number of residual corticocollicular neurons in the left and right temporal association area and the right dorsal auditory field. Scale bars, 200 µm. (F) Same as (A) for a different coronal section of the same mouse. (G, H) Higher magnification images show tdTomato-labeled corticocollicular neurons in the right visual cortex and thalamocollicular neurons in the right peripeduncular nucleus. Scale bars, 200 µm. (I, J) Higher magnification images of the temporal regions surrounding the lesion sites showing a very small number of residual corticocollicular neurons in the left and right ectorhinal cortex and the right dorsal auditory field. Scale bars, 200 µm. While the lesion procedure spared some auditory cortex tissue in this animal, its visual appearance and the fact that barely any corticocollicular neurons could be found suggests that this residual tissue was almost completely destroyed. Consequently, we categorized this animal as having a (near-)complete lesion, meaning that 5% or less of the auditory cortex was left intact. Cortical area abbreviations: Au1, primary auditory; AuD, secondary auditory, dorsal; AuV, secondary auditory, ventral; Ect, ectorhinal; TeA, temporal association; V2L, secondary visual, lateral.
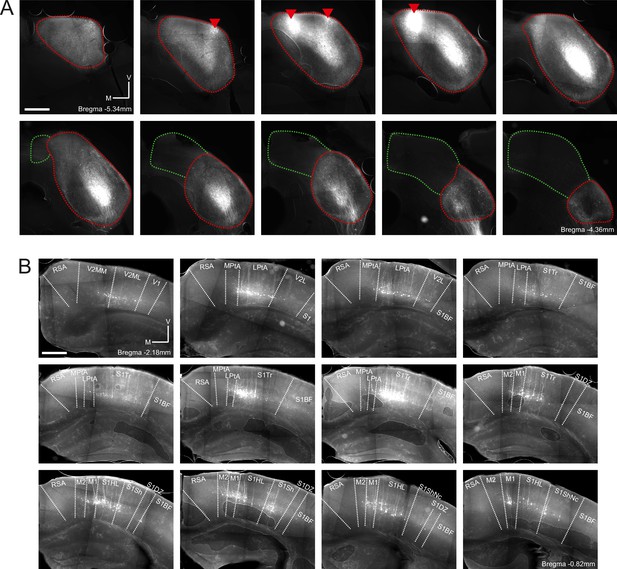
Retrograde labeling of corticocollicular neurons in non-temporal areas of the cerebral cortex is not the result of viral leakage into the superior colliculus.
(A) Coronal sections showing the right midbrain of one example mouse (same mouse as in Figure 2—figure supplement 2). Sections are ordered caudo-rostrally from top left to bottom right. Red lines indicate the approximate outline of the inferior colliculus, and green lines indicate the approximate outline of the superior colliculus. Red triangles indicate rAAV2-retro-tdTomato injection locations. In addition to the labeling near the injection sites, widespread retrograde labeling is found in the central nucleus of the inferior colliculus. No labeled cell bodies were found in the superior colliculus. Scale bar, 500 µm. (B) Coronal sections showing corticocollicular neurons in non-temporal areas of the right cerebral cortex labeled as a result of the rAAV2-retro-tdTomato injections in the inferior colliculus illustrated in (A). Sections are ordered caudo-rostrally from top left to bottom right. Area borders were drawn onto the images according to Paxinos et al., 2001. Scale bar, 500 µm. Cortical area abbreviations: LPta, lateral parietal association; MPta, medial parietal association; M1: primary motor; M2: secondary motor; RSA, retrosplenial agranular; S1BF, primary somatosensory, barrel field; S1DZ, primary somatosensory, dysgranular region; S1HL, primary somatosensory, hindlimb region; S1Sh, primary somatosensory, shoulder region; S1ShNc, primary somatosensory, shoulder/neck region; S1Tr, primary somatosensory, trunk field; V1, primary visual; V2L, secondary visual, lateral; V2ML, secondary visual, mediolateral; V2MM, secondary visual, mediomedial.
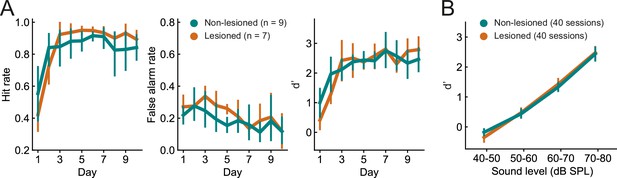
Lesioned and non-lesioned mice are indistinguishable in their click detection learning rate and sensitivity.
(A) Hit rate, false alarm rate, and d’ over time for lesioned and non-lesioned animals. (B) d’ as a function of sound level. The sound levels used were not identical across all mice and were, therefore, combined into 10 dB wide bins. Error bars indicate 95% confidence intervals.
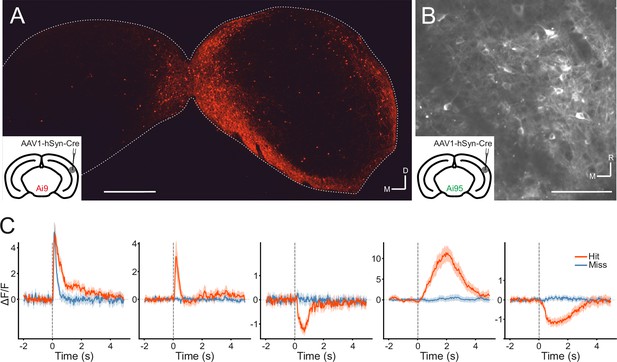
Transsynaptic targeting and two-photon calcium imaging of corticorecipient inferior colliculus (IC) shell neurons.
(A) Coronal section of the left and right IC of a tdTomato-reporter (Ai9) mouse in which AAV1.hSyn.Cre.WPRE had been injected into the right auditory cortex three weeks before perfusion. The transsynaptically transported virus drove the expression of Cre recombinase and tdTomato in neurons that receive input from the auditory cortex, including the corticorecipient neurons in the IC. tdTomato-labeled neurons were predominantly found in the shell of the ipsilateral (right) IC. Scale bar, 500 µm. (B) In vivo two-photon micrograph taken approximately 100 µm below the dorsal surface of the right IC of a GCaMP6f-reporter mouse (Ai95D) in which GCaMP6f expression had been driven in corticorecipient IC neurons by injection of AAV1.hSyn.Cre.WPRE into the right auditory cortex. See Figure 4—video 1 for the corresponding video recording. Scale bar, 100 µm. (C) Example average response profiles of five corticorecipient IC neurons for different trial outcomes. Vertical line at time 0 s indicates the time of click presentation. Shaded areas represent 95% confidence intervals.
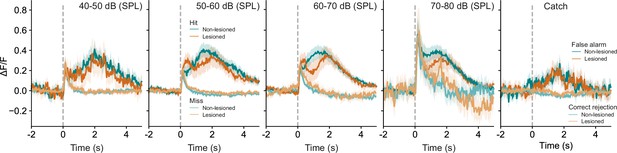
Averaged response profiles for stimulus and catch trials.
Stimulus trials are binned into four different sound level ranges and separated into hit and miss trials. Catch trials are separated into false alarms and correct rejections. Shaded areas represent 95% confidence intervals.
Two-photon calcium imaging was performed approximately 100 µm below the dorsal surface of the right inferior colliculus (IC) of a GCaMP6f-reporter mouse (Ai95D) engaged in a sound detection task.
GCaMP6f expression had been driven in corticorecipient IC neurons by injection of AAV1.hSyn.Cre.WPRE into the right auditory cortex. Video is played at twice the speed of acquisition and corresponds to the micrograph shown in Figure 4B.
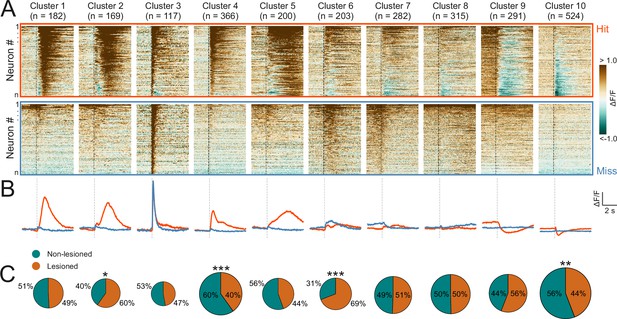
Corticorecipient inferior colliculus (IC) neurons display heterogeneous response profiles.
(A) Peri-stimulus time histograms for all neurons in the dataset separated by cluster identity: hit trials (top) vs miss trials (bottom). (B) Averaged response profiles obtained by taking the mean across all neurons in a cluster separately for hit (red) and miss (blue) trials. (C) Pie charts illustrate the proportion of neurons from lesioned and non-lesioned mice in each cluster. The size of each pie chart is proportional to the total number of neurons in each cluster. Given the unequal number of neurons from lesioned (952 neurons) and non-lesioned (1697 neurons) mice, the pie charts were normalized to the overall sample size such that a 50/50 split indicates a lesioned/non-lesioned distribution that is identical to that of the overall population. Asterisks indicate a significant difference between the lesioned/non-lesioned distribution in the given cluster and that in the overall population. *p<0.05, **p<0.01, ***p<0.001, two-sided one proportion Z-test.
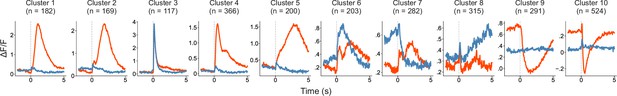
Rescaled response profiles for each cluster.
Averaged response profiles were obtained by taking the mean across all neurons in a cluster separately for hit (red) and miss (blue) trials. Same as Figure 5B except that here each panel has an individualized y-axis range.
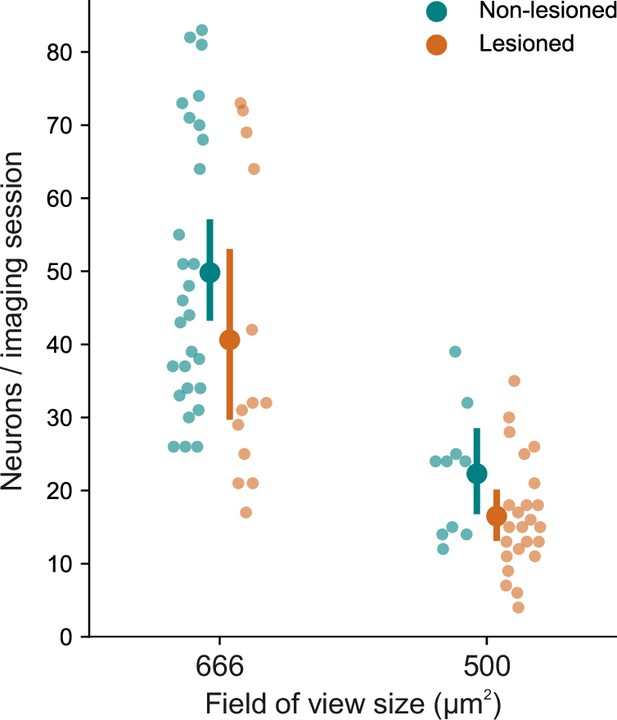
Number of sessions for each imaging field of view size.
A greater number of recordings happened to be made with a larger field of view in non-lesioned (28 of 38) than in lesioned (13 of 37) mice. Consequently, the number of neurons recorded in non-lesioned mice was greater than that recorded in lesioned mice (1697 vs 952). Error bars represent 95% confidence intervals.
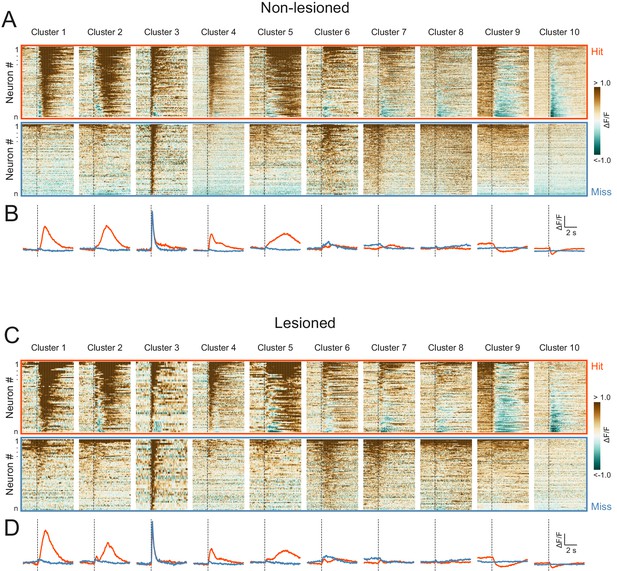
High correspondence between cluster profiles of lesioned and non-lesioned mice.
(A) Peri-stimulus time histograms for all neurons recorded in non-lesioned mice separated by cluster identity: hit trials (top) vs miss trials (bottom). (B) Averaged response profiles obtained by taking the mean across all neurons in each cluster separately for hit (red) and miss (blue) trials. (C, D) Same as (A) and (B) for neurons recorded in lesioned mice.
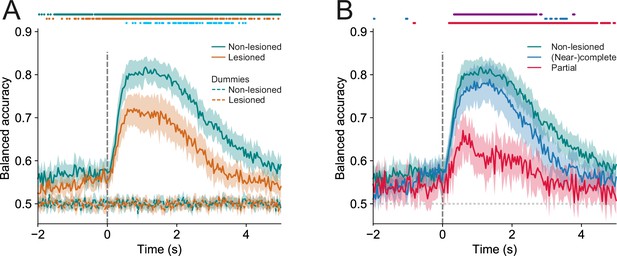
Trial outcomes can be accurately decoded from neural activity in lesioned and non-lesioned mice.
(A) Average decoding accuracy of logistic regression models as a function of time against dummy models with a score of 0.5 meaning chance performance and a score of 1 being the maximum. Data shown depict the mean model accuracy across 37 (lesioned) and 38 (non-lesioned) sessions, respectively. Dots at the top indicate the time points (frames) where the model performance was significantly different between trained and dummy models for non-lesioned mice (teal) or lesioned mice (orange) (p<0.05, one-sided Wilcoxon signed-rank test or paired t-test with Bonferroni correction, depending on whether normality assumption was met), and between the trained models for non-lesioned vs lesioned mice (blue) (p<0.05, one-sided Mann-Whitney U test or t-test with Bonferroni correction, depending on whether normality assumption was met). (B) Same as (A) but the average model accuracy is plotted separately for mice with (near-)complete (22 sessions) and partial lesions (15 sessions). Dots at the top indicate the time points where the model performance was significantly different between partial vs (near-)complete mice (purple), (near-)complete vs non-lesioned mice (blue), and partial vs non-lesioned mice (red) (p<0.05, one-sided Mann-Whitney U test or t-test with Bonferroni correction, depending on whether normality assumption was met). Shaded areas represent 95% confidence intervals.
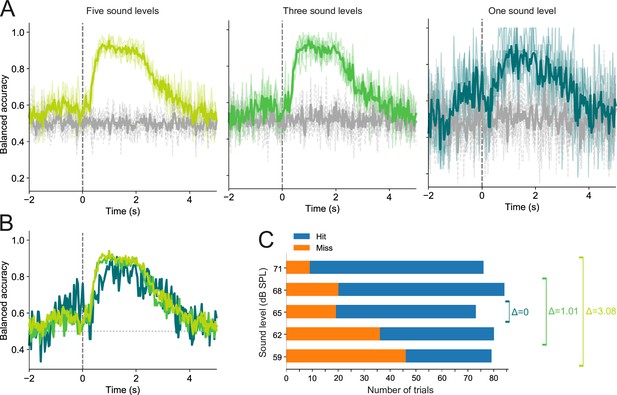
Trial outcome decoding is not meaningfully affected by differences in sound level distributions between hit and miss trials.
(A) Decoding results for one imaging session based on trials in which stimuli were presented at five (left), three (middle), or a single sound level (right). Thin colored lines show the results of each of the five cross-validation folds. Thick colored lines indicate averages across all five folds. Gray lines show results for the corresponding dummy models. (B) Superimposed averages from (A). (C) Hit and miss trial distributions for each of the five sound levels, as well as the mean sound level difference (Δ) between hit and miss trials for the three decoding conditions shown in (A) and (B). The mean difference was 3.08 dB, 1.01 dB, and 0 dB for the five, three, and one sound level conditions, respectively.
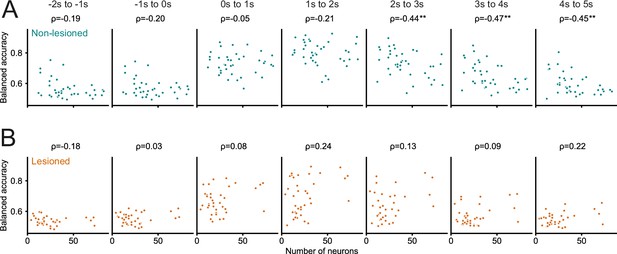
Greater number of recorded neurons was not associated with better decoding performance.
(A, B) Decoding performance (balanced accuracy) of the logistic regression models averaged over different 1 s time periods relative to stimulus onset as a function of the number of neurons recorded in a given session. A greater number of neurons obtained in a field of view was not associated with better decoding performance. Values above panels indicate Spearman’s rank correlation coefficient ρ. The only statistically significant relationship between the number of recorded neurons and decoding performance was found for late trial periods in non-lesioned mice (A), and indicated that for time periods >2 s after stimulus onset a smaller sample size was associated with better decoding performance. **p<0.01.
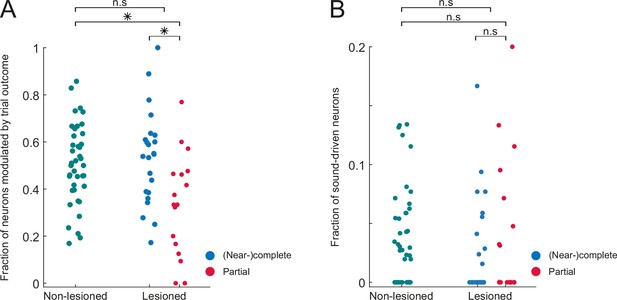
Similar fractions of task-modulated and sound-driven neurons in lesioned and non-lesioned mice.
(A) Fraction of neurons per session that exhibit a significant difference in response magnitude between hit and miss trials. (B) Fraction of neurons per session that exhibit a significant stimulus response in miss trials. *p<0.01, Mann Whitney U test.
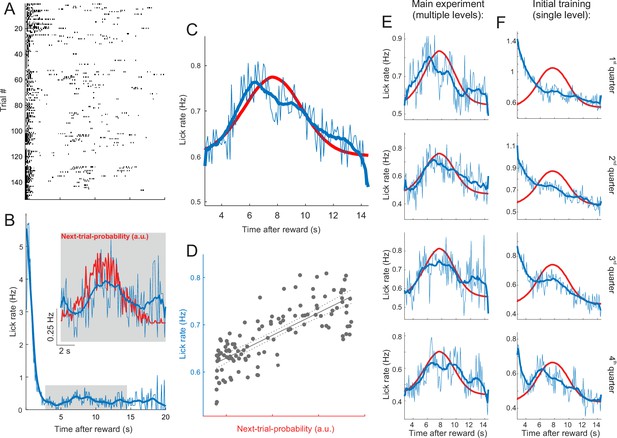
Lick rates in peri-catch trial periods approximate next-trial-probability.
(A) Peri-catch trial lick raster for all catch trials that followed a hit trial for one example mouse. The peri-catch trial period was defined as the period from the reward delivery in the hit trial to the onset of the trial following the catch trial. (B) Lick rate averaged across the peri-catch trial periods shown in (A) and binned into 100ms wide bins. The thick blue line shows the smoothed (20-point running average) lick rate. The inset gives a magnified view of the average lick rate during the period indicated by the gray rectangle. The red line illustrates the distribution of ‘reward-to-next-trial-onset’ intervals experienced by the example mouse. Given that licks are plotted time-locked to reward delivery, we plotted the distribution of intervals between reward delivery and the onset of the next trial rather than the inter-trial interval (ITI) distribution. In practice, the difference between the two is roughly the latency between the stimulus and the first lick and thus barely distinguishable at this scale. As the distribution indicates the probability of the next trial presentation as a function of time since the preceding reward delivery we refer to it as ‘next-trial-probability’. (C) Same as the inset in (B) averaged across all mice. Next-trial-probability was smoothed with a 20-point running average. (D) Next-trial probability as a predictor of lick rate. The dotted lines indicate the 95% confidence bounds around the regression fit. Adjusted R2=0.59. Although the next-trial probability is a good predictor of changes in the average lick rate, the lick rate at the peak of the distribution is merely about a quarter higher than at its tails where next-trial probability approaches zero. Furthermore, to put the average lick rates into perspective, note that mice tend to lick in bouts, typically consisting of two to six licks in very quick succession (see lick raster in (A)), and that, consequently, the lick rate exceeds the underlying bout rate by a factor of about four. (E) Same as (C) but with peri-catch trials binned into four quarters before averaging in order to illustrate changes in lick behavior across different stages of the experiment. (F) Same as (E) for all peri-catch trials during the initial training with a single-level stimulus. While the peri-catch trial lick rate profile changed substantially over the course of the initial training (F) and started to approximate the stimulus probability distribution towards the end of training, it remained broadly stable throughout the main experiment (E). In order to increase the statistical power of this analysis, we included data from several additional mice used in other projects. These additional mice received the same training and performed the same task, but differed from those in the main dataset in that they had a different genetic background and/or had been fitted with a cranial implant for cortical rather than midbrain imaging. N for panels (C–F) = 34 mice.





