Mesenchymal Meis2 controls whisker development independently from trigeminal sensory innervation
Figures
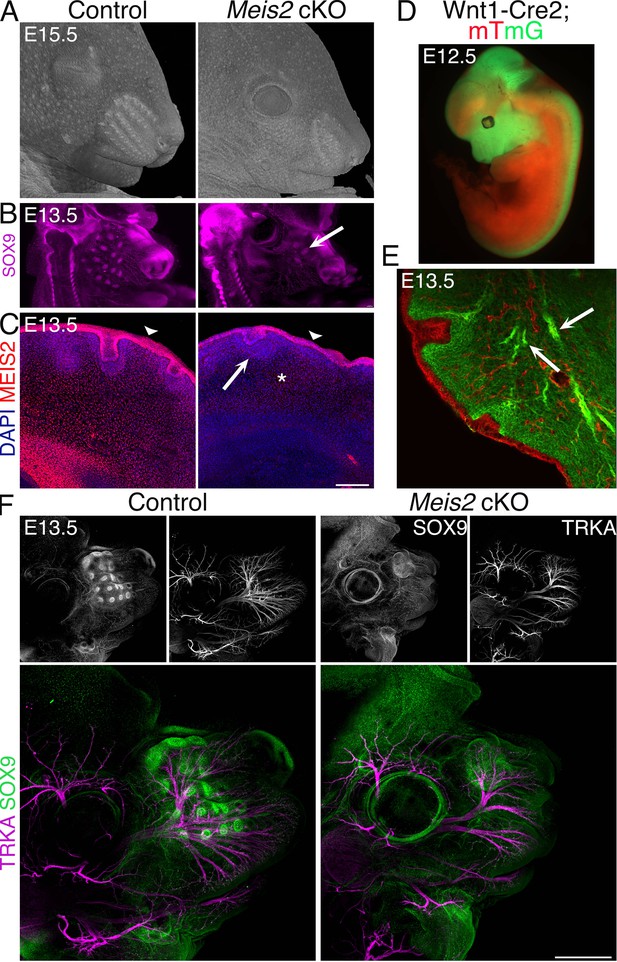
Severe whisker follicle (WF) development in Meis2 cKO embryos.
(A) Micro-computed tomography (micro-CT) images of control and Meis2 cKO embryos at embryonic day 15.5 (E15.5) showing aberrant whisker phenotype in Meis2 cKO mice. (B) Light-sheet microscopy of Sox9 whole-mount immunostaining of WF. Arrow indicates example of an ‘escaper’ whisker. (C) MEIS2 immunofluorescence on frontal frozen sections of E13.5 snouts showing MEIS2 expression in the dermis, dermal condensate (DC), and epithelium including placode (Pc). Arrowheads indicate epithelial expression of MEIS2 in both control and Meis2 cKO mice. Arrow and asterisk show disappearing MEIS2 expression in the DC and dermis, respectively, in the mutant. Scale bar: 150 μm. (D) Wnt1-Cre; mTmG embryos showing Cre recombination specificity in the craniofacial area, midbrain, and dorsal spinal cord. Recombined cells are labeled by membrane-localized GFP in green, and non-recombined cells labeled by membrane-localized tdTomato in red. (E) Frontal sections of Wnt1-Cre; mTmG snouts documenting Cre recombination in the neural crest-derived mesenchyme, cranial nerve projections without recombination in the overlying epithelium. Arrows show trigeminal (TG) nerve branches. (F) Whole-mount staining of Meis2 cKO heads at E13.5 with SOX9 and TRKA antibodies showing almost absence of WFs and compromised branching of the TG nerve in mutants. Scale bar: 500 μm.
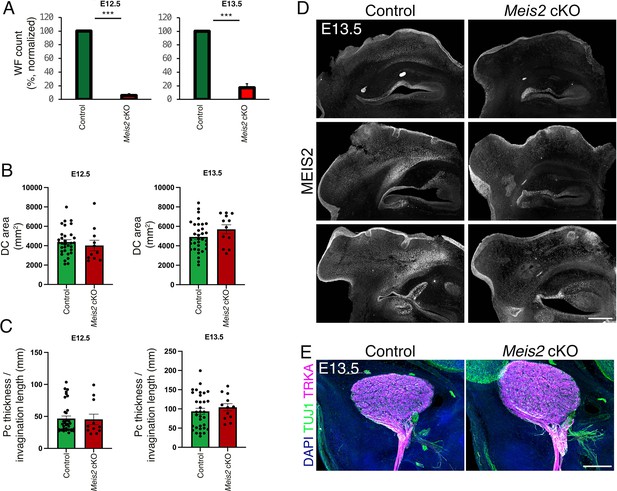
Normal sizes of escaper whisker follicles (WFs) and trigeminal (TGs) in Meis2 cKO mice.
(A) Quantification of WF numbers at embryonic day 12.5 (E12.5) (left) and E13.5 (right) showing substantial decrease in number of WFs in the Meis2 cKO mice (n=at least 3 embryos for E12.5, n=at least 8 embryos for E13.5, mean ± sem, t-test p<0.001 for both). (B–C) Quantification of 2D DC area (B) and placode (Pc) thickness of non-invaginated or length of the invaginated follicles (C) showing comparable values between control and Meis2 cKO at E12.5 (left) and E13.5 (right). N=4 embryos for E12.5 and 3 embryos for E13.5, and at least 30 WFs for controls and 10 for Meis2 cKO mice. p-Values for all graphs are larger than 0.05, Student’s t-test. Data are presented as mean ± sem. (D) Three examples of 100 μm frozen frontal sections through snout stained with MEIS2 antibody showing disappearing MEIS2 staining in the Meis2 cKO dermis. Scale bar: 300 μm. (E) 100 μm frozen sections through TG ganglia stained with TUJ1 and TRKA antibodies showing non-defective TG ganglion size or exit of the TG nerve from the ganglia. Scale bar: 250 μm.
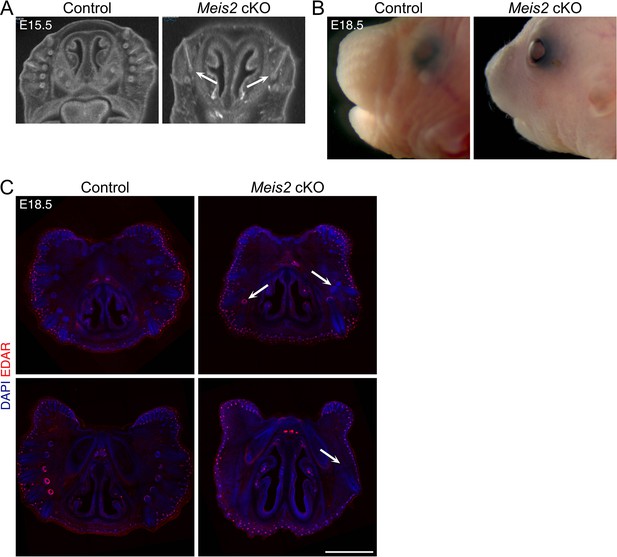
Whisker phenotype persists at embryonic day 18.5 (E18.5) Meis2 cKO mice.
(A) Micro-computed tomography (micro-CT) images through the snout of E15.5 control and Meis2 cKO mice showing abnormal whisker phenotype in the mutant. Arrows show some escaper whisker follicles (WFs). (B) Bright-field images of control and Meis2 cKO mice at E18.5. (C) Two examples of 100 μm frozen sections of snouts from E18.5 control and Meis2 cKO stained with DAPI (blue) and EDAR (red) showing only a few whiskers in the mutant. In contrast, EDAR-labeled hair follicles appeared to be normal in the mutant. Arrows show some escaper WFs. Scale bar: 1 mm.
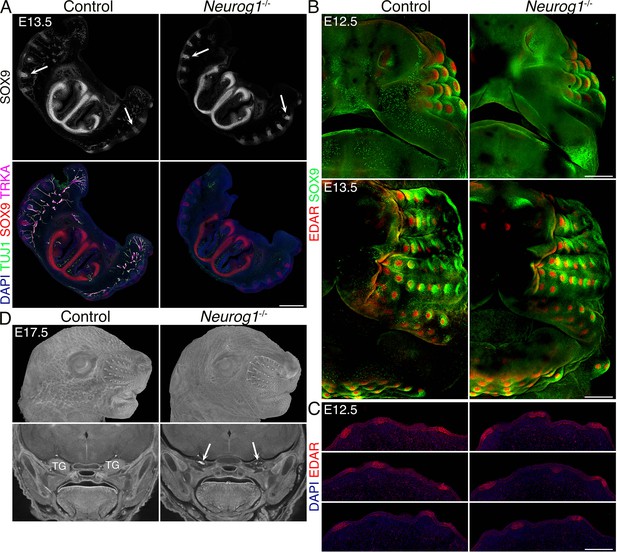
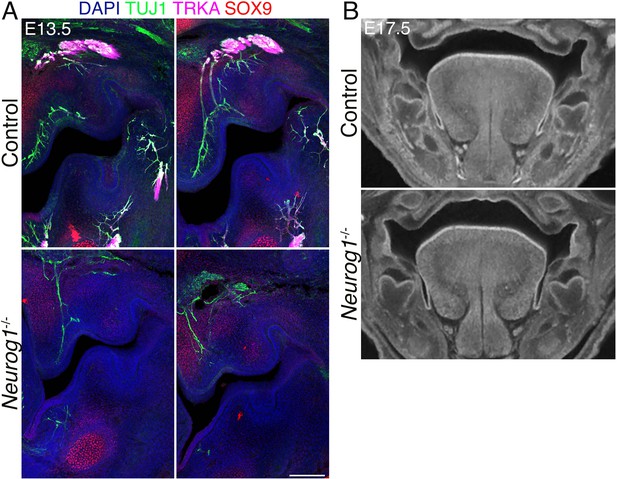
Neurog1-/- (KO) embryos lack the trigeminal (TG) nerve, but whisker follicle (WFs) development is normal.
(A) Triple immunostaining of 100 μm sections shows the absence of TG nerve projections (TUJ1+, TRKA+) and normal WF (SOX9+) in Neurog1-/- mice. Arrows in black and white images show examples of invagination of whisker placodes labeled by SOX9 antibody. Scale bar: 500 μm. (B) Whole-mount immunostaining of WFs with SOX9 and EDAR antibodies showing normal WF morphology and patterning in mutants at embryonic day 12.5 (E12.5) (top) and 13.5 (bottom). Scale bars: 300 μm. (C) EDAR staining of 10 μm sections at E12.5 showing normal initiation of placode formation in Neurog1 mutants. Scale bar: 150 μm. (D) Micro-computed tomography (micro-CT) images reveal normally developed whiskers (top) at E17.5 in mutants while TGs are lacking (bottom, arrows).
Normal tooth development in Neurog1-/- mice.
(A) 100 μm frozen sections of molar teeth of control and Neurog1-/- mice at embryonic day 13.5 (E13.5) stained with SOX9, TUJ1, and TRKA antibodies showing the normal phenotype of the mutant. Scale bar: 150 μm. (B) Micro-computed tomography (micro-CT) images around the molar tooth level at E17.5 in Neurog1-/- mice.
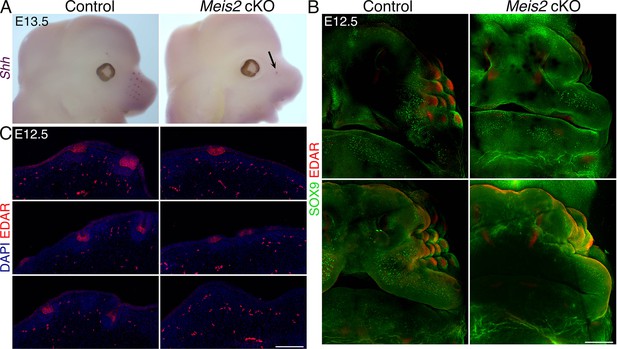
Induction and progression of whisker follicle (WF) development is compromised in Meis2 cKO.
(A) Whole-mount in situ hybridization of Shh mRNA documenting loss of WFs in mutants. Arrow shows an escaper whisker. (B) Whole-mount immunostaining of WFs with SOX9 and EDAR antibodies showing absence of WFs in Meis2 cKO at embryonic day 12.5 (E12.5). Two examples for each genotype are shown. Scale bar: 300 μm. (C) EDAR staining of 10 μm sections showing placode formation arrest in mutants. Scale bar: 150 μm.
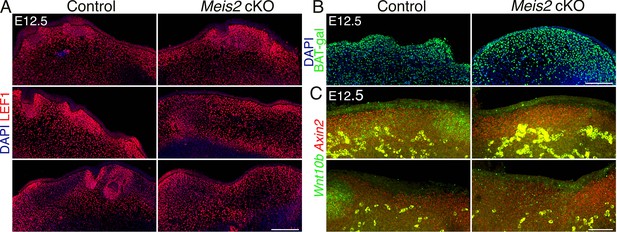
WNT signaling in the epithelium is affected by deletion of Meis2 in the dermal mesenchyme.
(A) LEF1 staining of 10 μm paraffin frontal sections of snouts showing abundance of LEF1 in the dermal mesenchyme, while in the epithelium, it is concentrated in placodes in regions of whisker follicle (WF) appearance. Similar to missing placodes, LEF1 is also lost in Meis2 cKO epithelium. Scale bars: 150 μm. (B) Beta-galactosidase immunostaining of sections from WNT reporter BAT-gal controls and Meis2 cKO; BAT-gal mutants showing widespread galactosidase signal in the dermis and epithelium. Scale bars: 150 μm. (C) In situ hybridization HCR-FISH using probes for Axin2 and Wnt10b. In the epithelium, Wnt10b is detected in placodes, and the signal is lost in Meis2 cKO. Similarly, upregulation of Axin2 is not observed in the expected WF loci. In the dermal mesenchyme, Axin2 mRNA is detected throughout the dermis regardless of WFs. This pattern was not changed in Meis2 cKO. Scale bar: 50 μm.
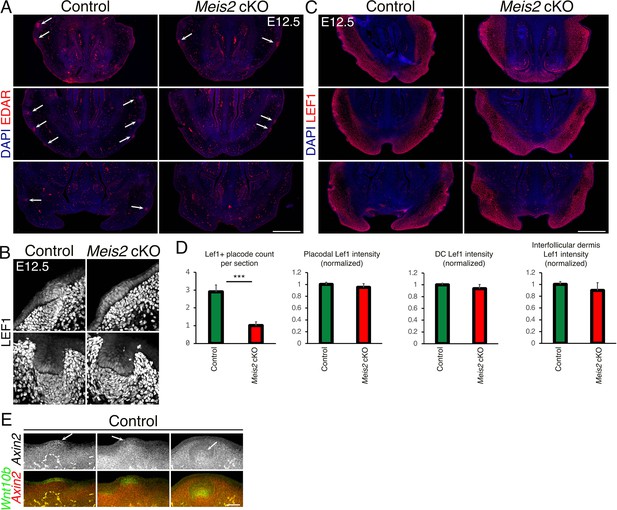
EDAR and LEF1 expression in Meis2 cKO mice.
(A) Three examples of 10 μm FFPE sections stained with EDAR antibody showing affected expression of EDAR in Meis2 cKO mice. Arrows indicate EDAR-positive sites which are observed less frequently in the mutant snout. Scale bar: 500 μm. (B) Two representative images of normally developed escaper whisker follicles (WFs) in the mutants by LEF1 staining. LEF1 expression is similar in just forming placodes (top) and invaginated (bottom). Additionally, LEF1 expression shows a decline in the DC regions compared to peri-DC. Scale bar: 30 μm. (C) Three representative micrographs of LEF1-stained 10 μm FFPE snout sections showing normal expression of LEF1 in the dermis but the decreased number of LEF1+ placodes in the epithelium of the mutant snout. Scale bar: 500 μm. (D) Quantification of Lef1+ placode count and Lef1 fluorescence intensities in placodes, DCs, or interfollicular upper dermis. Data are presented as mean ± sem, ***p=0.0002. (E) In situ hybridization HCR-FISH using probes for Axin2 and Wnt10b in control snout at embryonic day 12.5 (E12.5) showing placodal expression of Wnt10b (green) and Axin2 (arrows, red) as well as widespread expression of Axin2 in the dermis. Scale bar: 60 μm.
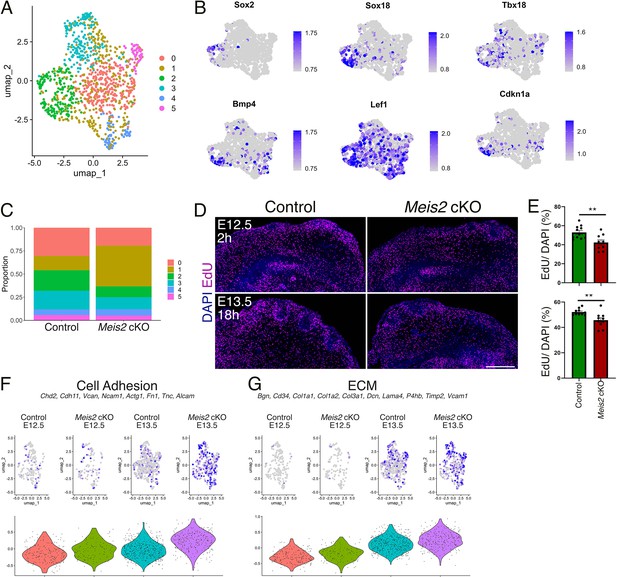
Dermal condensation and proliferation are reduced in Meis2 cKO.
(A) Uniform Manifold Approximation and Projection (UMAP) diagram of single-cell RNA-seq (scRNA-seq) analysis of the dermal mesenchyme subset with six-cluster resolution in which cluster 2 (green) represents dermal condensate (DC) of whisker follicles (WFs). (B) UMAP representations of typical DC markers Sox2, Sox18, Tbx18, Bmp4, Lef1, and Cdkn1 shown by FeaturePlots. (C) Relative cell numbers in six clusters showing a higher cell count in cluster 1 (nonspecialized cells) at the expense of clusters 0 (dividing cells) and 2 (DC). (D) Analysis of cell proliferation by EdU incorporation after a 2 hr pulse at embryonic day 12.5 (E12.5) and an 18 hr pulse at E13.5. (E) Overall proliferation rate was reduced in Meis2 cKO from 52.96±2.05% to 42.38 ± 2.77% (mean ± sem, t-test p=0.0057) in 2 hr pulse (top) and from 52.17±0.81% to 45.76 ± 1.85% (mean ± sem, t-test p=0.0052) in 18 hr pulse (bottom). Each data point indicates the average value for one section. n=2 mice and at least nine sections. (F) Increase of cell adhesion module score in mutants at E13.5 by scRNA-seq analysis. Module scores are generated by indicated genes. (G) Increase of extracellular matrix (ECM) module score in mutants at E13.5 by scRNA-seq analysis. Module scores are generated by indicated genes.
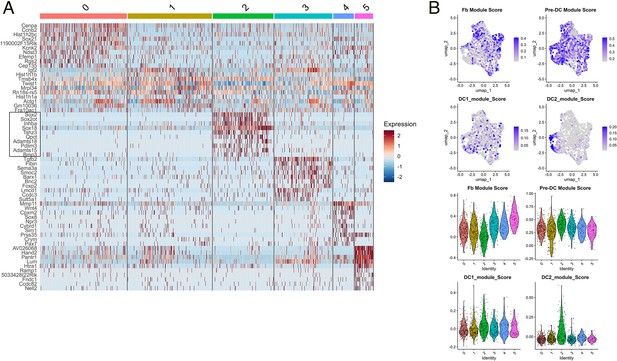
Identification of dermal condensate (DC) cluster in single-cell RNA sequencing (scRNA-seq) datasets.
(A) Heatmap of top 10 markers for each identified cluster in scRNA-seq data. Each column represents individual cells, whereas each row represents indicated marker genes on the left. Genes in the black box are top markers of cluster 2 (DC). (B) FeaturePlots and VlnPlots of fibroblasts (Fb), pre-DC, DC1, and DC2 module scores in each cluster. See Materials and methods for selected genes used for generating module scores.
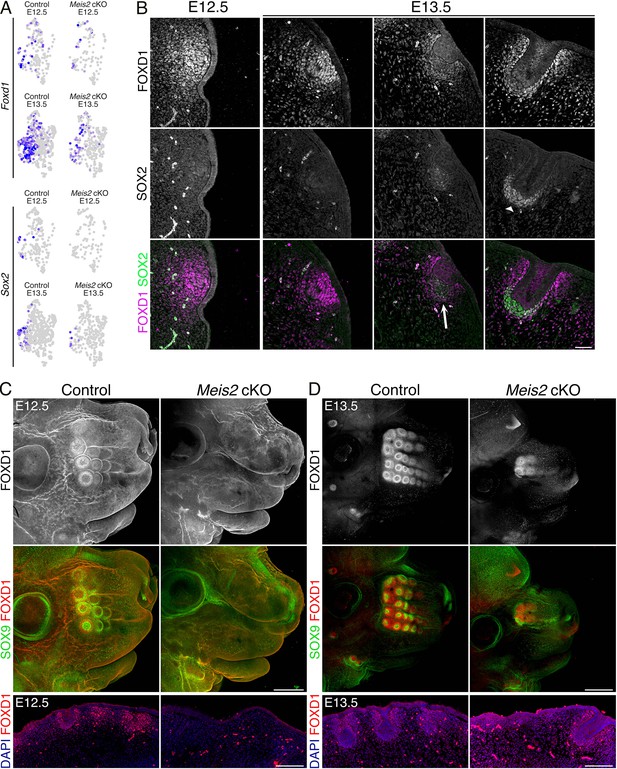
Expression of the earliest pre-dermal condensate (DC) and DC marker Foxd1 is lost in Meis2 cKO.
(A) Uniform Manifold Approximation and Projection (UMAP) representations of decline in Foxd1 (top) and Sox2 (bottom) expression in DC cluster (#2) shown by FeaturePlots. Expression of both genes is significantly reduced in mutants. (B) Immunofluorescence of FOXD1 and SOX2 at initial stages of whisker follicle (WF) showing FOXD1 in the pre-DC stage while the expression shifts from DC to peri-DC regions during WF progression. SOX2 appears as a typical DC marker with no earlier expression. Scale bar: 50 μm. (C–D) Whole-mount immunostaining of FOXD1 and SOX9 of heads from controls and Meis2 cKO at embryonic day 12.5 (E12.5) (C) and E13.5 (D). It shows the absence of WFs including the pre-DC marker FOXD1 at the E12.5 stage when proximal columns of WFs have already formed. A few WF escapers develop in mutants at E13.5 in proximal regions while at least six WF columns have formed in controls. Scale bars: 400 μm (C) and 500 μm (D). Lower panels: FOXD1 staining in FFPE sections. Scale bars: 150 μm.
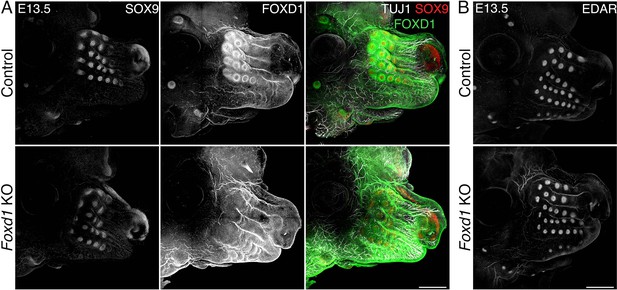
Whisker follicles (WFs) normally develop in loss-of-function Foxd1 mutants.
(A) Whole-mount immunostaining of FOXD1, TUJ1, and SOX9 of heads from controls and Foxd1-null mutants at embryonic day 13.5 (E13.5) showing normal formation of WFs in mutants in which FOXD1 signal disappears. Normal WF development is also reflected in normal WF innervation represented by TUJ1 staining. Scale bars: 500 μm. (B) Whole-mount immunostaining of EDAR confirmed normal placode (Pc) appearance in Foxd1-null mutants. Scale bars: 500 μm.
Videos
Embryonic day 13.5 (E13.5) whole-mount half head of a control mouse stained with TRKA antibody and scanned with a spinning disk microscope at 1 μm z-step size.
Embryonic day 13.5 (E13.5) whole-mount half head of a Meis2 cKO mouse stained with TRKA antibody and scanned with a spinning disk microscope at 1 μm z-step size.
Compared to its control littermate in Video 1, peripheral trigeminal (TG) nerve branching is considerably defected in the mutant.
Additional files
-
Supplementary file 1
Seurat cluster markers of dermal fibroblast.
List of markers of all identified clusters for integrated samples of dermal fibroblasts from embryonic day 12.5 (E12.5) and E13.5 Meis2 cKO mice and their control littermates extracted by FindAllMarkers Seurat function. Dermal fibroblast cell cluster is subset using single-cell RNA sequencing (scRNA-seq) data by Hudacova et al., 2025.
- https://cdn.elifesciences.org/articles/100854/elife-100854-supp1-v1.xlsx
-
Supplementary file 2
clusterProfiler gene ontology (GO) for cluster 0 markers.
clusterProfiler GO in biological processes run by using top 100 markers of cluster 0 of dermal fibroblast cell cluster from embryonic day 12.5 (E12.5) and E13.5 Meis2 cKO and their control litters. GO terms were extracted by enrichGO function with default parameters.
- https://cdn.elifesciences.org/articles/100854/elife-100854-supp2-v1.xlsx
-
Supplementary file 3
DEGs between dermal condensate (DC) cluster (cluster 2) of control and Meis2 cKO mice.
DEGs between DC cluster of integrated embryonic day 12.5 (E12.5) and E13.5 Meis2 cKO mice and their control littermates, extracted by FindMarkers Seurat function where ident.1 and ident.2 arguments were set to control cluster 2 and Meis2 cKO cluster 2, respectively.
- https://cdn.elifesciences.org/articles/100854/elife-100854-supp3-v1.xlsx
-
MDAR checklist
- https://cdn.elifesciences.org/articles/100854/elife-100854-mdarchecklist1-v1.docx





