The skin is a significant but overlooked anatomical reservoir for vector-borne African trypanosomes
Figures
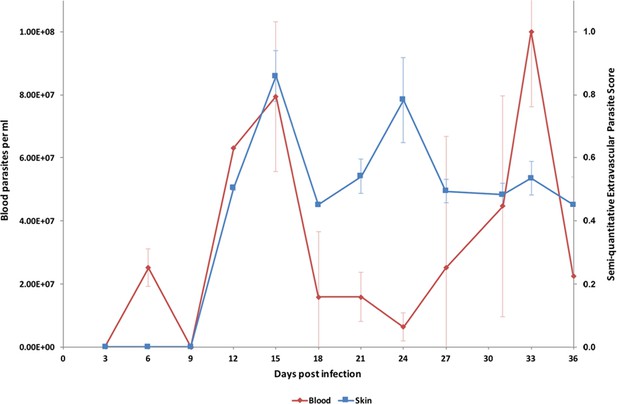
Parasite densities in the blood and in the extravascular tissue of the skin over a time-course.
The blood parasitaemia of T.b. brucei strain STIB247 (red) and the semi-quantitative score of extravascular parasites in the skin (blue) are shown over a 36-day time-course following infection in Balb/C mice. Blood parasitaemia was measured using phase microscopy using methodology outlined in (Lumsden, 1963). Skin parasite burden is an average of five high-power fields scored by histological analysis (0 = no parasites detectable; 1 = low numbers of parasites; 2 = moderate numbers of parasites; 3 = large numbers of parasites). Standard error shown (n = 3).
-
Figure 1—source data 1
Semi-quantitative evaluation of the parasite burden in skin sections (STIB247) Every three days for 36 days of a STIB247 T.b.
brucei infection, five mice were culled (three infected, two control) and skin sections stained with parasite-specific anti-IGS65 antibody. Parasite burden was assessed by two pathologists blinded to group assignment and experimental procedures. Presence of parasites defined as intravascular (parasites within the lumen of dermal or subcutaneous small to medium-sized vessels) and extravascular (parasites located outside blood vessels, scattered in the connective tissue of the dermis or in the subcutis) was evaluated in 5 high-power fields at x40 magnification with a 0 to 3 semi-quantitative grading scale (0 = no parasites detectable; 1 = low numbers of parasites; 2 = moderate numbers of parasites; 3 = large numbers of parasites).
- https://doi.org/10.7554/eLife.17716.003
-
Figure 1—source data 2
Daily parasitaemia during STIB247 infection in Balb/C mice The daily parasitaemia during a 36-day STIB247 T.b.
brucei infection was estimated using phase microscopy and methodology outlined in (Lumsden, 1963).
- https://doi.org/10.7554/eLife.17716.004
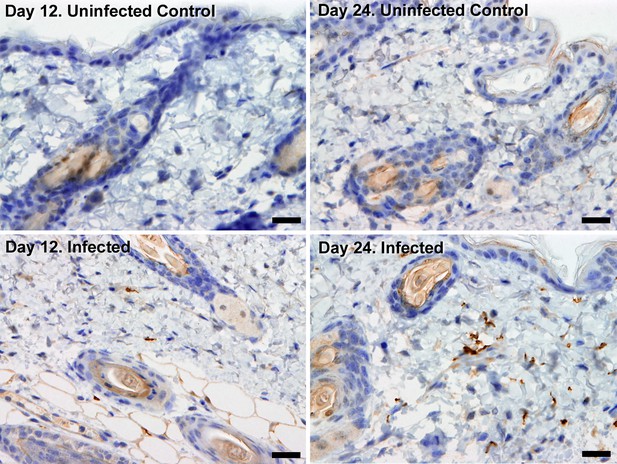
Extravascular localisation of trypanosomes during an infection.
Histological sections of dorsal skin from uninfected and infected Balb/C mice stained with trypanosome-specific anti-ISG65 antibody (brown), counterstained with Gill’s Haematoxylin stain (blue) at 12 days and 24 days post-inoculation with T.b. brucei strain STIB247. Parasites are visible in extravascular locations of the skin including the deep dermis and subcutaneous adipose tissue from day 12. The scale bar represents 20 µm.
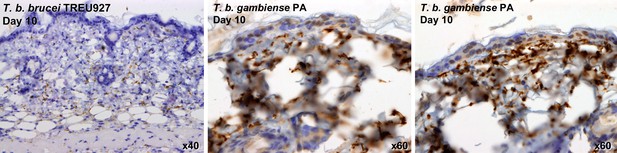
Skin invasion by T.b. brucei strain TREU927 and T.b. gambiense strain PA.
Histological sections of dorsal skin from a mouse infected with T.b. brucei strain TREU927 at 20x magnification and two mice infected with T.b. gambiense strain PA at 40x magnification 10-days post-inoculation. Trypanosome-specific anti-ISG65 antibody reveals the presence of extravascular parasites (brown) and the slides were counterstained with Gill’s Haematoxylin stain (blue) to reveal host skin structure.
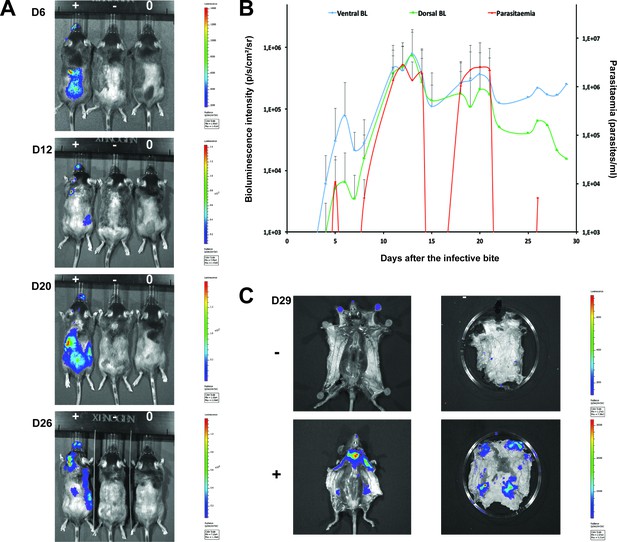
Dynamics of parasite distribution in the extravascular tissue of the skin and in the blood during a representative course of infection following natural transmission.
A total of seven mice were infected by the single infective bite of an individual G.m. morsitans on the belly with the T.b. brucei AnTat1.1E AMLuc/tdTomato strain. Panels A and C depict representative patterns. (A) Examples of bioluminescence profiles of 3 mice (+ bitten by an infected fly, - bitten by an uninfected fly and 0 not bitten) 6, 12, 20 and 26 days after the bite are shown. (B) Ventral (blue) and dorsal (green) bioluminescence (BL) intensities (in p/s/cm2/sr on the left Y-axis) and parasitaemia (in parasites/ml in red on the right Y-axis) were measured daily for 29 days and plotted as mean ± SD (n = 7 mice). (C) The entire skins of mice (+) and (-) were dissected for bioluminescence imaging 29 days after the bite. For the mouse (+), Figure 3—figure supplement 1 shows the bioluminescence profile of dissected organs, Figure 3—figure supplement 2 presents the skin inflammation, and Figure 3—figure supplement 3 shows labelled parasites in skin sections.
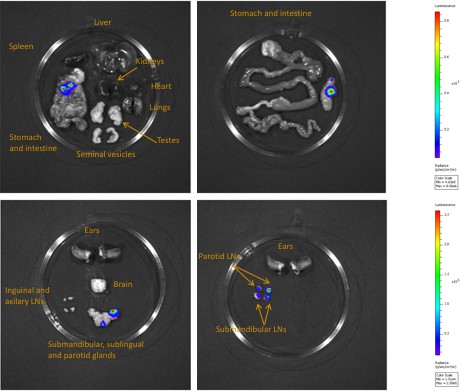
Bioluminescence mostly originates from parasites in the skin.
Mouse (+) was sacrificed and dissected for bioluminescence imaging 29 days after the infective bite. Figure 3C shows the bioluminescence profile of its entire skin and dissected organs are shown here.
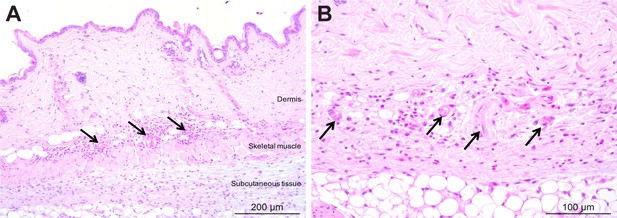
Mild inflammation of skin tissues one month after an infection by natural transmission.
After 29 days, the most bioluminescent skin region of mouse (+) was dissected, fixed in paraformaldehyde, embedded in paraffin and stained with HE. Multifocal inflammatory infiltrates containing neutrophils were located in the dermis and subcutaneous tissue and associated with oedema (arrows in A). Inflammatory foci were generally centred on blood vessels (arrows in B).
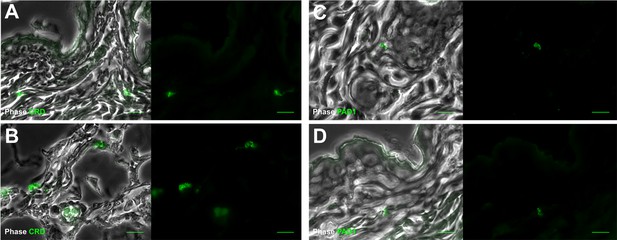
Extravascular parasites in the skin express both VSGs and PAD1 surface markers.
After 29 days, the most bioluminescent skin region of mouse (+) was dissected, fixed in paraformaldehyde, embedded in paraffin and treated for IFA with the anti-CRD antibody that predominately labels parasites expressing VSGs (A–B), or the anti-PAD1 antibody specific to transmission form 'stumpy' cells (C–D).
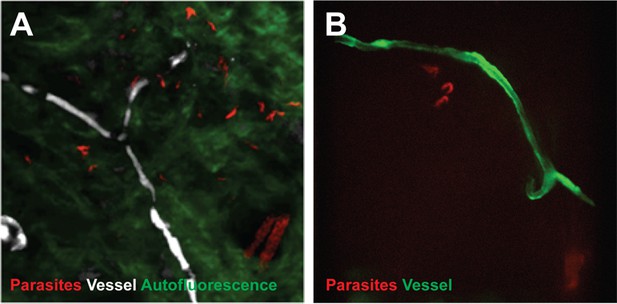
Extravascular localisation of trypanosomes during an infection visualised using multi-photon microscopy (A) and spinning-disk confocal microscopy (B).
(A) Still-image extracted from video (Video 1) of multi-photon live imaging of dorsal skin during a trypanosome infection. Intravenous non-targeted quantum dots (white) highlight blood vessels. T.b. brucei STIB 247 parasites transfected with mCherry to aid visualisation (red) are clearly visible and motile outside the vasculature and within the extravascular skin matrix (green). (B) Still-image extracted from (Video 3) of spinning-disk confocal live imaging of the ear of an Kdr (Flk1) C57BL/6J Rj mouse during a trypanosome infection. T.b. brucei AnTat1.1E AMLuc/tdTomato parasites expressing tdTomato (red) are moving in the extravascular region surrounding a vessel of the dermis (green).
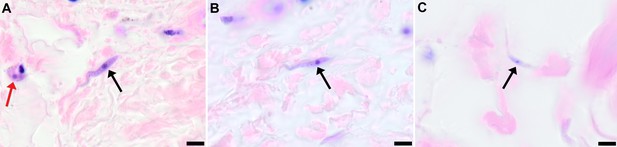
Extravascular localisation of trypanosomes in previously unidentified human cases of trypanosomiasis
Histological sections of skin collected from previously unidentified cases of human trypanosomiasis from the Democratic Republic of Congo, showing the presence of extravascular parasites in biopsies from three individuals (A, B and C). Skin biopsies were collected as part of a national onchocerciasis screening programme that took place in the same geographic region as an active trypanosomiasis focus. Slides were stained with Giemsa and examined under oil immersion at 100x magnification. In addition to visible slender forms (black arrows) in the extravascular tissue of the skin, a clearly identifiable stumpy transmission form with typical morphology and an unattached undulating membrane is also present in the skin of one individual (red arrow in A). The scale bar represents 5 µm.
Videos
Extravascular trypanosomes visualised in the skin using 2-photon microscopy.
Intravital multi-photon imaging of the flank skin during trypanosome infection 10 days after IP inoculation. An intravenous injection of non-targeted quantum dots prior to imaging allowed visualisation of blood vessels. mCherry STIB247 T.b. brucei parasites (red) are observed moving in the extravascular region surrounding blood vessels of the dermis (white). Collagen auto-fluorescence is visible as green.
Extravascular trypanosomes visualised in the skin using spinning-disk confocal microscopy.
Spinning-disk confocal live imaging of the ear of anKdr (Flk1) C57BL/6J Rj mouse during a trypanosome infection after natural transmission. T.b. brucei AnTat1.1E AMLuc/tdTomato parasites expressing tdTomato (red) are observed moving in the extravascular region surrounding blood and / or lymphatic vessels of the dermis (green).
Extravascular trypanosomes visualised in the skin using spinning-disk confocal microscopy.
https://doi.org/10.7554/eLife.17716.014Extravascular trypanosomes visualised in the skin using spinning-disk confocal microscopy.
https://doi.org/10.7554/eLife.17716.015Tables
Skin parasites are ingested during tsetse pool-feeding. Mice were IP infected with T.b. brucei AnTat1.1E AMLuc/TY1/tdTomato and the parasitaemia and bioluminescence were monitored daily until the day of xenodiagnosis. The number of parasites in the blood was determined using a haemocytometer or a flux cytometer. The number of parasites in the skin was estimated from the measured bioluminescence intensity by using a standard curve (Table 1—source data 1 and Table 1—source data 2). Batches of teneral flies were fed on different skin regions of mice infected with differing levels of bioluminescence across the skin and with differing levels of parasitaemia (Table 1—source data 2 and Table 1—source data 3). Fly batches A4A, A2A, B1A, 3B and 3A were used to assess tsetse transmission in hosts with low numbers of blood parasites but high numbers skin parasites, while fly batches 1B, 1A, 4B, 4A, 2B, 2A, B2B and B4A were used to investigate the impact of high numbers of parasites in both the skin and blood. Flies were dissected and their midguts checked for the presence of fluorescent trypanosomes after two days to determine the proportion of infected flies (Table 1—source data 4A–B). For some of these experiments, results of an in-depth quantification of parasite stages by IFA is provided in Supplementary file 4. Stumpy forms were observed only in the blood of mice with parasitaemia values highlighted in purple. Bioluminescence was detected in the skin of mice with values highlighted in grey.
| Fly batches | Parasites in blood (per ml) | Parasites in skin (per cm2) | Dissected flies | Fly infection rates (%) |
|---|---|---|---|---|
| C1 | 0 | 0 | 32 | 0% |
| C | 0 | 0 | 8 | 0% |
| A4B | < 104 | < 103 | 16 | 0% |
| B1B | 2.2 × 104 | < 103 | 13 | 0% |
| A4A | < 104 | 6.6 × 105 | 17 | 35% |
| A2A | 1.1 × 104 | 3.8 × 106 | 7 | 86% |
| B1A | 2.2 × 104 | 4.6 × 107 | 16 | 31% |
| 3B | 4.4 × 104 | 2.6 × 104 | 14 | 36% |
| 3A | 4.4 × 104 | 2.6 × 104 | 16 | 38% |
| 1B | 1.8 × 105 | 8.0 × 103 | 12 | 67% |
| 1A | 1.8 × 105 | 8.0 × 103 | 14 | 79% |
| 4B | 2.2 × 105 | 1.2 × 104 | 17 | 53% |
| 4A | 2.2 × 105 | 1.2 × 104 | 18 | 56% |
| 2B | 1.6 × 106 | 8.0 × 103 | 14 | 36% |
| 2A | 1.6 × 106 | 3.2 × 104 | 18 | 39% |
| B4B | 4.3 × 106 | 6.7 × 107 | 10 | 80% |
| B4A | 4.3 × 106 | 6.7 × 107 | 17 | 100% |
-
Table 1—source data 1
Characterisation of the AnTat 1.1E AMLuc/TY1/tdTomato sub-clone.
(A) The in vitro growth of the selected AnTat1.1E AMLuc/TY1/tdTomato sub-clone (red) was similar to that of the parental wild-type strain (blue). Bloodstream forms were cultured in HMI11, counted daily in a Muse cytometer (Merck-Millipore) and diluted after 4 days. (B) A parasite density / bioluminescence intensity analysis was performed by measuring the bioluminescence in successive 2-fold dilutions in 96-micro-well plates with an IVIS Spectrum imager (Perkin Elmer). When plotted as mean ± SD (n = 3), parasite densities and bioluminescence intensities were correlated when the bioluminescence levels were higher than 10 (Berthier et al., 2016) p/s/cm²/sr, corresponding to about 10 (Koffi et al., 2006) parasites, allowing estimation of the parasite density from in vivo imaging over this threshold. This standard curve was used to estimate the number of parasites in the skin from measured values of bioluminescence. (C) This correlation was verified by quantification in a microplate reader Infinite 200 (Tecan) at the very beginning of the first in vivo experiment as well as the end of the last one (mean ± SD, n = 3).
- https://doi.org/10.7554/eLife.17716.017
-
Table 1—source data 2
Parasite densities in extravascular tissue of the skin and in the blood of mice used for differential xenodiagnosis.
Mice were injected IP with AnTat1.1E AMLuc/TY1/tdTomato and monitored daily for bioluminescence and parasitaemia. (A) Bioluminescence profile of four mice (- uninfected control and (1–3) three infected mice) four days after infection. (B) The entire skins of the uninfected control mouse (-) and mouse 3 were dissected for bioluminescence imaging four days after infection. (C) Parasite densities in the blood and in the skin (calculated from the mean dorsal bioluminescence intensity measurement and from the standard curve in Table 1—source data 1, in parasites/cm (Fakhar, 2013) in blue) were calculated daily over one week and plotted as mean ± SD (n = 13 mice).
- https://doi.org/10.7554/eLife.17716.018
-
Table 1—source data 3
Skin parasites are sufficient to initiate a tsetse infection.
Schematics summarising the principal results from the xenodiagnosis experiment. In a mouse with no detected transmissible parasites in the blood (absence of stumpy forms by IFA and absence of infection of flies fed on a non-bioluminescent region of the skin), flies can ingest transmissible parasites from the bioluminescent region of the skin (left panel). When a mouse presents transmissible forms in the blood, fly infection rates increase with the concomitant ingestion of parasites from the skin (right panel). Values correspond to those obtained for mouse A4 and B4.
- https://doi.org/10.7554/eLife.17716.019
-
Table 1—source data 4
Parasite stage determination by labelling of specific surface markers.
Parasites recovered from infected tsetse midguts (A–B) or included in bloodsmears (C) were fixed in methanol for 5 s and stained either with the anti-GPEET antibody detecting early procyclic forms (red in A–B) and the L8C4 antibody labelling the flagellum PFR (green in A–B), or with the anti-PAD1 antibody detecting intermediate and stumpy forms (green in C), respectively.
- https://doi.org/10.7554/eLife.17716.020
Additional files
-
Supplementary file 1
Histopathological assessment of inflammation in the skin during STIB247 infection.
The extent of cutaneous inflammatory cell infiltration during the 36-day STIB247 experiment was assessed on haematoxylin and eosin stained sections with a semi-quantitative scoring system applied by two pathologists blinded to group assignment and experimental procedures.The extent of mixed inflammatory cell infiltration in the dermis and/or subcutis was assessed on a 0 to 3 grading scale (0 = no inflammation or only few scattered leukocytes; 1 = low numbers of inflammatory cells; 2 = moderate numbers of inflammatory cells; 3 = large numbers of inflammatory cells). Ten high-power fields (HPFs) were scored for each skin sample.
- https://doi.org/10.7554/eLife.17716.022
-
Supplementary file 2
Histopathological assessment of inflammation in the skin during TREU927 infection.
The extent of cutaneous inflammatory cell infiltration at day 10 of infection by strain TREU927 experiment was assessed on haematoxylin and eosin stained sections with a semi-quantitative scoring system applied by two pathologists blinded to group assignment and experimental procedures. The extent of mixed inflammatory cell infiltration in the dermis and/or subcutis was assessed on a 0 to 3 grading scale (0 = no inflammation or only few scattered leukocytes; 1 = low numbers of inflammatory cells; 2 = moderate numbers of inflammatory cells; 3 = large numbers of inflammatory cells). Ten high-power fields (HPFs) were scored for each skin sample.
- https://doi.org/10.7554/eLife.17716.023
-
Supplementary file 3
Expression of PAD1 relative to ZFP3.
The relative abundance stumpy cells in the skin of three BALB/c was estimated using qPCR at day 11 post-inoculation with T.b. brucei strain TREU927. Mice were culled and perfused to remove blood parasites and a 2 cm (Fakhar, 2013) region of skin removed from the flank. The tissue was homogenised and RNA extracted. 100 ng of RNA from each sample was reverse-transcribed and qPCR performed to estimate the cycle thresholds (CT) of the stumpy marker PAD1 and the endogenous control ZFP3. As CT is inversely proportional to amount of target cDNA in the sample and PAD1 and ZFP3 have similar qPCR efficiencies, a comparison of the delta (∆) of CT between PAD1 and ZFP3 transcripts reveals the relative ratio of PAD1 to ZFP3 transcripts and hence the proportion of differentiated parasites transcribing the PAD1 gene.
- https://doi.org/10.7554/eLife.17716.024
-
Supplementary file 4
Both the respective densities and the proportions of transmissible forms of parasites in the skin and in the blood govern the tsetse infection rates during pool feeding.
For some of the xenodiagnosis experiments shown in Table 1, identification and quantification of parasite stages was performed by IFA on blood smears and skin sections.Stumpy forms were observed only in the blood of mice with parasitaemia values highlighted in light grey. Bioluminescence was detected in the skin of mice with values highlighted in dark grey. The number of parasites in the skin was calculated according to the values obtained in the standard in vitro assay (Table 1—source data 1) and is therefore probably an underestimate. Tsetse flies were dissected 2 days after xenodiagnosis. Populations of intermediate and stumpy form cells were assessed in blood smears and in successive skin sections stained either with the anti-CRD antibody or the anti-PAD1 antibody (see Materials and method section). Populations of early procyclic cells were assessed in dissected fly midguts stained with the anti-GPEET antibody (see Materials and method section). ND: not determined.
- https://doi.org/10.7554/eLife.17716.025





