Activation of targetable inflammatory immune signaling is seen in myelodysplastic syndromes with SF3B1 mutations
Figures
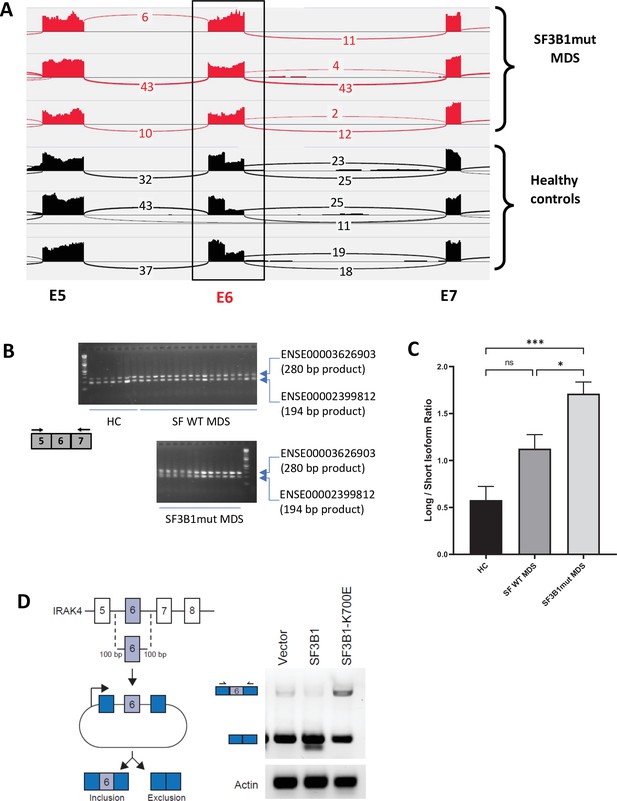
SF3B1 mutations are associated with inclusion of exon 6 in myelodysplastic syndromes (MDS) .
(A) Sashimi plots representing full length IRAK4 exon 6 inclusion or exclusion in CD34+ cells from healthy controls (HC) (n=3), and from MDS patients with mutation in SF3B1 (MDS-SF3B1mut; n=3) based on RNA-sequencing junction reads. (B) RT-PCR analysis of CD34+ cells from HC (n=7), from MDS patients with no splicing factor mutations (MDS-SFWT; n=17), and from MDS patients with mutation in SF3B1 (MDS-SF3B1mut; n=12) using primers flanking IRAK4 exon 6. (C) Densitometric quantification of IRAK4 exon 6 inclusion calculated as the ratio of the long isoform versus the short isoform from panel B. Data represent the mean ± SEM. P-values were obtained using Kruskal–Wallis test with Dunn’s multiple comparisons test. * p<0.05, *** p<0.001 (D) Schematic of IRAK4 exon 6 splicing reporter. IRAK4 exon 6 and 50 bp of flanking introns were cloned into a splicing reporter (pFlare5A)55 and stably expressed into HEK293 cells. SF3B1 and SF3B1-K700E were transfected and IRAK4 exon 6 splicing was measured by RT-PCR using primers within pFlare5A. WT: wild type.
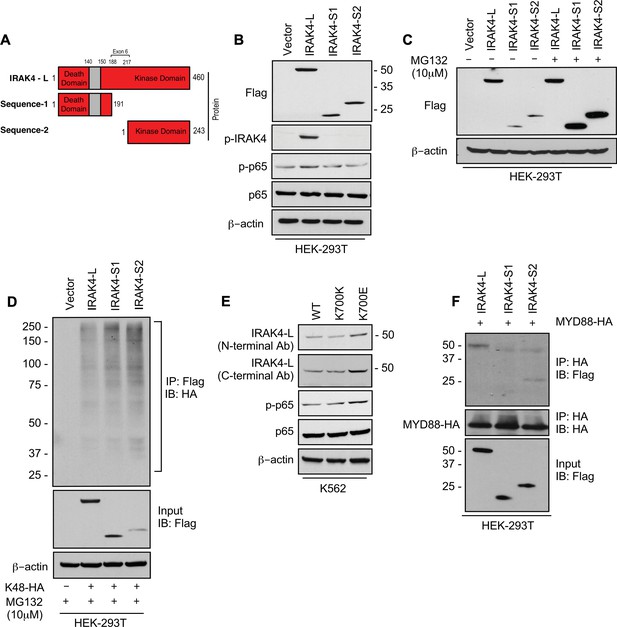
SF3B1 mutation leads to longer, stable, and functionally active isoform of IRAK4.
(A) Schematic showing that IRAK4 protein consists of a kinase domain and death domain that associates with MYD88. The segment of exon 6 that is not included in wild type (WT) controls encodes amino acids 188–217 and can lead to two smaller isoforms by generation of premature stop codon or use of alternative translational start site. (B) HEK-293T cells were transfected with Flag tagged plasmids of IRAK4-long isoform containing full length exon 6, IRAK4-S1, and IRAK4-S2. p-IRAK4, p-p-65, and p65 were determined by immunoblotting. (C) Flag tagged plasmids of IRAK4-long isoform containing full length exon 6, IRAK4-S1, and IRAK4-S2 were transfected in HEK293T cells and treated with proteasome inhibitor MG132 (10 uM) overnight followed by immunoblotting. IRAK4-S1 and IRAK4-S2 constructs that led to smaller IRAK4 protein bands had lower protein expression and were accumulated upon proteasomal inhibition. (D) Ubiquitination of ectopic IRAK4 was determined in HEK-293T cells transfected with indicated plasmids in presence of MG132 (10 µM) by immunoprecipitating with HA-specific antibody and immunoblotting with Flag. (E) Immunoblotting analysis for indicated proteins in isogenic K562 cells with SF3B1 WT and K700E mutation. (F) HEK-293T cells were transfected with Flag tagged IRAK4 isoforms and HA tagged MYD88. MYD88 was immunoprecipitated with HA-specific antibody and its association with IRAK4 was probed by immunoblotting with specific antibodies.
-
Figure 2—source data 1
Expression levels of IRAKa and its signaling partners in HEK293T cells after transfection with indicated plasmids.
- https://cdn.elifesciences.org/articles/78136/elife-78136-fig2-data1-v1.zip
-
Figure 2—source data 2
Expression levels of IRAK4 isforms transfected in HEK293T in presence of proteosome inhibitor.
- https://cdn.elifesciences.org/articles/78136/elife-78136-fig2-data2-v1.zip
-
Figure 2—source data 3
Ubiquitination of ectopic IRAK4 in HEK293T in presence of MG132.
- https://cdn.elifesciences.org/articles/78136/elife-78136-fig2-data3-v1.zip
-
Figure 2—source data 4
Expression levels of proteins in inflmmation pathway in K562 Ctl and K700E cells.
- https://cdn.elifesciences.org/articles/78136/elife-78136-fig2-data4-v1.zip
-
Figure 2—source data 5
Association of IRAK4 isoforms with MyD88.
- https://cdn.elifesciences.org/articles/78136/elife-78136-fig2-data5-v1.zip

Amino acid sequences of IRAK4-long and the shorter IRAK4-Sequence 1 and IRAK4-Sequence 2 isoforms.
The amino acids shown in red color (exon 6) are retained in IRAK4-long isoform.
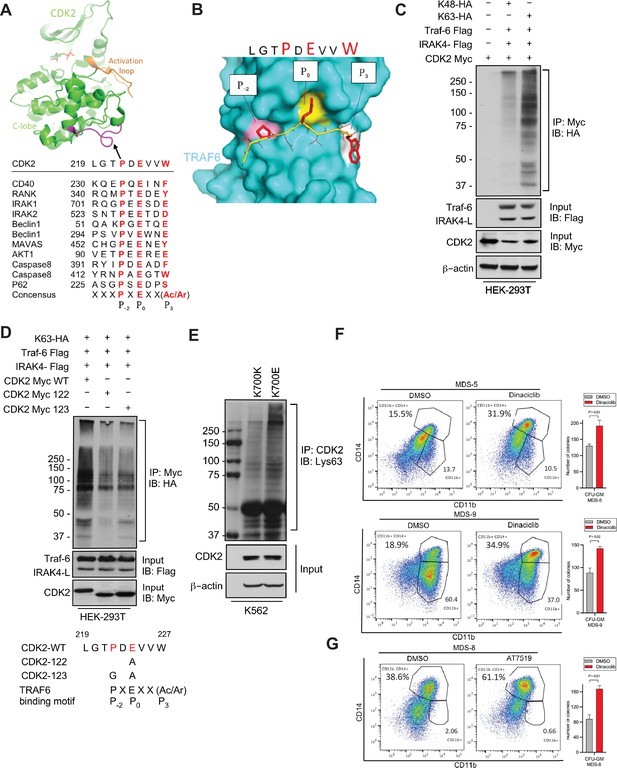
IRAK4/TRAF6 signaling regulates CDK2 ubiquitination in SF3B1 mutant cells.
(A) CDK2 structure (PDB:1GY3) bound to ATP is shown in cartoon representation. The putative TRAF6 binding peptide (magenta) is located at a loop between αG and αH in the C-lobe of the kinase. Sequence alignment of CDK2 peptide with common binding sequences with TRAF6. The consensus pattern with conserved P-2, P0, and P3 positions is shown at the bottom. (B) MM-GBSA docking of CDK2 peptide to substrate binding domain of TRAF6 (PDB:1LB5). The top 1 populated cluster is shown. TRAF6 and CDK2 peptide are shown in ribbon and surface representation, respectively. Positions P-2, P0, and P3 correspond to three distinct sub-pockets in the binding site, formed largely by residues Y473, M450, G472 (pink), A458, G470, K469 (yellow), and H376, V374, R392 (white). Capital lettering of conserved residues P, E, and W within the shown peptides depicts successful docking in corresponding sub-pockets. (C) Lys63-linked proteins were immunoprecipitated in K562 cells with wild type (WT) and K700E SF3B1 mutation and CDK2 ubiquitination was determined by CDK2 specific antibody. (D) Ubiquitination of CDK2 was determined in HEK-293T cells transfected with indicated plasmids by immunoprecipitating with HA specific antibody and immunoblotting with either Lys48 or Lys63 antibody. (E) Lys63-linked proteins were immunoprecipitated in HEK-293T cells transfected with indicated plasmids and CDK2 ubiquitination was determined by Myc-tag specific antibody. Sequence alignment of optimum amino-acids sequence predicted for TRAF6 with WT and mutant CDK2 (CDK2-122 and CDK2-123) (F–G) Myelodysplastic syndromes (MDS) patient derived samples (bone marrow/peripheral blood) were treated with DMSO or Dinaciclib (10 nM) or AT7519 (10 nM) for 14 days on methylcellulose clonogenic assays. The samples were evaluated for colony formation CFU-GM (colony forming unit - granulocyte macrophage) and for myeloid differentiation on colonies and were subjected to flow cytometry analysis.
-
Figure 3—source data 1
Ubiquitination of CDK2 in HEK293T cells.
- https://cdn.elifesciences.org/articles/78136/elife-78136-fig3-data1-v1.zip
-
Figure 3—source data 2
Ubiquitination of CDK2 mutants in HEK293T cells.
- https://cdn.elifesciences.org/articles/78136/elife-78136-fig3-data2-v1.zip
-
Figure 3—source data 3
Ubiquitination of CDK2 in K562 CTL and K700E cells.
- https://cdn.elifesciences.org/articles/78136/elife-78136-fig3-data3-v1.zip
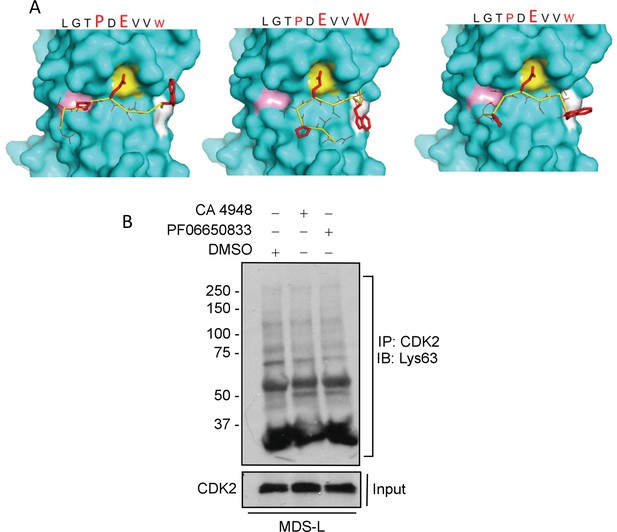
Modeling interaction between TRAF6 and CDK2 and CDK2 ubiquitination after treatment with IRAK4 inhibitors.
(A) MMGBSA docking (see Materials and methods) of CDK2 peptide to substrate binding domain of TRAF6 (PDB:1LB5). Various potential clusters are shown. TRAF6 and CDK2 peptide are shown in ribbon and surface representation, respectively. High-lettering of conserved residues P, E, W within the shown peptides depicts successful docking in corresponding sub-pockets. (B) Lys63 ubiquitination of CDK2 was determined in myelodysplastic syndromes (MDS)-L cells treated with IRAK4 inhibitors. Immunoprecipitation with CDK2 and immunoblotting with anti-Lys63 ubiquitin antibody shows decreased overall smear after IRAK4 inhibition.
-
Figure 3—figure supplement 1—source data 1
Ubiquitination of CDK2 in MDS-L cells with IRAK4 inhibitors CA-4948 (Emavusertib) and PF06650833.
- https://cdn.elifesciences.org/articles/78136/elife-78136-fig3-figsupp1-data1-v1.zip
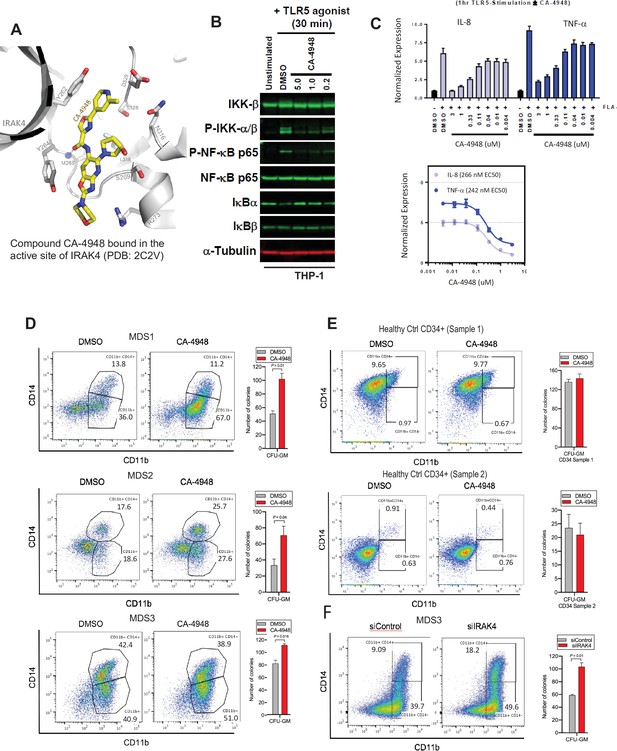
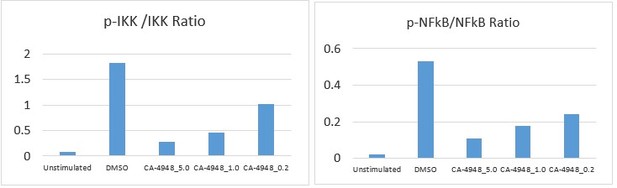
IRAK4 inhibition with CA-4948 promotes differentiation in myelodysplastic syndromes (MDS)/acute myeloid leukemia (AML).
(A) Crystal structure of CA-4948 bound in the active site of IRAK4 (PDB:7C2V). Interacting residues of IRAK4 are shown in sticks. (B) Immunoblotting analysis for indicated proteins in THP-1 cells that were stimulated by toll like receptors (TLR)-5 and treated either with DMSO or CA-4948. (C) mRNA levels of IL8 and TNF-a in OCI-AML2 cells stimulated by TLR-5 ligand and were either treated with DMSO or CA-4948. (D) MDS patient derived samples with SF3B1 mutation were treated with IRAK4 inhibitor (CA4948) or control in methylcellulose clonogenic assays and the analyzed for myeloid colony formation. Colonies were picked and analyzed by FACS for myeloid differentiation markers CD11b and CD14. (E) Healthy CD34+ stem and progenitor cells were grown in clonogenic assays with and without IRAK4 inhibitor (CA4948). Myeloid colonies were counted at day 14 and colonies were analyzed by FACS. (F) MDS patient sample with SF3B1 mutation was treated with siRNAs against IRAK4 and control and grown in clonogenic assays. The sample was evaluated for myeloid colony formation and for differentiation by analyzing their CD11b and CD14 expression with flow cytometry.
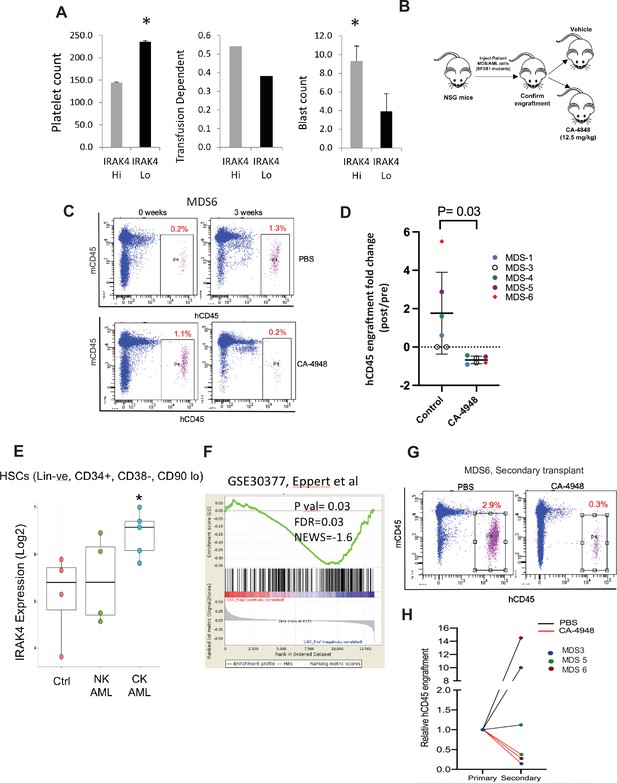
IRAK4 overexpression is associated with adverse clinical features in human myelodysplastic syndromes (MDS), and its inhibition leads to reduction in MDS clones in vivo.
(A) IRAK4 expression in 183 samples from MDS patient bone marrow CD34+ cells was correlated with platelet counts, RBC transfusion dependency, and blast counts. Cohort with high IRAK4 expression (>median) were associated with lower platelet counts, higher transfusion dependence, and higher blast counts (t-test, p<0.05). (B) NSG mice were xenografted with MDS cells with SF3B1 mutation. After engraftment, mice were treated with either vehicle or IRAK4 inhibitor (CA-4849, 12.5 mg/kg) and bone marrow aspirates were used to evaluate human cells engraftment by flow cytometry. (C–D) Summary of human cell engraftment for xenografted mice treated with either IRAK4 inhibitor (CA-4948) or vehicle. Representative MDS sample xenograft with SF3B1 K700E mutation is shown. (E) IRAK4 expression in sorted HSCs (Lineage –ve, CD34+, CD38−) cells from MDS/acute myeloid leukemia (AML) cases (N=9) and controls (N=5). Significantly increased IRAK4 expression is seen in samples with complex karyotype (CK) when compared to HC (t-test, p<0.05). (F) Gene expression signature associated with high IRAK4 expression (>median, from set of 183 MDS CD34+ cells) was compared to know leukemic stem cell signature and showed significant similarity by GSEA analysis. (G–H) MDS bone marrow cells were isolated and purified from mice either treated with vehicle or CA-4948 in and then xenografted in secondary recipient NSG mice. Human cell engraftment was evaluated after 4 weeks by flow cytometry on bone marrow aspirates.
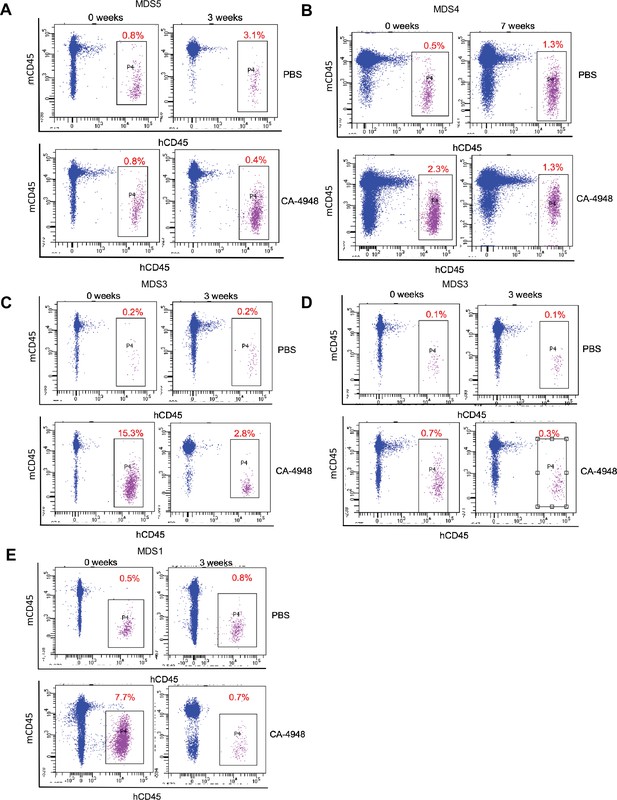
IRAK4 inhibition leads to reduction in myelodysplastic syndromes (MDS) clones in vivo.
NSG mice were xenografted with MDS cells with SF3B1 mutation. After engraftment, mice were treated with either vehicle or IRAK4 inhibitor (CA-4849, 12.5 mg/kg) and bone marrow aspirates were used to evaluate human cells engraftment by flow cytometry.
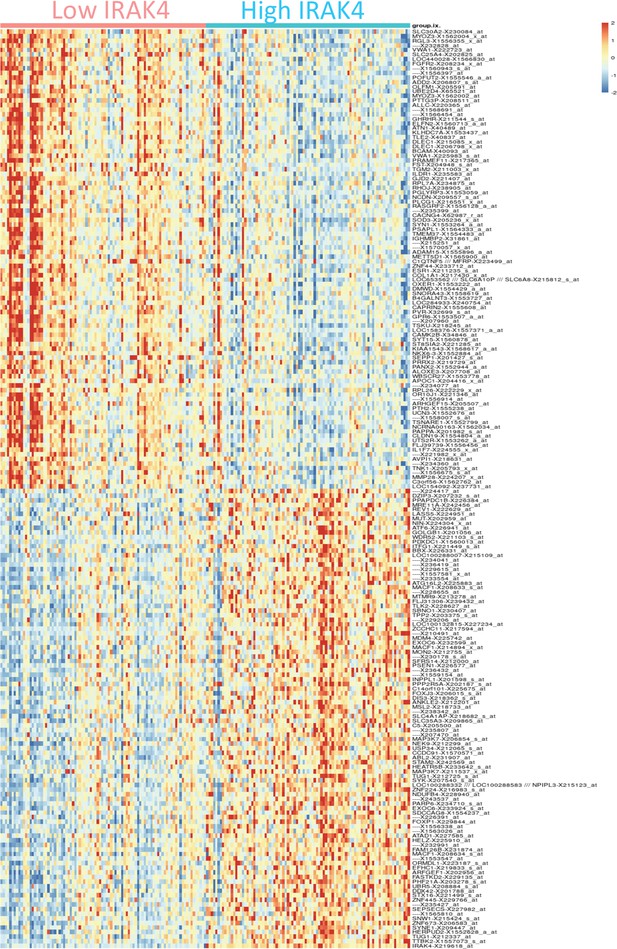
Gene expression signature associated with high IRAK4 expression.
Top 100 differentially expressed genes between cohorts with high and low IRAK4 (based on median expression) are shown.
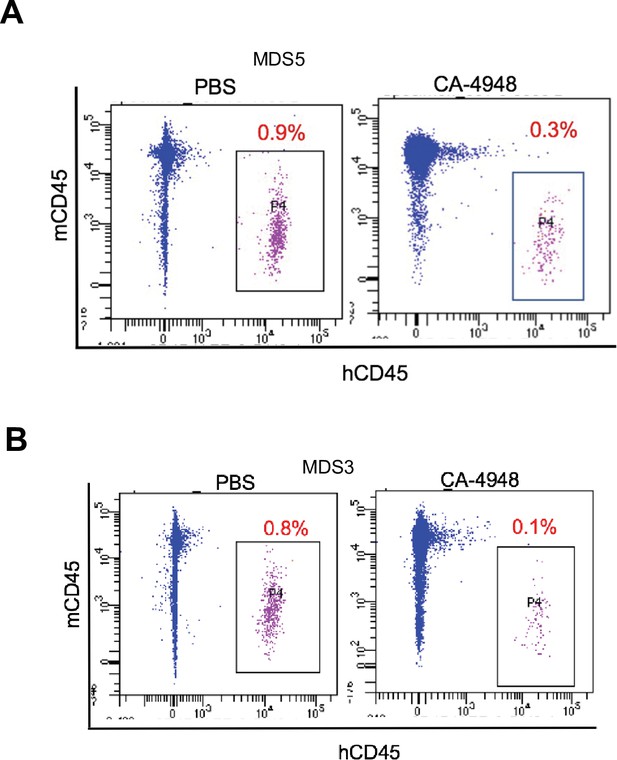
IRAK4 inhibition leads to reduction in myelodysplastic syndromes (MDS) clones in secondary transplant recipient mice.
MDS bone marrow cells were isolated and purified from NSG mice either treated with vehicle or CA-4948 in and then xenografted in secondary recipient NSG mice. Human cell engraftment was evaluated after 4 weeks by flow cytometry on bone marrow aspirates.
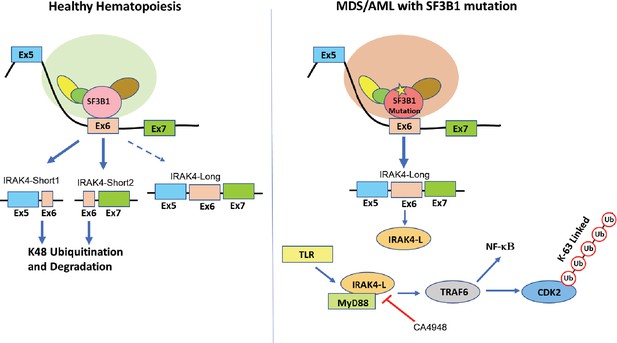
Proposed schematic of myelodysplastic syndromes (MDS) pathogenesis due to mis-splicing of IRAK4 because of SF3B1 mutations.
Additional files
-
Supplementary file 1
Patient samples.
- https://cdn.elifesciences.org/articles/78136/elife-78136-supp1-v1.pptx
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/78136/elife-78136-transrepform1-v1.docx