TGF-β signaling and Creb5 cooperatively regulate Fgf18 to control pharyngeal muscle development
Figures
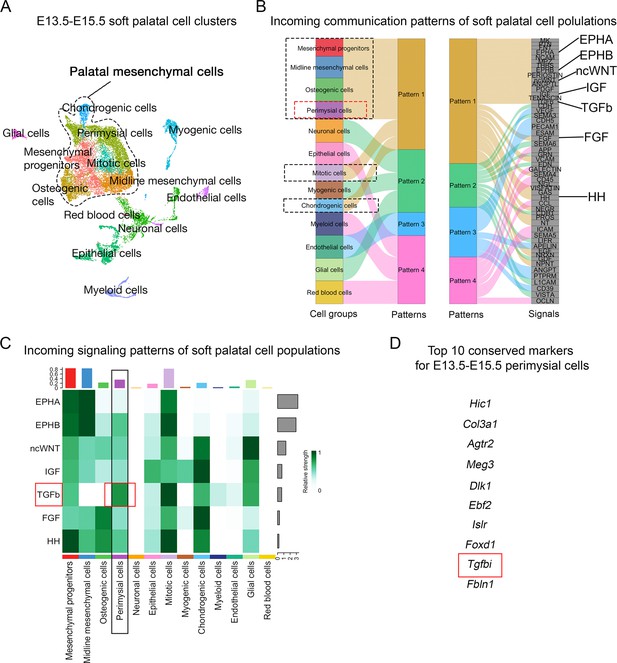
CellChat analysis of E13.5-E15.5 soft palatal scRNAseq data predicts TGF-β signaling as a key pathway activated in the perimysial cells.
(A) UMAP plot of soft palatal cell populations identified by Seurat integration analysis. (B) Alluvial plot of the incoming signaling patterns for soft palatal cell populations. The black dotted boxed area indicates the palatal mesenchymal cell populations grouped into the same incoming patterns. The red dotted box highlights the perimysial cells. (C) Heatmap of the contribution of the incoming pathways of interest to soft palatal cell populations. The black box highlights signaling activity in the perimysial cells and the red box highlights TGF-β signaling activity in the perimysial cells. (D) Top conserved markers for perimysial cells identified through Seurat integration analysis.
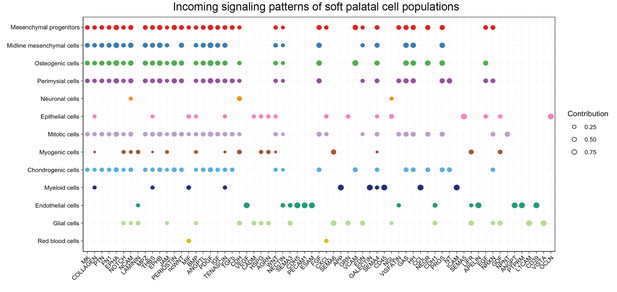
CellChat analysis of scRNAseq data identified incoming signaling pathways for all cell types in the soft palate.
Bubble plot showing significant incoming signaling patterns for all the cell types of developing soft palate from E13.5-E15.5. Different colors in the plot represent different cell groups. Sizes in the bubble plot are proportional to the communication probability.
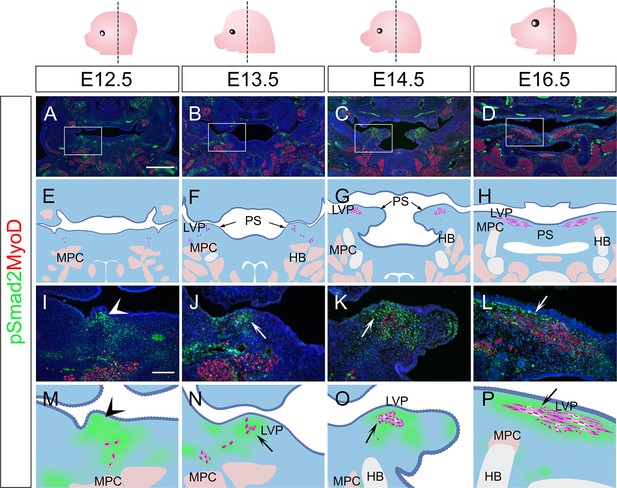
TGF-β signaling is activated in perimysial cells surrounding the developing LVP myogenic cells.
(A–P) Immunofluorescence (A–D and I–L) and schematic drawings (E–H and M–P) of myogenic marker MyoD (red) and TGF-β signaling readout pSmad2 (green) expression in coronal sections of LVP region at E12.5, E13.5, E14.5, and E16.5. Boxed areas in A, B, C, and D are shown magnified in I, J, K, and L, respectively. Schematic drawings in E-H and M-P correspond to A-D and I-L, respectively. Arrows in F and G indicate the palatal shelves. White and black arrows in J-L and N-P point to the LVP myogenic sites in the palatal shelves. Arrowheads indicate the absence of MyoD+ signal in mouse pharyngeal wall at E12.5 in I and M, and arrows indicate the presence from E13.5 onwards in J-L and N-P. The magenta and salmon colors represent the LVP myogenic cells and the other pharyngeal muscles, respectively. HB, hyoid bone; LVP, levator veli palatini; MPC, middle pharyngeal constrictor; PS, palatal shelves. Top panel schematics depict the orientation and level of the sections. N=3 for all experiments. Scale bars in A and I indicate 500 μm in A-D and 100 μm in I-L, respectively.
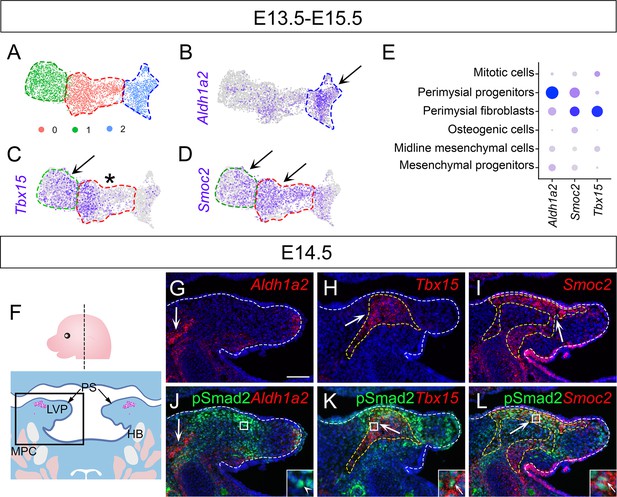
Tbx15+ and Smoc2+ perimysial cells are present in the region where TGF-β signaling is active.
(A) UMAP plot for three perimysial cell clusters subsetted and reclustered from E13.5-E15.5 soft palatal scRNAseq analysis. (B–D) Expression of putative marker genes (Aldh1a2, Tbx15, and Smoc2) that are expressed differentially in the 3 perimysial clusters from E13.5-E15.5 soft palatal scRNAseq analysis. Black arrows indicate abundance of the expression and asterisks indicate the deficiency of expression of the marker genes in the clusters. (E) DotPlot of feature genes for different perimysial subpopulation cell types. The intensity of the blue color of signature genes corresponds to the level of expression in the specific cell type on the x-axis. (F) Schematic drawing of the plane of section (upper panel) and zoomed in area (lower panel) for G-L. The magenta and salmon colors represent the LVP myogenic cells and the other pharyngeal muscles, respectively. HB, hyoid bone; LVP, levator veli palatini; MPC, middle pharyngeal constrictor; PS, palatal shelves. (G–L) Co-staining of pSmad2 immunostaining (green) and Aldh1a2 (red), Tbx15 (red), or Smoc2 (red) RNAScope in situ hybridizations in the coronal sections of LVP region at E14.5. Boxed areas in J-L are enlarged as insets. The white and yellow dotted lines outline the palatal shelves and Tbx15+ or Smoc2+ area in G-L, respectively. White arrows in G-L point to the region with Aldh1a2, Tbx15 or Smoc2 expression. White arrowhead in the inset in J indicates an example of the absence of Aldh1a2 expression in a pSmad2+ cell in the palatal shelves, while white arrows in insets in K and L indicate the presence of Tbx15 or Smoc2 expression in pSmad2+ cells. N=3. Scale bar in G indicates 100 μm for G-L and 20 μm for the insets in G-L.
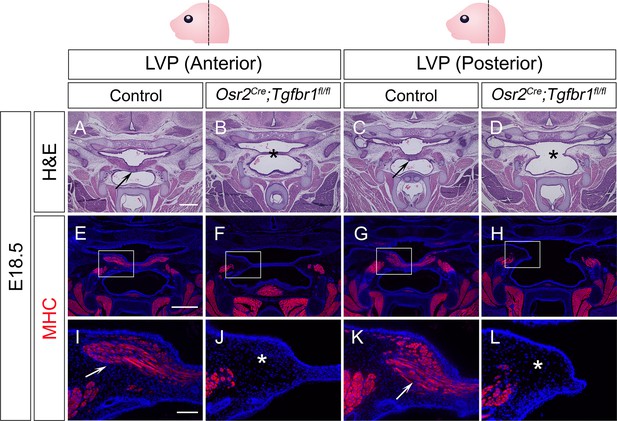
Osr2Cre;Tgfbr1fl/fl mice exhibit palatal shelf and LVP defects at E18.5.
(A–D) H&E staining of coronal sections at LVP region from control and Osr2Cre;Tgfbr1fl/fl mice at E18.5. Arrows indicate the normal palatal shelf in A and C; asterisks indicate the deformed and cleft palatal shelf in B and D, respectively. N=4. (E–L) Immunofluorescence of MHC (red) in coronal sections at LVP region from control and Osr2Cre;Tgfbr1fl/fl mice at E18.5. Boxed areas in E, F, G, and H are enlarged in I, J, K, and L, respectively. Arrows indicate the presence of muscle fibers in I and K; asterisks indicate their absence in J and L. N=3. Top panel schematics depict the orientation and level of the sections. Tgbfr1fl/fl or Tgbfr1fl/+ littermates were used as controls for Osr2Cre;Tgfbr1fl/fl mice. Scale bars in A and E indicate 500 µm in A-D and E-H, respectively; the scale bar in I indicates 100 µm in I-L.
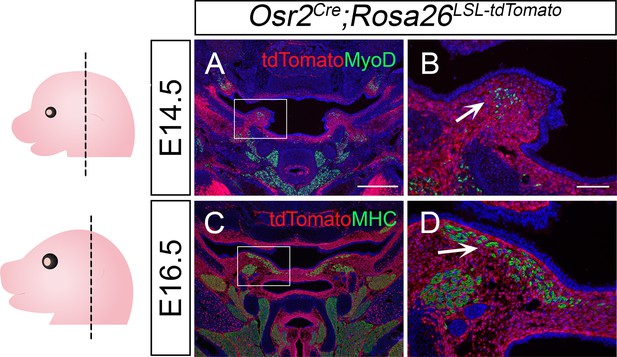
Osr2Cre targets palatal mesenchymal cells surrounding soft palatal myogenic cells.
(A–D) Immunofluorescence of myogenic marker MyoD (green) or MHC (green) and tdTomato (red) in the coronal sections of LVP region of E14.5 and E16.5 in Osr2Cre;Rosa26LSL-tdTomato mice. Boxes in A and C are shown at higher magnification in B and D, respectively. Arrows in B and D indicate that the green MyoD or MHC signal does not overlap with the red tdTomato signal at E14.5 or E16.5. Left panel schematics depict the orientation and level of the sections. N=3. The scale bar in A indicates 500 µm in A and C; the scale bar in B indicates 100 µm in B and D.
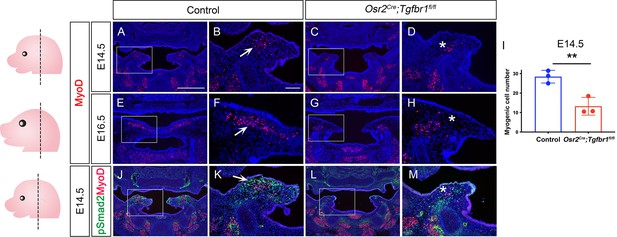
Osr2Cre;Tgfbr1fl/fl mice exhibit palatal shelf and myogenic defects in the LVP region from E14.5 onwards.
(A–H) Immunofluorescence of myogenic marker MyoD (red) in coronal sections of palatal shelves of control and Osr2Cre;Tgfbr1fl/fl mice at E14.5 (A–D) and E16.5 (E–H) LVP region. Boxes in A, C, E, and G are shown at higher magnification in the B, D, F, and H, respectively. Arrows in B and F indicate the presence of MyoD+ cells in the control, and asterisks in D and H indicate reduced MyoD+ cells in the Osr2Cre;Tgfbr1fl/fl mice. (I) Quantification of myogenic cell number in control and Osr2Cre;Tgfbr1fl/fl mice at E14.5. **, p≤0.01. Statistical significance was assessed by unpaired t-test with two-tailed calculations. Data is presented as mean ± SEM. (J–M) Immunofluorescence of MyoD (red) and pSmad2 (green) in coronal sections of the LVP region of E14.5 control and Osr2Cre;Tgfbr1fl/fl mice. Boxed areas in J and L are enlarged in K and M, respectively. The arrows indicate the presence of signal and asterisks correspond to loss of signal. N=3 for all experiments. Tgbfr1fl/fl or Tgbfr1fl/+ littermates were used as controls for Osr2Cre;Tgfbr1fl/fl mice. Left panel schematics depict the orientation and level of the sections. The scale bar in A indicates 500 µm in A, C, E, G, J, and L; the scale bar in B indicates 100 µm for the rest of the figures.
-
Figure 3—figure supplement 2—source data 1
Source data for Figure 3—figure supplement 2I.
- https://cdn.elifesciences.org/articles/80405/elife-80405-fig3-figsupp2-data1-v1.xlsx
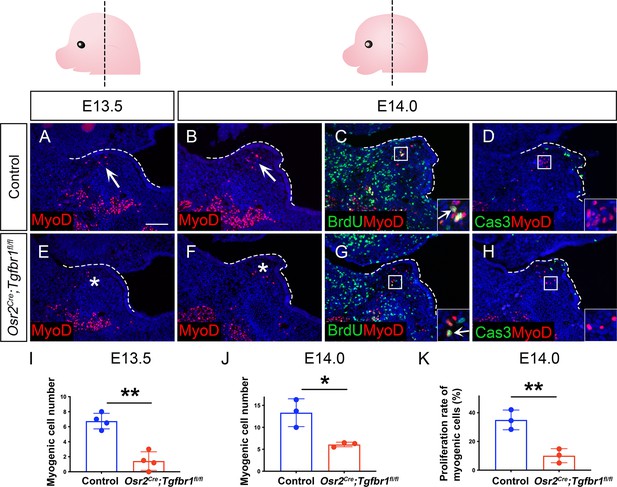
Myogenic defects occur in Osr2Cre;Tgfbr1fl/fl mice prior to E14.5.
(A–H) Immunofluorescence of MyoD (red) (A–B, E–F) or co-staining with BrdU (green) (C, G) or Caspase 3 (green) (D, H) in coronal sections of palatal shelves in the LVP region of control and Osr2Cre;Tgfbr1fl/fl mice at E13.5 and E14.0. Boxed areas in C, D, G, and H are enlarged as insets. Arrows indicate a positive signal and asterisks indicate reduction of signal in the palatal shelf region. The palatal shelves are outlined by white dotted lines. (I–K) Quantification of myogenic cell numbers at E13.5 (I) and E14.0 (J) and proliferation rate of myogenic cells at E14.0 (K). **, p≤0.01; *, p≤0.05. Statistical significance was assessed by unpaired t-test with two-tailed calculations. Data is presented as mean ± SEM. Top panel schematics depict the orientation and level of the sections. N=3 for all experiments except I (N=4). Tgbfr1fl/fl or Tgbfr1fl/+ littermates were used as controls for Osr2Cre;Tgfbr1fl/fl mice. Scale bar in A indicates 100 μm for A-H.
-
Figure 3—figure supplement 3—source data 1
Source data for Figure 3—figure supplement 3I.
- https://cdn.elifesciences.org/articles/80405/elife-80405-fig3-figsupp3-data1-v1.xlsx
-
Figure 3—figure supplement 3—source data 2
Source data for Figure 3—figure supplement 3J.
- https://cdn.elifesciences.org/articles/80405/elife-80405-fig3-figsupp3-data2-v1.xlsx
-
Figure 3—figure supplement 3—source data 3
Source data for Figure 3—figure supplement 3K.
- https://cdn.elifesciences.org/articles/80405/elife-80405-fig3-figsupp3-data3-v1.xlsx
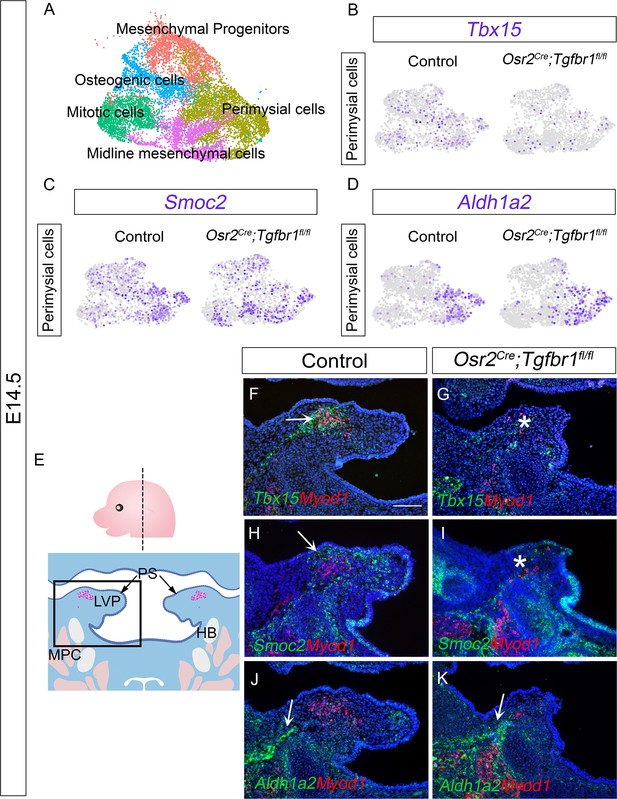
The Tbx15+ and Smoc2+ subpopulations are more affected than the Aldh1a2+ subpopulation in Osr2Cre;Tgfbr1fl/fl palatal mesenchyme at E14.5.
(A) UMAP plot of palatal mesenchymal cell clusters from integrated scRNAseq analysis of control and Osr2Cre;Tgfbr1fl/fl soft palates at E14.5. (B–D) Feature plot view of expression patterns of perimysial fibroblast markers Tbx15 (B) and Smoc2 (C) and perimysial progenitor marker gene Aldh1a2 (D) in perimysial cells from integrated control and Osr2Cre;Tgfbr1fl/fl palatal mesenchymal cell clusters at E14.5. (E) Schematic drawing of orientation and level of the sections (top panel) and coronal sections of LVP region at E14.5 (bottom panel). Boxed area in E indicates the region of F-K. Magenta and salmon colors represent the LVP myogenic cells and the other pharyngeal muscles, respectively. HB, hyoid bone; LVP, levator veli palatini; MPC, middle pharyngeal constrictor; PS, palatal shelves. (F–K) RNAScope in situ hybridization for Tbx15 (green), Smoc2 (green), or Aldh1a2 (green) colocalization with myogenic markers Myod1 (red) in coronal sections of LVP region of control and Osr2Cre;Tgfbr1fl/fl mice at E14.5. Arrows in F, H, J, and K indicate Tbx15, Smoc2, or Aldh1a2 expression. Asterisks in G and I show reduced Tbx15 or Smoc2 expression. N=3. Tgbfr1fl/fl or Tgbfr1fl/+ littermates were used as controls for Osr2Cre;Tgfbr1fl/fl mice. The scale bar in F indicates 100 μm in F-K.
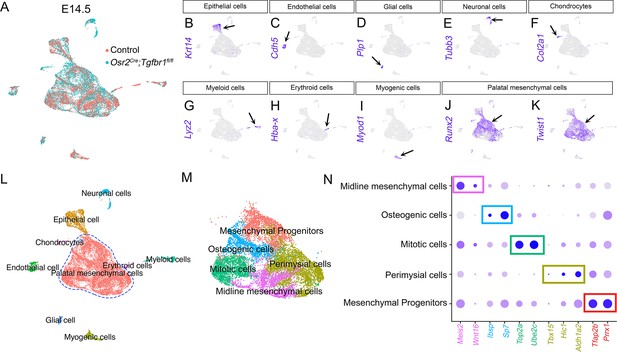
scRNAseq analysis identified distinct populations of palatal mesenchymal cells in E14.5 Osr2Cre;Tgfbr1fl/fl and control (Tgbfr1fl/fl or Tgbfr1fl/+) soft palates.
(A) Integration analysis of scRNAseq from the soft palates of control and Osr2Cre;Tgfbr1fl/fl mice at E14.5. (B–K) Expression patterns of marker genes distinguishing non-mesenchymal cells from palatal mesenchymal cells (Runx2+/Twist1+). (L) Identification and characterization of different cellular populations in the E14.5 Osr2Cre;Tgfbr1fl/fl and control soft palates. (M) UMAP plot of integrated palatal mesenchymal cells of E14.5 Osr2Cre;Tgfbr1fl/fl and control soft palates based on clustering. (N) Marker genes from the integration of scRNAseq analysis of individual cellular clusters between Osr2Cre;Tgfbr1fl/fl and control mice at E14.5. Note highlighted markers for perimysial cells: Tbx15, Hic1, and Aldh1a2.
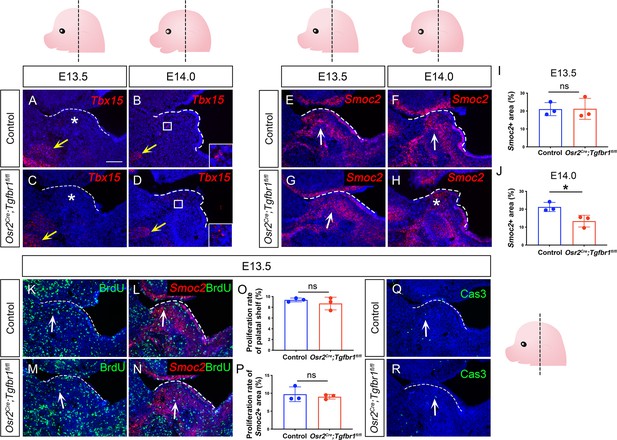
Expression of perimysial markers Tbx15 and Smoc2 in E13.5-E14.0 Osr2Cre;Tgfbr1fl/fl and control soft palates.
(A–H) RNAScope in situ hybridization for Tbx15 (red) (A–D) and Smoc2 (red) (E–H) in coronal sections of the LVP region of E13.5 and E14.0 control and Osr2Cre;Tgfbr1fl/fl mice. Boxed areas in B and D are shown enlarged in insets. White arrows point to a positive signal while asterisks indicate absence or reduction of signal in the palatal shelf region. Yellow arrows in A-D point to positive signals in the middle pharyngeal constrictor regions. (I–J) Quantification of the percentage of Smoc2 expressing area out of the entire palatal shelf region for control and Osr2Cre;Tgfbr1fl/fl mice at E13.5 (I) and E14.0 (J). *, p≤0.05; ns, not significant. Statistical significance was assessed by unpaired t-test with two-tailed calculations. Data is presented as mean ± SEM. (K–N) Immunofluorescence of BrdU (green) (K and M) or co-staining with Smoc2 RNAScope in situ hybridization (red) (L and N) in coronal sections of the LVP region in control and Osr2Cre;Tgfbr1fl/fl mice at E13.5. White arrows point to an example of a positive BrdU+ signal in the palatal shelf region (K and M) or in the Smoc2+ cells (L and N). (O–P) Quantification of the percentage of BrdU+ proliferative cells for the entire the palatal shelf (O) or Smoc2+ cells in the palatal shelf (P). ns, not significant. Statistical significance was assessed by unpaired t-test with two-tailed calculations. Data is presented as mean ± SEM. (Q–R) Immunofluorescence of Caspase 3 (green) in coronal sections of the LVP region in control and Osr2Cre;Tgfbr1fl/fl mice at E13.5. The schematics in the top panel and bottom right panel depict the orientation and level of the sections. The white dotted line outlines the palatal shelf region. Tgfbr1fl/fl or Tgfbr1fl/+ littermates were used as control for Osr2Cre;Tgfbr1fl/fl mice. N=3 for all experiments. Scale bar in A indicates 100 μm for A-H, K-N, Q, and R.
-
Figure 4—figure supplement 2—source data 1
Source data for Figure 4—figure supplement 2I.
- https://cdn.elifesciences.org/articles/80405/elife-80405-fig4-figsupp2-data1-v1.xlsx
-
Figure 4—figure supplement 2—source data 2
Source data for Figure 4—figure supplement 2J.
- https://cdn.elifesciences.org/articles/80405/elife-80405-fig4-figsupp2-data2-v1.xlsx
-
Figure 4—figure supplement 2—source data 3
Source data for Figure 4—figure supplement 2O.
- https://cdn.elifesciences.org/articles/80405/elife-80405-fig4-figsupp2-data3-v1.xlsx
-
Figure 4—figure supplement 2—source data 4
Source data for Figure 4—figure supplement 2P.
- https://cdn.elifesciences.org/articles/80405/elife-80405-fig4-figsupp2-data4-v1.xlsx
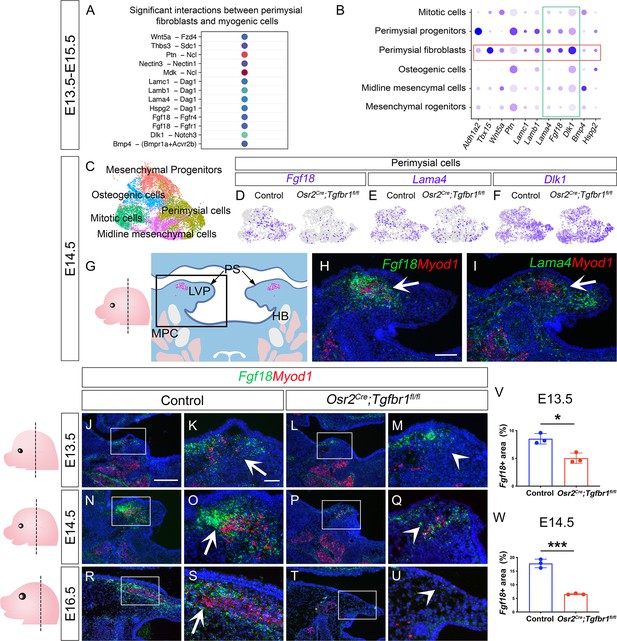
CellChat analysis of soft palatal scRNAseq data identifies putative perimysial-to-myogenic signaling molecules downregulated in Osr2Cre;Tgfbr1fl/fl perimysial fibroblasts.
(A) CellChat analysis of scRNAseq integration data of E13.5-E15.5 soft palatal tissue showing significant interactions between perimysial fibroblasts and myogenic cells. (B) Dot plot of expression patterns of perimysial fibroblast-derived signals in individual cellular clusters of integration scRNAseq from E13.5-E15.5 soft palatal tissue. Note that highlighted signals, Lama4, Fgf18, and Dlk1, are more enriched in the perimysial fibroblasts. (C) UMAP plot of palatal mesenchymal cell clusters from integrated scRNAseq analysis of control and Osr2Cre;Tgfbr1fl/fl soft palates at E14.5. (D–F) Feature plot view of Fgf18 (D), Lama4 (E), or Dlk1 (F) expression in perimysial cells from the integrated control and Osr2Cre;Tgfbr1fl/fl palatal mesenchymal cell clusters at E14.5. (G) Schematic drawing of orientation and level of the sections (left panel) and coronal section of the LVP region (right panel) at E14.5. Boxed area in G indicates the region shown in H-I. The magenta and salmon colors represent the LVP myogenic cells and the other pharyngeal muscles, respectively. HB, hyoid bone; LVP, levator veli palatini; MPC, middle pharyngeal constrictor; PS, palatal shelves. (H–I) RNAScope in situ hybridization for Myod1 (red) and Fgf18 (green) (H) or Lama4 (green) (I) in the coronal section of the LVP region at E14.5. White arrows in H and I point to a positive signal. (J–U) RNAScope in situ hybridization for Myod1 (red) and Fgf18 (green) in the coronal section of the LVP region at E13.5 (J–M), E14.5 (N–Q), and E16.5 (R–U) in control and Osr2Cre;Tgfbr1fl/fl mice. Boxed areas in J, L, N, P, R, and T are enlarged in K, M, O, Q, S, and U, respectively. Arrows in K, O, and S point to positive signal; arrowheads in M, Q, and U indicate lack of signal. Left panel schematics depict the orientation and level of the sections. (V–W) Quantification of the percentage of Fgf18 expressing area out of the entire the palatal shelf region for control and Osr2Cre;Tgfbr1fl/fl mice at E13.5 (V) and E14.5 (W). *, p≤0.05; ***, p≤0.001. Statistical significance was assessed by unpaired t-test with two-tailed calculations. Data is presented as mean ± SEM. N=3 for all experiments. Tgbfr1fl/fl or Tgbfr1fl/+ littermates were used as controls for Osr2Cre;Tgfbr1fl/fl mice. The scale bar in H indicates 100 μm in H and I; the scale bar in J indicates 250 μm in J, L, N, P, R, and T; the scale bar in K indicates 50 μm for the rest of the figures.
-
Figure 5—source data 1
Source data for Figure 5U.
- https://cdn.elifesciences.org/articles/80405/elife-80405-fig5-data1-v1.xlsx
-
Figure 5—source data 2
Source data for Figure 5V.
- https://cdn.elifesciences.org/articles/80405/elife-80405-fig5-data2-v1.xlsx
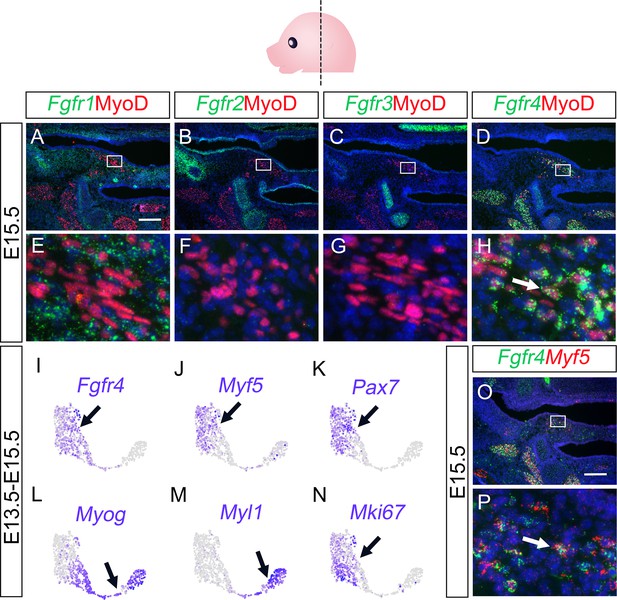
Fgf18 receptor Fgfr4 is predominantly expressed by myogenic cells in the LVP regions.
(A–H) Immunofluorescence of MyoD (red) and RNAScope in situ hybridization for Fgfr1 (green) (A, E), Fgfr2 (B, F), Fgfr3 (C, G), and Fgfr4 (D, H) in coronal sections of LVP region at E15.5. Boxed areas in A-D are enlarged in E-H, respectively. N=3. (I–N) Expression of Fgfr4, myogenic markers, and proliferative marker in the myogenic cell population from E13.5-E15.5 soft palatal scRNAseq analysis. Note the expression of Fgfr4 (I) overlaps mainly with that of the myogenic progenitor markers (Myf5/Pax7) (J and K), and proliferative markers (Mki67) (N), but not with markers of the more differentiated myogenic cells (Myog/Myl1) (L and M). (O–P) RNAScope in situ hybridization for Fgfr4 (green) and Myf5 (red) in coronal sections of the LVP region at E15.5. Boxed area in O is enlarged in P. Arrow in P indicates an example of Fgfr4 expression by Myf5+ cells. N=3. Scale bars in A and O indicate 200 μm for A-D and O, and 20 μm for E-H and P, respectively.
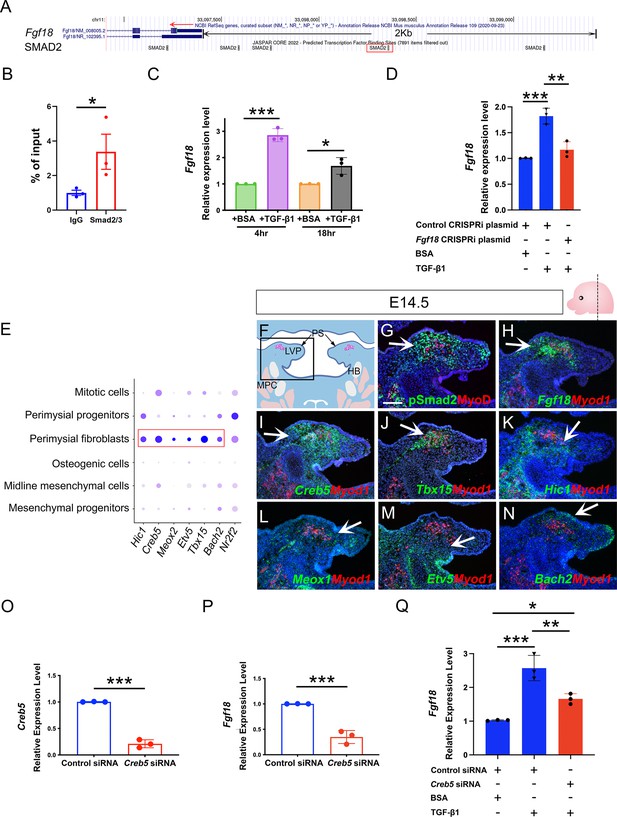
Perimysial fibroblast-specific regulon Creb5, identified by SCENIC analysis, cooperates with TGF-β signaling to regulate Fgf18.
(A) UCSC binding prediction of SMAD2 binding motif to the promoter of Fgf18 gene. The boxed area indicates the binding site with the highest score. (B) Cut and Run analysis shows significantly more enriched Smad2/3 binding to the promoter region of the Fgf18 gene close to the predicted binding site than that of IgG in the soft palatal tissue of the control mice at E14.5. *, p-value ≤0.05. Statistical significance was assessed by unpaired t-test with two-tailed calculations. Data is presented as mean ± SEM. (C) qPCR analysis of Fgf18 expression in E14.5 soft palatal mesenchymal cell culture after treatment with 5 ng/ml TGF-β1 or BSA compared after 4 or 18 hr. Note the increase of Fgf18 at 4 hr is higher than at 18 hr. *, p≤0.05; ***; p≤0.001. Statistical significance was assessed by unpaired t-test with two-tailed calculations. Data is presented as mean ± SEM. (D) qPCR analysis of Fgf18 expression of E14.5 soft palatal mesenchymal cell culture transfected with Fgf18 or Control CRISPRi plasmid followed by treatment with 5 ng/ml TGF-β1 or BSA for 4 hr. ‘+’ or ‘-’ under the plots indicates the presence or absence of the indicated treatment. **, p≤0.01; ***, p≤0.001. Statistical significance was assessed by ANOVA. Data is presented as mean ± SEM. (E) Dotplot of perimysial fibroblast regulon expression pattern in individual cellular clusters of integrated scRNAseq from E13.5-E15.5 soft palatal tissue. Highlighted regulons are more enriched in the perimysial fibroblasts. (F) Schematic drawing of coronal section of the LVP region at E14.5. Boxed area in E indicates the region shown in G-N. The magenta and salmon colors represent the LVP myogenic cells and the other pharyngeal muscles, respectively. HB, hyoid bone; LVP, levator veli palatini; MPC, middle pharyngeal constrictor; PS, palatal shelves. (G) Immunofluorescence of MyoD (red) and pSMAD2 (green) in the coronal section of the LVP region at E14.5. (H–N) RNAScope in situ hybridization for Myod1 (red) and Fgf18 (green) (H), Creb5 (green) (I), Tbx15 (green) (J), Hic1 (green) (K), Meox1 (green) (L), Etv5 (green) (M), or Bach2 (green) (N) in coronal sections of the LVP region at E14.5. Arrows indicate positive signals. (O–P) qPCR analysis of Creb5 (O) and Fgf18 expression (P) after Creb5 siRNA treatment on E14.5 soft palatal mesenchymal cell culture compared with the control siRNA. ***, p≤0.001. Statistical significance was assessed by unpaired t-test with two-tailed calculations. Data is presented as mean ± SEM. (Q) qPCR analysis of Fgf18 expression following Creb5 siRNA treatment combined with 5 ng/ml TGF-β1 or BSA on E14.5 soft palatal mesenchymal cell culture, compared with the control siRNA. ‘+’ or ‘-’ under the plots indicates the presence or absence of the indicated treatment. *, p≤0.05; **, p≤0.01; ***, p≤0.001. Statistical significance was assessed by ANOVA. Data is presented as mean ± SEM. N=3 for all experiments. Tgbfr1fl/fl or Tgbfr1fl/+ littermates were used as controls for Osr2Cre;Tgfbr1fl/fl mice. Scale bar in G indicates 100 μm in G-N.
-
Figure 6—source data 1
Source data for Figure 6B.
- https://cdn.elifesciences.org/articles/80405/elife-80405-fig6-data1-v1.xlsx
-
Figure 6—source data 2
Source data for Figure 6C.
- https://cdn.elifesciences.org/articles/80405/elife-80405-fig6-data2-v1.xlsx
-
Figure 6—source data 3
Source data for Figure 6D.
- https://cdn.elifesciences.org/articles/80405/elife-80405-fig6-data3-v1.xlsx
-
Figure 6—source data 4
Source data for Figure 6O.
- https://cdn.elifesciences.org/articles/80405/elife-80405-fig6-data4-v1.xlsx
-
Figure 6—source data 5
Source data for Figure 6P.
- https://cdn.elifesciences.org/articles/80405/elife-80405-fig6-data5-v1.xlsx
-
Figure 6—source data 6
Source data for Figure 6Q.
- https://cdn.elifesciences.org/articles/80405/elife-80405-fig6-data6-v1.xlsx
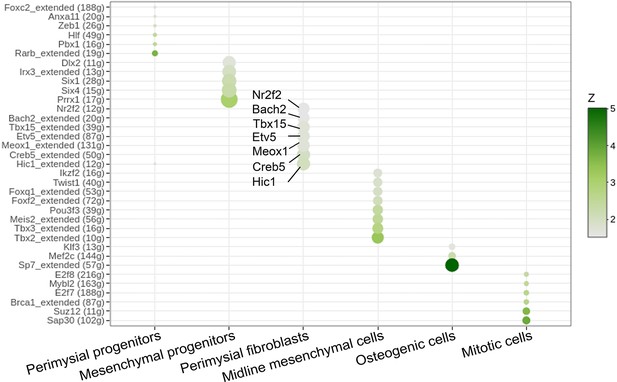
SCENIC analysis identified individual cell type-specific regulons from E13.5-E15.5 soft palatal scRNAseq data.
Dot plot indicates predicated regulons for perimysial progenitors, mesenchymal progenitors, perimysial fibroblasts, midline mesenchymal cells, osteogenic cells, and mitotic cells. Note the regulons identified for perimysial fibroblasts are Hic1, Creb5, Meox1, Etv5, Tbx15, Bach2, and Nr2f2.
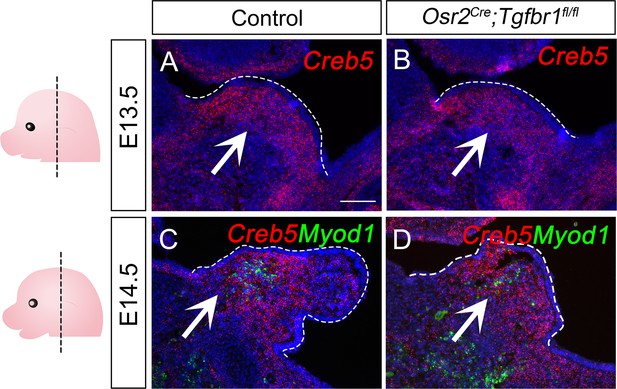
Creb5 expression in E13.5-E14.5 Osr2Cre;Tgfbr1fl/fl and control soft palates.
(A–B) RNAScope in situ hybridization for Creb5 (red) expression in coronal sections of LVP region of E13.5 control and Osr2Cre;Tgfbr1fl/fl mice. (C–D) RNAScope in situ hybridization for Creb5 (red) and Myod1 (green) expression in coronal sections of LVP region of E14.5 control and Osr2Cre;Tgfbr1fl/fl mice. White arrows point to positive signals. Dotted line outlines the palatal shelf regions. N=3 for all experiments. Tgfbr1fl/fl or Tgfbr1fl/+ littermates were used as controls for Osr2Cre;Tgfbr1fl/fl mice. Scale bar in A indicates 100 μm for A-D.
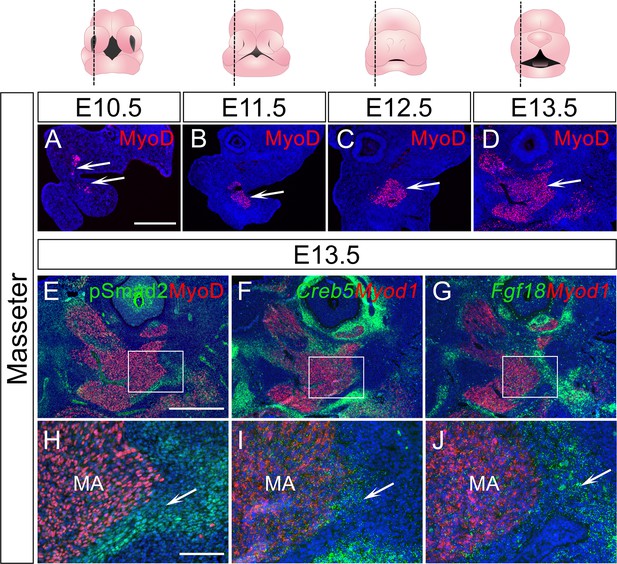
TGF-β signaling, Creb5, and Fgf18 are expressed in a similar region of the perimysial cells of the masseter at E13.5.
(A–D) Immunofluorescence of MyoD (red) in sagittal sections of control mouse head in the masseter region at E10.5 (A), E11.5 (B), E12.5 (C), and E13.5 (D). White arrows in A-D point to forming masseter. (E and H) Immunofluorescence of MyoD (red) and pSMAD2 (green) in sagittal sections of control mouse head in the masseter muscle region at E13.5. Boxed area in E is enlarged in H. The white arrow in H points to a positive signal. (F–G and I–J) RNAScope in situ hybridization for Myod1 (red) and Creb5 (green) (F and I) or Fgf18 (green) (G and J) in sagittal sections of control mouse heads at in the masseter region E13.5. Boxed areas in F and G are enlarged in H and I, respectively. White arrows in I and J point to positive signals. MA, masseter. N=3 for all experiments. Scale bars in A and E indicate 500 μm for A-D and E-F, respectively; scale bar in H indicates 100 μm in H-J.
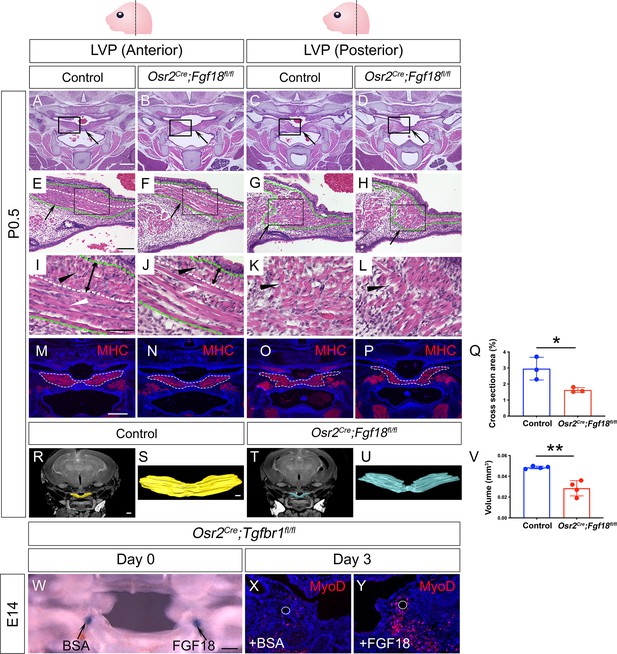
Myogenic cells are reduced in the LVP region of Osr2Cre;Fgf18fl/fl mice, while Fgf18 increases MyoD+ myogenic cells in Osr2Cre;Tgfbr1fl/fl soft palate slice cultures.
(A–L) H&E staining in coronal sections of LVP region at P0.5 from control and Osr2Cre;Fgf18fl/fl mice. Boxed areas in A-D and E-H are enlarged in E-H, and I-L, respectively. Black arrows point to the palatal shelf in A-D and the LVP in E-H. Green dotted line outlined the LVP in E-H. Black and white triangles point to perpendicular muscle fibers in I-L and parallel fibers in I-J. The white dotted line indicates the boundaries of perpendicular and parallel fibers in E, F, I, and J. Double-ended arrows indicate the thickness of perpendicular fibers in I and J. N=4. (M–Q) Immunofluorescence and quantification of MHC (red) staining on coronal section of the LVP region of control Osr2Cre;Fgf18fl/fl mice at P0.5. MHC+ areas in the LVP region for quantification of cross-section areas are outlined by white dashed lines in M-P. The percentage of MHC-stained area out of the whole image area is used for quantification in Q. *, p≤0.05. N=3. Statistical significance was assessed by unpaired t-test with two-tailed calculations. Data is presented as mean ± SEM. (R–V) CT scanning and quantitative analysis of the muscle volume of control and Osr2Cre;Tgfbr1fl/fl LVP at P0.5. A representative reconstructed control LVP from CT scanning is indicated in yellow (R and S) and an Osr2Cre;Tgfbr1fl/fl reconstructed LVP is indicated in teal (T and U). **, p≤0.01. N=4. Statistical significance was assessed by unpaired t-test with two-tailed calculations. Data is presented as mean ± SEM. (W) A 300 μm coronal slice of the LVP region at E14 from Osr2Cre;Fgf18fl/fl mouse for slice culture following bead implantation. Arrows point to BSA- or FGF18-treated bead. (X–Y) Immunofluorescence of MyoD (red) in the coronal section of LVP region from Osr2Cre;Fgf18fl/fl mouse cultured for 3 days with BSA bead (X) and FGF18 bead (Y). White circles indicate the location of the BSA beads in X and FGF18 beads in Y. N=3. Fgf18fl/fl or Fgf18fl/+ littermates were used as controls for Osr2Cre;Fgf18fl/fl mice. Scale bars in A, E, I, M, R, S, and W indicate 500 μm in in A-D, 100 μm in E-H, 50 μm in I-L, 400 μm in M-P, 400 μm in R and T, 100 μm in S and U, and 100 μm in W-Y, respectively.
-
Figure 7—source data 1
Source data for Figure 7Q.
- https://cdn.elifesciences.org/articles/80405/elife-80405-fig7-data1-v1.xlsx
-
Figure 7—source data 2
Source data for Figure 7V.
- https://cdn.elifesciences.org/articles/80405/elife-80405-fig7-data2-v1.xlsx
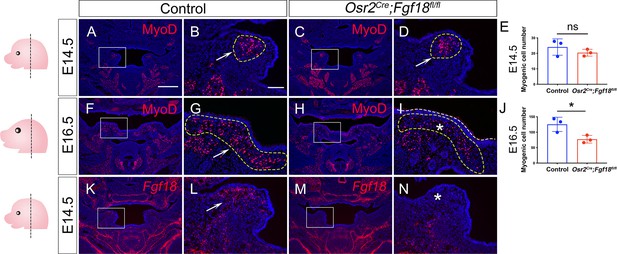
Osr2Cre;Fgf18fl/fl mice exhibit myogenic defects during LVP development.
(A–J) Immunofluorescence and quantification of MyoD (red) in coronal section of LVP region at E14.5 (A–E) and E16.5 (F–J) from control and Osr2Cre;Fgf18fl/fl mice. Boxed areas in A, C, F, and H are enlarged in B, D, G, and I, respectively. White arrows in B, D, and G point to the presence of MyoD+ cells. The white asterisk in I indicates a decrease of MyoD+ cells in E16.5 Osr2Cre;Fgf18fl/fl mice. Note that the myogenic cell numbers in E14.5 are comparable between control and Osr2Cre;Fgf18fl/fl mice at E14.5 (E) but are significantly decreased in the mutant at E16.5 (J). *, p≤0.05; ns, not significant. Statistical significance was assessed by unpaired t-test with two-tailed calculations. Data is presented as mean ± SEM. Yellow dotted line outlines the myogenic regions where the qualification analysis was performed. (K–N) BaseScope in situ hybridizations of Fgf18 Exon1C (red) in the coronal section of LVP region at E14.5 from control and Osr2Cre;Fgf18fl/fl mice. Boxed areas in K and M are enlarged in L and N, respectively. The white arrow in L points to a positive signal, and the asterisk in N points to a reduced signal. Left panel schematics depict the orientation and level of the sections. N=3 for all experiments. Fgf18fl/fl or Fgf18fl/+ littermates were used as controls for Osr2Cre;Fgf18fl/fl mice. Scale bar in A indicates 500 μm in A, C, F, H, K, and M; scale bar in B indicates 100 μm in B, D, G, I, L, and N.
-
Figure 7—figure supplement 1—source data 1
Source data for Figure 7—figure supplement 1E.
- https://cdn.elifesciences.org/articles/80405/elife-80405-fig7-figsupp1-data1-v1.xlsx
-
Figure 7—figure supplement 1—source data 2
Source data for Figure 7—figure supplement 1J.
- https://cdn.elifesciences.org/articles/80405/elife-80405-fig7-figsupp1-data2-v1.xlsx
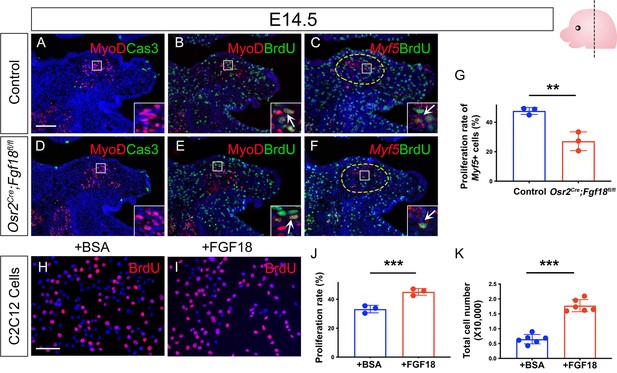
Loss of Fgf18 in Osr2Cre;Fgf18fl/fl mice leads to defective proliferation of Myf5+ myogenic cells during LVP development, while exogenous FGF18 can increase the proliferation of C2C12 myogenic cells.
(A–F) Immunofluorescence and RNAScope in situ hybridization for MyoD (red) and Caspase 3 (A and D), MyoD (red) and BrdU (green) (B and E), and Myf5 (red) and BrdU (green) (C and F) in coronal sections of LVP region at E14.5 from control and Osr2Cre;Fgf18fl/fl mice. Insets are enlarged from the boxed areas. Yellow dotted line outlines the myogenic regions where the qualification analysis was performed. N=3. Fgf18fl/fl or Fgf18fl/+ littermates were used as controls for Osr2Cre;Fgf18fl/fl mice. The schematic in the top right corner depicts the orientation and level of the section. (G) Quantification of the percentage of BrdU+/My5 + double-positive cells out of the total Myf5 + cells in the LVP region from E14.5 control and Osr2Cre;Fgf18fl/fl mice. **, p≤0.01. N=3. Statistical significance was assessed by unpaired t-test with two-tailed calculations. Data is presented as mean ± SEM. (H–J) Immunofluorescence and quantification of BrdU (red) staining on C12C12 myogenic ells following 1 day of treatment with BSA or 500 ng/ml FGF18. Proliferative rate is calculated by the percentage of BrdU+ cells out of the total number of cells. ***, p≤0.001. N=3. Statistical significance was assessed by unpaired t-test with two-tailed calculations. Data is presented as mean ± SEM. (K) Quantification of total number of C12C12 myogenic cells after culture for 3 days with BSA or 500 ng/ml FGF18. ***, p≤0.001. N=6. Statistical significance was assessed by unpaired t-test with two-tailed calculations. Data is presented as mean ± SEM. Scale bar in A indicates 100 μm for A-F and 30 μm for the insets in A-F; scale bar in H indicates 200 μm for H-I.
-
Figure 7—figure supplement 2—source data 1
Source data for Figure 7—figure supplement 2G.
- https://cdn.elifesciences.org/articles/80405/elife-80405-fig7-figsupp2-data1-v1.xlsx
-
Figure 7—figure supplement 2—source data 2
Source data for Figure 7—figure supplement 2J.
- https://cdn.elifesciences.org/articles/80405/elife-80405-fig7-figsupp2-data2-v1.xlsx
-
Figure 7—figure supplement 2—source data 3
Source data for Figure 7—figure supplement 2K.
- https://cdn.elifesciences.org/articles/80405/elife-80405-fig7-figsupp2-data3-v1.xlsx
Tables
| Reagent type(species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Strain, strain background (M. musculus) | Osr2Cre | Rulang Jiang, Cincinnati Children’s Hospital; Chen et al., 2009 | ||
| Strain, strain background (M. musculus) | Tgfbr1fl/fl | Dudas et al., 2006; Larsson et al., 2001 | ||
| Strain, strain background (M. musculus) | Fgf18fl/fl | David Ornitz, Washington University School of Medicine; Hagan et al., 2019 | ||
| Strain, strain background (M. musculus) | Rosa26LSL-tdTomato | Jackson Laboratory; Madisen et al., 2010 | Stock No. 007905 RRID:IMSR_JAX:007905 | |
| Strain, strain background (M. musculus) | C57BL/6J | Jackson Laboratory | Stock No. 000664 RRID:IMSR_JAX:000664 | |
| Sequence-based reagent | RNAScope Probe-Mm-Aldh1a2 | Advanced Cell Diagnostics | Cat# 447391 | |
| Sequence-based reagent | RNAScope Probe-Mm-Tbx15 | Advanced Cell Diagnostics | Cat# 558761 | |
| Sequence-based reagent | RNAScope Probe- Mm-Smoc2 | Advanced Cell Diagnostics | Cat# 318541 | |
| Sequence-based reagent | RNAScope Probe-Mm-Fgf18 | Advanced Cell Diagnostics | Cat# 495421 | |
| Sequence-based reagent | RNAScope Probe-Mm-Lama4 | Advanced Cell Diagnostics | Cat# 494901 | |
| Sequence-based reagent | RNAScope Probe- Mm-Fgfr1 | Advanced Cell Diagnostics | Cat# 443491 | |
| Sequence-based reagent | RNAScope Probe- Mm-Fgfr2 | Advanced Cell Diagnostics | Cat# 443501 | |
| Sequence-based reagent | RNAScope Probe- Mm-Fgfr3 | Advanced Cell Diagnostics | Cat# 440771 | |
| Sequence-based reagent | RNAscope Probe- Mm-Fgfr4 | Advanced Cell Diagnostics | Cat# 443511 | |
| Sequence-based reagent | RNAScope Probe- Mm-Myf5 | Advanced Cell Diagnostics | Cat# 492911 | |
| Sequence-based reagent | RNAScope Probe-Mm-Hic1 | Advanced Cell Diagnostics | Cat# 464131 | |
| Sequence-based reagent | RNAScope Probe-Mm-Creb5 | Advanced Cell Diagnostics | Cat# 572891 | |
| Sequence-based reagent | RNAScope Probe-Mm-Meox1-C2 | Advanced Cell Diagnostics | Cat# 530641-C2 | |
| Sequence-based reagent | RNAScope Probe-Mm-Etv5 | Advanced Cell Diagnostics | Cat# 316961 | |
| Sequence-based reagent | RNAScope Probe-Mm-Bach2-C3 | Advanced Cell Diagnostics | Cat# 887121-C3 | |
| Sequence-based reagent | RNAScope Probe-Mm-Myod1 | Advanced Cell Diagnostics | Cat# 316081 | |
| Sequence-based reagent | RNAScope Probe- Mm-Myod1-C2 | Advanced Cell Diagnostics | Cat# 316081-C2 | |
| Sequence-based reagent | BaseScope Probe- BA-Mm-Fgf18-3zz-st-C1 | Advanced Cell Diagnostics | Cat# 1118021-C1 | |
| Antibody | Mouse monoclonal, Myosin heavy chain (MHC) | DSHB | Cat# MF20 | (1:25) |
| Antibody | Rabbit polyclonal, RFP | Rockland | Cat# 600-401-379 | (1:500) |
| Antibody | Rabbit monoclonal, pSmad2 | Cell Signaling Technology | Cat# 3108 RRID:AB_490941 | (1:500) |
| Antibody | Rabbit monoclonal, MyoD | Abcam | Cat# ab133627 RRID:AB_2890928 | (1:200) |
| Antibody | Rat monoclonal, BrdU | Abcam | Cat# ab6326 RRID: AB_305426 | (1:100) |
| Antibody | Rabbit Polyclonal, Cleaved Caspase-3 | Cell Signaling Technology | Cat# 9661 RRID: AB_2341188 | (1:100) |
| Antibody | Rabbit monoclonal, Smad2/3 | Cell Signaling Technology | Cat# 8685 RRID:AB_10889933 | (1:20) |
| Antibody | Rabbit monoclonal, IgG | Cell Signaling Technology | Cat# 3900 RRID:AB_1550038 | (1:20) |
| Antibody | Goat polyclonal anti-Mouse Alexa Fluor 488 | Life Technologies | Cat# A-11001 RRID:AB_2534069 | (1:200) |
| Antibody | Goat polyclonal anti-Rat Alexa Fluor 488 | Life Technologies | Cat# A-11006 RRID:AB_141373 | (1:200) |
| Antibody | Goat polyclonal anti-Rabbit Alexa Fluor 488 | Life Technologies | Cat# A-11008 RRID:AB_143165 | (1:200) |
| Antibody | Goat polyclonal anti-Rabbit Alexa Fluor 568 | Life Technologies | Cat# A-11036 RRID:AB_10563566 | (1:200) |
| Antibody | Goat polyclonal anti-rabbit IgG Antibody (H+L), HRP | Vector Laboratories | Cat# PI-1000 RRID:AB_2336198 | (1:200) |
| Peptide, recombinant protein | Recombinant human TGF-β1 | R&D Systems | Cat# 7754-BH | |
| Peptide, recombinant protein | Recombinant human FGF18 | Peprotech | Cat# 100–28 | |
| Cell line (Mus musculus) | C2C12 | ATCC | Cat# CRL-1772 RRID:CVCL_0188 | |
| Commercial assay or kit | RNAScope Multiplex Fluorescent Kit v2 | Advanced Cell Diagnostics | Cat# 323110 | |
| Commercial assay or kit | BaseScope detection reagent kit v2-Red | Advanced Cell Diagnostics | Cat# 323910 | |
| Commercial assay or kit | TSA Plus Cyanine 3 System | Perkin Elmer | Cat# NEL744001KT | |
| Commercial assay or kit | TSA Plus Fluoresceine System | Perkin Elmer | Cat# NEL771B001KT | |
| Commercial assay or kit | RNeasy Micro Kit | QIAGEN | Cat# 74004 | |
| Commercial assay or kit | iScript Advanced cDNA Synthesis Kit | Bio-Rad | Cat# 1725038 | |
| Commercial assay or kit | SsoFast EvaGreen Supermix | Bio-Rad | Cat# 1725201 | |
| Commercial assay or kit | Chromium single-cell 3’ v2 reagent kit | 10x Genomics | Cat# PN-120267 | |
| Commercial assay or kit | Cut and Run assay kit | Cell Signaling | Cat# 86652 | |
| Software, algorithm | Cell ranger | 10x Genomics.Inc | RRID:SCR_017344 | |
| Software, algorithm | Seurat | Satija lab | RRID:SCR_016341 | |
| Software, algorithm | CellChat | Jin Lab | ||
| Software, algorithm | SCENIC | Aerts lab | RRID:SCR_017247 | |
| Software, algorithm | GraphPad Prism | GraphPad Software | RRID:SCR_002798 |





