Dynamic compartmentalization of the pro-invasive transcription factor NHR-67 reveals a role for Groucho in regulating a proliferative-invasive cellular switch in C. elegans
Figures
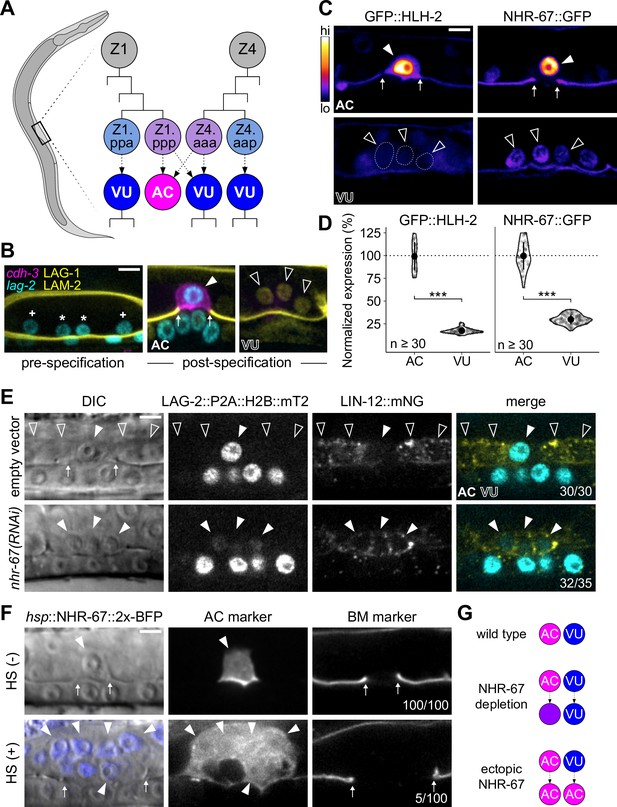
Invasive AC fate correlates to high levels of NHR-67.
(A) Schematic of C. elegans anchor cell (AC, magenta) and ventral uterine (VU, blue) cell fate specification from the Z1 and Z4 somatic gonad precursor cell lineages (p, posterior daughter; a, anterior daughter). (B) Micrographs depicting AC and VU cell differentiation over developmental time. AC/VU precursors express LAG-2 (H2B::mTurquoise), which eventually becomes restricted to the AC, whereas VU cells express LAG-1 (mNeonGreen) post-specification. The differentiated AC (cdh-3p::mCherry::moeABD) then invades through the underlying basement membrane (LAM-2::mNeonGreen). (C–D) Representative heat map micrographs (C) and quantification (D) of GFP-tagged HLH-2 and NHR-67 expression in the AC and VU cells at the time of AC invasion. (E) Expression of Notch (lin-12::mNeonGreen) and Delta (lag-2::P2A::H2B::mTurquoise2) following RNAi-induced knockdown of NHR-67 compared to empty vector control. (F) Micrographs depicting the ectopic invasive ACs (cdh-3p::mCherry::moeABD, arrowheads) and expanded basement membrane (laminin::GFP, arrows) gap observed following heat shock-induced expression of NHR-67 (hsp::NHR-67::2x-BFP) compared to non-heat shocked controls. (G) Schematic summarizing AC and VU cell fates that result from perturbations of NHR-67 levels. For all figures: asterisk (*), AC/VU precursor; plus (+), VU precursor; solid arrowhead, AC; open arrowhead, VU cell; arrows, basement membrane breach. Statistical significance determined by Student’s t-test (*p>0.05, **p>0.01, ***p>0.001). Scale bars, 5 µm.
-
Figure 1—source data 1
Raw data of GFP-tagged transcription factor expression in the anchor cell (AC) and ventral uterine (VU) cells, as reported in Figure 1C and D and Figure 1—figure supplement 1B and C.
In all source files: ROI, region of interest; BG, background; BS, background-subtracted.
- https://cdn.elifesciences.org/articles/84355/elife-84355-fig1-data1-v1.zip
-
Figure 1—source data 2
Raw data of LAG-2::P2A::H2B::mTurquoise2 and LIN-12::mNeonGreen expression in NHR-67-deficient anchor cells (ACs) compared to control AC and ventral uterine (VU) cells, as reported in Figure 1E and Figure 1—figure supplement 2A and B.
- https://cdn.elifesciences.org/articles/84355/elife-84355-fig1-data2-v1.zip
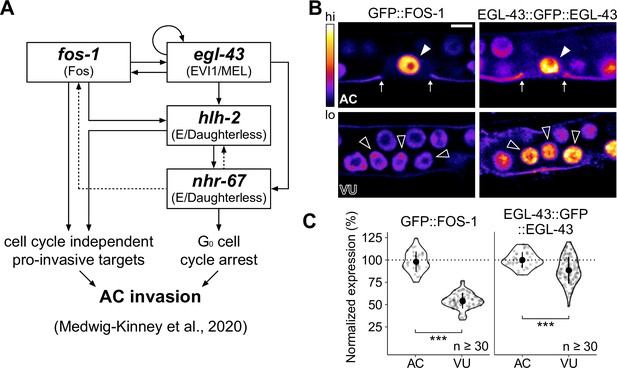
Expression of pro-invasive transcription factors EGL-43 and FOS-1 in the somatic gonad.
(A) Schematic of the anchor cell (AC) pro-invasive gene regulatory network comprised of four transcription factors: EGL-43, FOS-1, HLH-2, and NHR-67. (B–C) Representative heat-map micrographs (B) and quantification (C) of GFP-tagged EGL-43 and FOS-1 expression in the AC and ventral uterine (VU) cells.
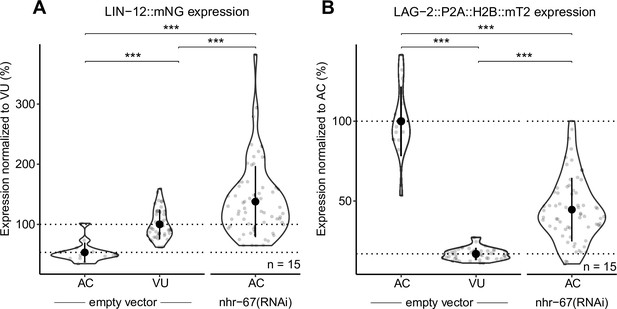
NHR-67-deficient anchor cells (ACs) express both Notch and Delta.
Quantification of Notch (LIN-12::mNeonGreen) (A) and Delta (LAG-2::P2A::H2B::mTurquoise2) (B) expression in NHR-67(RNAi) treated ACs compared to empty vector control AC and ventral uterine (VU) cells.
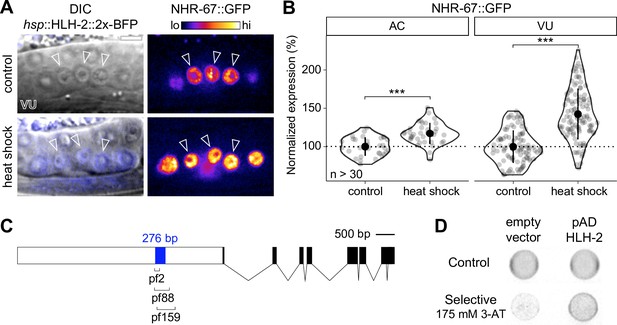
NHR-67 expression is downregulated in ventral uterine (VU) cells through direct transcriptional regulation by HLH-2.
(A–B) Representative heat map micrographs (A) and quantification (B) of NHR-67::GFP expression in VU cells following heat shock-induced expression of HLH-2 (2x-BFP) compared to non-heat shocked controls. (C) Schematic of a 276 bp putative regulatory element within the promoter of NHR-67 (Bodofsky et al., 2018), annotated with the location of three hypomorphic mutations (pf2, pf88, and pf159). (D) Yeast one-hybrid experiment pairing HLH-2 Gal4-AD prey with the 276 bp fragment of the NHR-67 promoter as bait on SC-HIS-TRP plates with and without competitive inhibitor 3-AT (175 mM).
-
Figure 2—source data 1
Raw data of NHR-67::GFP expression in the anchor cell (AC) and ventral uterine (VU) cells following heat-shock inducible expression of HLH-2, as reported in Figure 2A and B.
- https://cdn.elifesciences.org/articles/84355/elife-84355-fig2-data1-v1.zip
-
Figure 2—source data 2
Raw data of GFP::HLH-2 and NHR-67::TagRFP-T expression in the anchor cell (AC) and ventral uterine (VU) cells following uba-1 RNAi treatment compared to empty vector controls, as reported in Figure 2—figure supplement 1B–D.
- https://cdn.elifesciences.org/articles/84355/elife-84355-fig2-data2-v1.zip
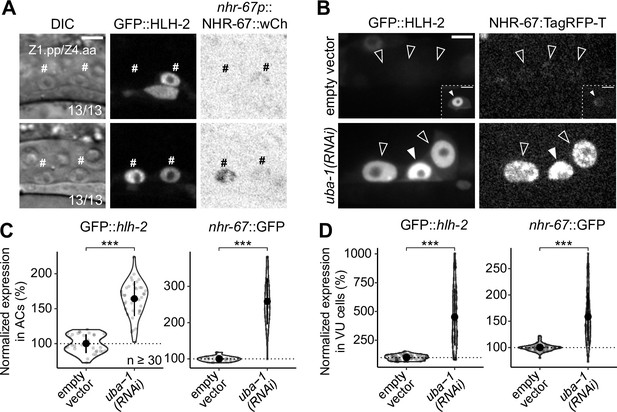
Onset of expression and regulatory interaction between NHR-67 and HLH-2 in the somatic gonad.
(A) Micrographs depicting the onset of GFP-tagged HLH-2 and a wCherry-labeled NHR-67 transgene (inverted to aid visualization) in Z1.pp and Z4.aa cells at early (top) and late (bottom) stages. (B–D) Representative micrographs (B) and quantification (C–D) of GFP-tagged HLH-2 and TagRFP-T-tagged NHR-67 in anchor cell (AC) (C) and ventral uterine (VU) cells (D) following uba-1(RNAi) compared to control. Insets depict different z-planes of the same image.
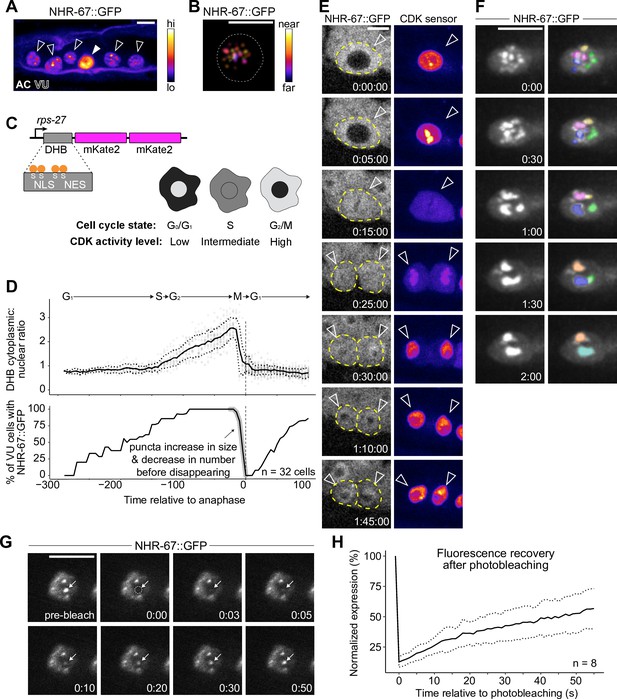
NHR-67 dynamically compartmentalizes in nuclei of ventral uterine (VU) cells.
(A) Heat-map maximum intensity projection of NHR-67::GFP showing protein localization in the anchor cell (AC) and VU cells. (B) Spatial color-coded projection of NHR-67::GFP punctae in VU cells, with nuclear border indicated with a dotted line. (C) Schematic of DNA Helicase B (DHB) based CDK sensor and its dynamic localization over the cell cycle. (D) Graphs depicting CDK activity levels and corresponding cell cycle state (top), and percentage of cells exhibiting NHR-67::GFP punctae (bottom) over time, aligned to anaphase. (E) Representative time-lapse of NHR-67::GFP over the course of a cell cycle, with cell membranes indicated with dotted lines. (F) Time-lapse depicting NHR-67::GFP punctae fusion prior to cell division. Right panels are pseudo-colored. (G–H) Representative images (G) and quantification (H) depicting fluorescence recovery of NHR-67::GFP following photobleaching of individual punctae (arrow).
-
Figure 3—source data 1
Raw data of CDK sensor (DHB) ratios in ventral uterine (VU) cells over time, as reported in Figure 3D and E.
- https://cdn.elifesciences.org/articles/84355/elife-84355-fig3-data1-v1.zip
-
Figure 3—source data 2
Raw data of NHR-67::GFP puncta expression following photobleaching overtime, as reported in Figure 3G and H.
- https://cdn.elifesciences.org/articles/84355/elife-84355-fig3-data2-v1.zip
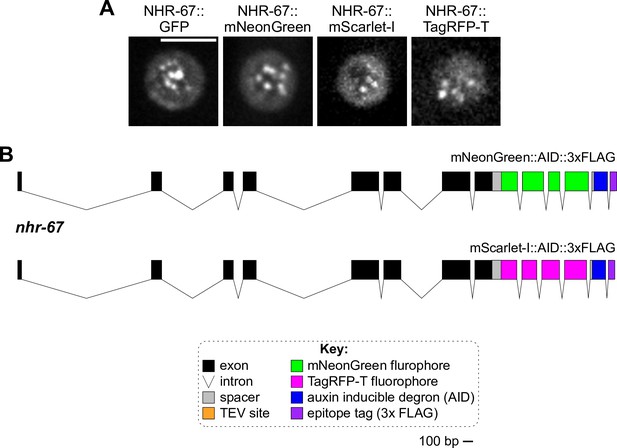
Knock-in alleles of NHR-67.
(A) Representative images of ventral uterine (VU) cells exhibiting punctae formed by NHR-67 tagged with GFP, mNeonGreen, mScarlet-I, and TagRFP-T. (B) Schematics of the new endogenously tagged loci generated in this paper for NHR-67. Scale bar, 100 base pairs (bp).
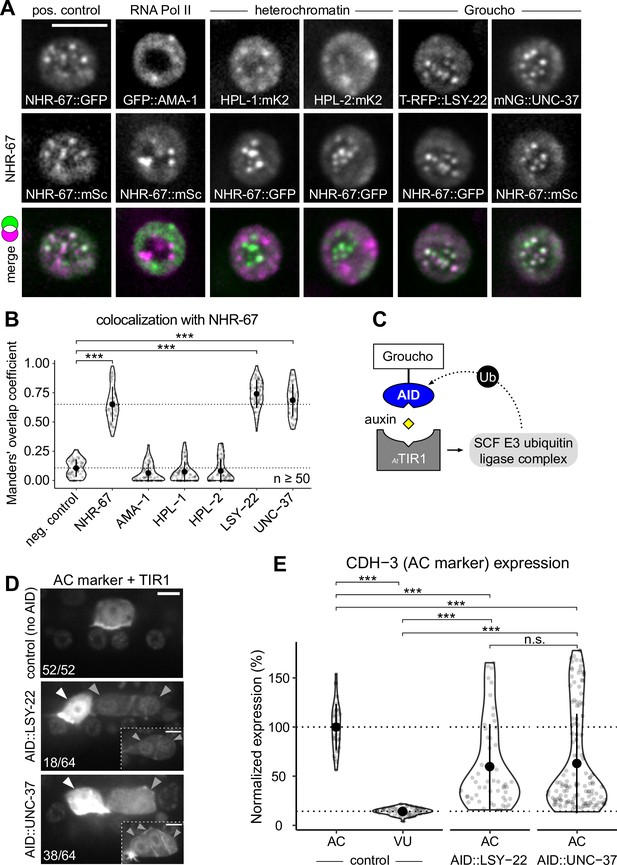
Groucho homologs LSY-22 an UNC-37 colocalize with NHR-67 punctae and contribute to maintenance of ventral uterine (VU) cell fate.
(A) Co-visualization of NHR-67 with RNA Polymerase II (GFP::AMA-1), HP1 heterochromatin proteins (HPL-1::mKate2 and HPL-2::mKate2), and Groucho homologs (TagRFP-T::LSY-22 and mNeonGreen::UNC-37) in VU cells using endogenously tagged alleles. (B) Quantification of colocalization, with plot reporting Manders’ overlap coefficients compared to negative controls (90-degree rotation of one channel) and positive controls. (C) Schematic of the auxin-inducible degron (AID) system, where AtTIR1 mediates proteasomal degradation of AID-tagged proteins in the presence of auxin. (D) Representative images of phenotypes observed following individual AID-depletion of UNC-37 and LSY-22 compared to control animals without AID-tagged alleles. All animals compared here are expressing TIR1 ubiquitously (rpl-28p::AtTIR1::T2A::mCherry::HIS-11) and an anchor cell (AC) marker (cdh-3p::mCherry::moeABD). Insets depict different z-planes of the same image. (E) Quantification of AC marker (cdh-3p::mCherry::moeABD) expression in ectopic ACs resulting from AID-depletion of UNC-37 and LSY-22 compared to control AC and VU cells.
-
Figure 4—source data 1
Raw data of protein colocalization in ventral uterine (VU) cells, as reported in Figure 4A and B and Figure 5D and E.
- https://cdn.elifesciences.org/articles/84355/elife-84355-fig4-data1-v1.zip
-
Figure 4—source data 2
Raw data of CDH-3 expression in the ectopic anchor cells (ACs) resulting from auxin-mediated depletion of AID-tagged LSY-22 or UNC-37, as reported in Figure 4D and E.
- https://cdn.elifesciences.org/articles/84355/elife-84355-fig4-data2-v1.zip

Knock-in alleles of LSY-22.
Schematics of the new endogenously tagged loci generated in this paper for LSY-22. Scale bar, 100 base pairs (bp).

UNC-37 mutants show ectopic expression of anchor cell (AC) markers.
Ectopic expression of AC marker (cdh-3p::mCherry::moeABD) in hypomorphic (unc-37(e262wd26)) and null (unc-37(wd17wd22)) alleles of unc-37 compared to wild-type unc-37. Insets depict different z-planes of the same image.
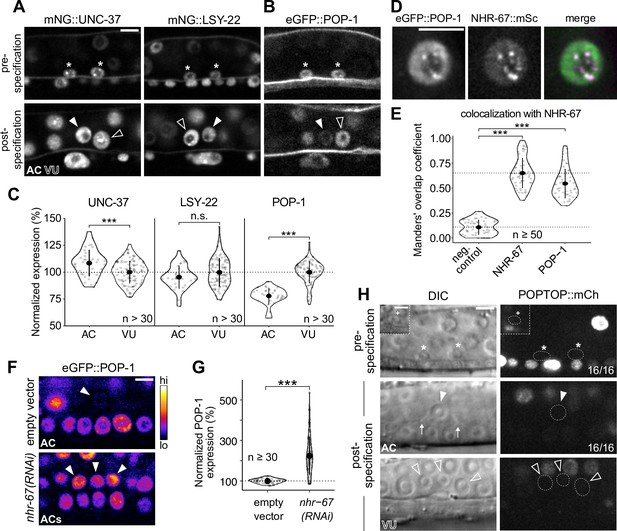
POP-1 is enriched in ventral uterine (VU) cells and colocalizes with NHR-67 punctae.
(A–B) Expression of mNeonGreen::UNC-37 and mNeonGreen::LSY-22 (A) and eGFP::POP-1 (B) in the anchor cell (AC)/VU precursors pre-specification (left), as well as in the AC and VU cells post-specification (right). (C) Quantification of UNC-37, LSY-22, and POP-1 expression at the time of AC invasion. (D) Co-visualization of NHR-67::mScarlet-I and eGFP::POP-1 in the VU cells. (E) Quantification of POP-1 and NHR-67 colocalization, with plot reporting Manders’ overlap coefficient compared to negative and positive controls. (F-G) Micrographs (F) and quantification (G) of eGFP-tagged POP-1 expression in proliferative ACs following RNAi depletion of NHR-67 compared to empty vector control. (H) Representative micrographs showing expression of POPTOP, a synthetic pop-1-activated reporter construct, in wild-type ACs, VU cells, and their precursors. Insets depict different z-planes of the same image.
-
Figure 5—source data 1
Raw data of mNG::UNC-37, mNG::LSY-22, and eGFP::POP-1 expression in the AC/VU precursors, the anchor cell (AC), and ventral uterine (VU) cells, as reported in Figure 5A–C and Figure 5—figure supplement 1A and B.
- https://cdn.elifesciences.org/articles/84355/elife-84355-fig5-data1-v1.zip
-
Figure 5—source data 2
Raw data of eGFP::POP-1 expression in anchor cells (ACs) resulting from RNAi knockdown of transcription factors and chromatin modifiers compared to empty vector controls, reported in Figure 5F and G and Figure 5—figure supplement 2A and B.
- https://cdn.elifesciences.org/articles/84355/elife-84355-fig5-data2-v1.zip
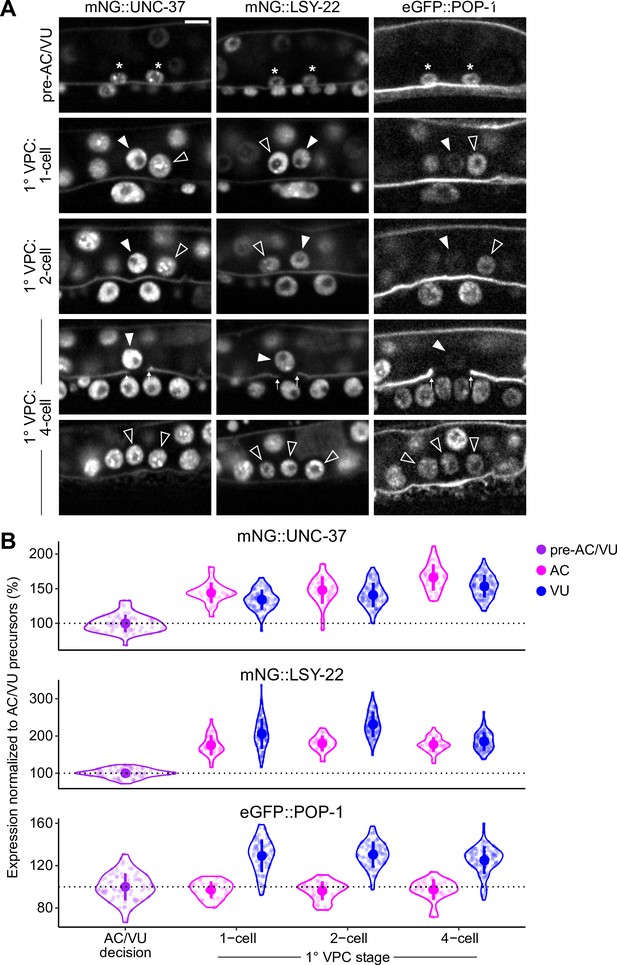
Expression of LSY-22, UNC-37, and POP-1 over developmental time.
(A–B) Developmental series (A) and quantified expression (B) of mNeonGreen::UNC-37, mNeonGreen::LSY-22, and eGFP::POP-1 expression in the AC/VU precursors, anchor cell (AC), and ventral uterine (VU) cells over time. Following AC/VU cell specification, animals are staged by the division of the underlying primary vulval precursor cells (1° VPCs).
POP-1 function in ventral uterine (VU) cells is distinct from the activating role in distal somatic gonad.
(A) Schematics representing the dual functions of POP-1. In the presence of Wnt signaling, POP-1 binds to its co-activator β-catenin (e.g. SYS-1) and activates transcription of its target genes. In the absence of Wnt signaling, POP-1 binds to its co-repressor Groucho (UNC-37) and represses transcription of its target genes. (B) Representative micrographs of eGFP::POP-1 and POPTOP (pes-10::7x-TCF::mCherry) expression in the anchor cell (AC), dorsal uterine cells (DU), spermatheca/sheath cells (SS), and VU cells. (C) Schematic of SYS-1 (β-catenin) expression in the Z1/Z4 lineage (based on Phillips et al., 2007).

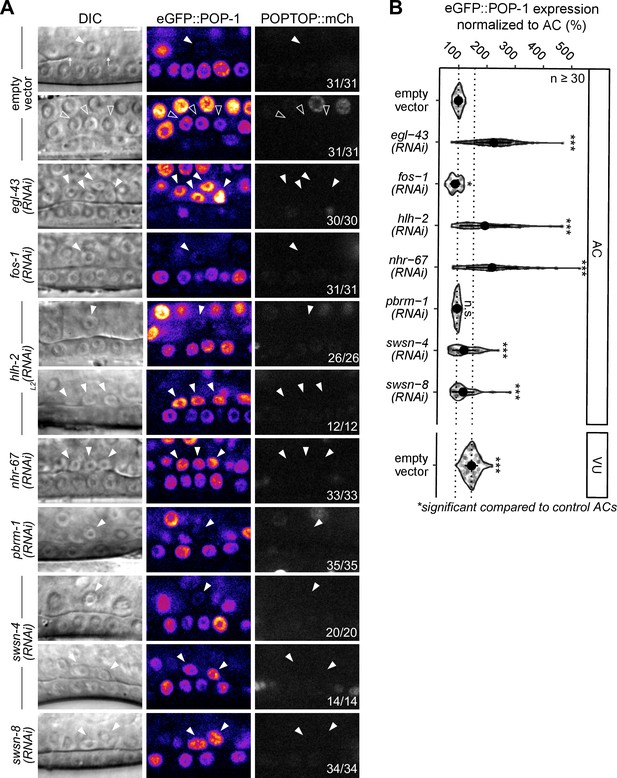
POP-1 expression is regulated by the cell cycle-dependent pro-invasion pathway.
(A–B) Representative micrographs of eGFP::POP-1 and POPTOP (pes-10::7x-TCF::mCherry) following RNAi-induced knockdown of pro-invasive transcription factors and chromatin modifiers compared to control anchor cell (AC) and ventral uterine (VU) cells. (B) Quantification of eGFP::POP-1 expression in ACs following RNAi treatments. Here, the presence of multiple ACs are the result of the failure of the AC to exit the cell cycle.
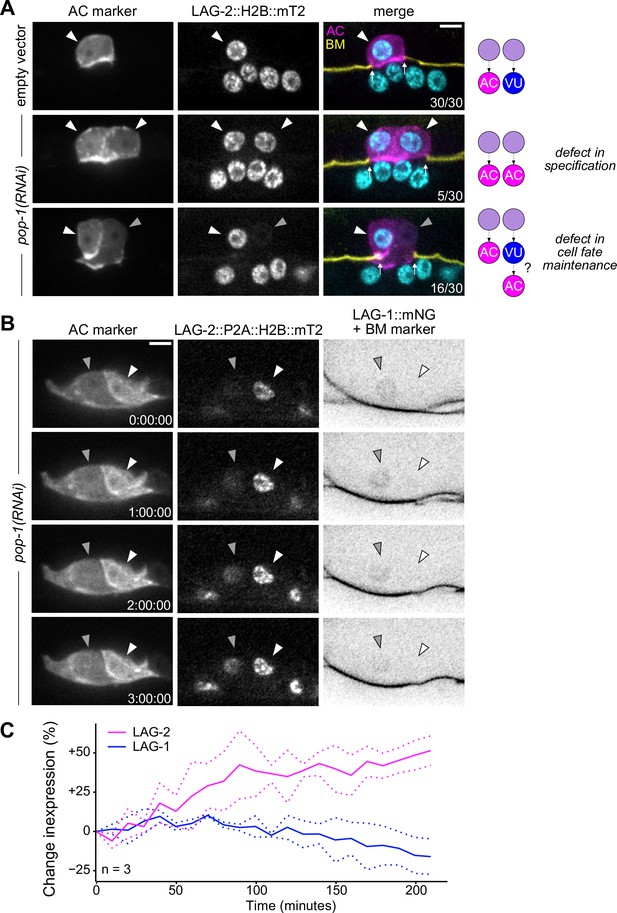
Ectopic anchor cells (ACs) arise through VU-to-AC cell fate transformation.
(A) Representative images of ectopic AC (cdh-3p::mCherry::moeABD; LAG-2::P2A::H2B::mTurquoise2) phenotypes observed following RNAi depletion of POP-1. Schematics (right) depict potential explanations for observed phenotypes. (B) Expression of AC markers and a VU cell marker (LAG-1::mNeonGreen, inverted to aid visualization) in pop-1(RNAi) treated animals over time. (C) Quantification of LAG-2 (magenta) and LAG-1 (blue) expression in transdifferentiating cells produced by pop-1(RNAi) over time.
-
Figure 6—source data 1
Raw data of LAG-2::P2A::H2B::mT2 and LAG-1::mNG expression during VU-to-AC transdifferentiation, as reported in Figure 6B and C.
- https://cdn.elifesciences.org/articles/84355/elife-84355-fig6-data1-v1.zip
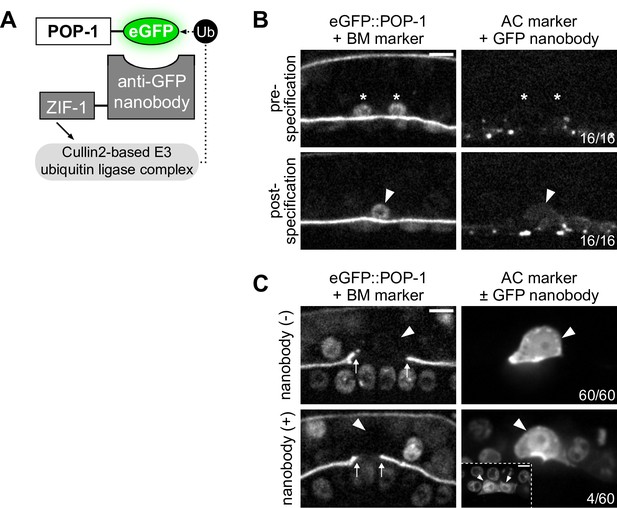
POP-1 functions to regulate AC/VU cell fates post-specification.
(A) Schematic of the anti-GFP nanobody protein degradation system (based on Wang et al., 2017). (B) Micrographs demonstrating that the anti-GFP nanobody (driven under the egl-43L promoter) is not expressed pre-specification or even shortly after when the presumptive anchor cell (AC) begins to express its differentiated cell reporter (cdh-3). (C) With decreased levels of pop-1, a low penetrance (~7%) of multi-AC phenotypes were observed.
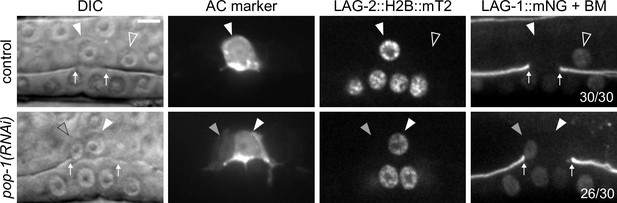
Ectopic anchor cells (ACs) resulting from pop-1 perturbation express ventral uterine (VU) cell markers.
Expression of AC markers (cdh-3p::mCherry::moeABD; LAG-2::P2A::H2B::mTurquoise2) and VU marker (LAG-1::mNeonGreen) in pop-1(RNAi) treated animals compared to empty vector control.
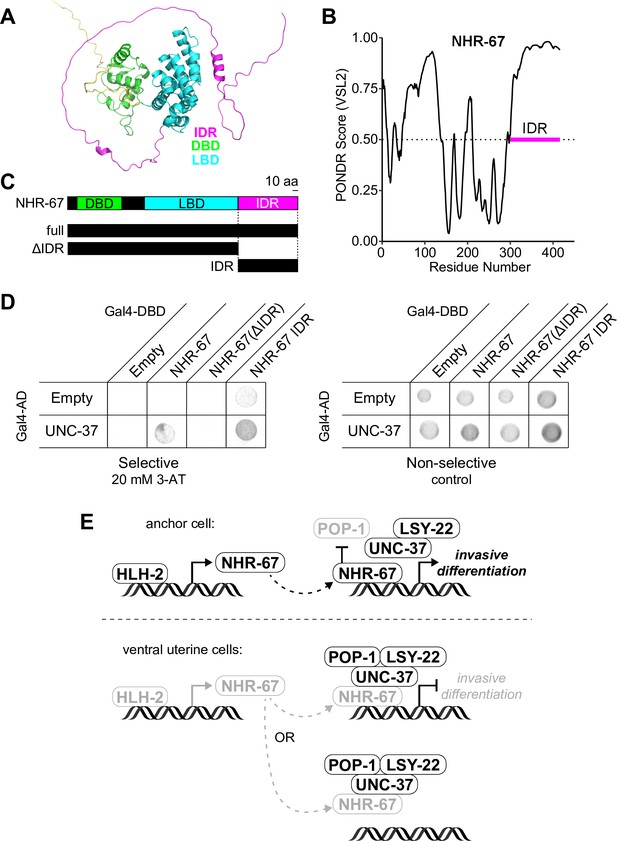
NHR-67 binds to UNC-37 through IDR-mediated protein-protein interaction.
(A) Predicted structure of NHR-67 generated by AlphaFold. (B) Measure of intrinsic disorder of NHR-67 using the PONDR VSL2 prediction algorithm. (C) Schematic of NHR-67 protein-coding sequences used for Yeast two-hybrid experiments with reference to its intrinsically disordered region (IDR, magenta), DNA binding domain (DBD, green), and ligand binding domain (LBD, cyan). Scale bar, 10 amino acids. (D) Yeast two-hybrid experiment shows pairing of UNC-37 with either full-length NHR-67 or the IDR alone allows for yeast growth in the presence of competitive inhibitor 3-AT (20 mM). (E) Possible models of the roles of NHR-67, UNC-37, LSY-22, and POP-1 in the maintenance of anchor cell (AC) and ventral uterine (VU) cell fate. In the ventral uterine cells, the association of NHR-67 with the Groucho/TCF complex may result in the repression of NHR-67 targets (top) or the sequestration of NHR-67 away from its targets (bottom).
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Strain, strain background (C. elegans) | DQM335 | Medwig-Kinney et al., 2020 | egl-43(bmd88[egl-43p::EGL-43::loxP::GFP::EGL-43]) II; qyIs225[cdh-3p::mCherry::moeABD] V; qyIs7[laminin::GFP] X. | |
| Strain, strain background (C. elegans) | DQM350 | Medwig-Kinney et al., 2020 | hlh-2(bmd90[hlh-2p::loxP::GFP::HLH-2]) I; qyIs225[cdh-3p::mCherry::moeABD] V; qyIs7[laminin::GFP] X. | |
| Strain, strain background (C. elegans) | DQM354 | This paper | nhr-67(syb509[nhr-67p::NHR-67::GFP]) IV; bmd66[loxP::egl-43p::GFP-nanobody::P2A::HIS-58::mCherry] I; qyIs225[cdh-3p::mCherry::moeABD] V; qyIs7[laminin::GFP] X. | |
| Strain, strain background (C. elegans) | DQM368 | Medwig-Kinney et al., 2020 | nhr-67(syb509[nhr-67p::NHR-67::GFP]) IV; qyIs225[cdh-3p::mCherry::moeABD] V; qyIs7[laminin::GFP] X. | |
| Strain, strain background (C. elegans) | DQM444 | Medwig-Kinney et al., 2020 | bmd121[hsp::NHR-67::2x-BFP] I; qyIs227[cdh-3p::mCherry::moeABD] I; qyIs7[laminin::GFP] X. | |
| Strain, strain background (C. elegans) | DQM515 | Medwig-Kinney et al., 2020 | fos-1(bmd138[fos-1p::loxP::GFP::FOS-1]) V; qyIs227[cdh-3p::mCherry::moeABD] I; qyIs7[laminin::GFP] X. | |
| Strain, strain background (C. elegans) | DQM704 | Medwig-Kinney et al., 2021 | nhr-67(bmd212[nhr-67p::NHR-67::TagRFP-T::AID]) IV; hlh-2(bmd90[hlh-2p::LoxP::GFP::HLH-2]) I. | |
| Strain, strain background (C. elegans) | DQM800 | This paper | pop-1(he335[pop-1p::eGFP::loxP::POP-1]) I; syIs187[pes-10::7XTCF-mCherry-let-858(3’UTR)+unc-119(+)]. | |
| Strain, strain background (C. elegans) | DQM811 | This paper | qyIs227[cdh-3p::mCherry::moeABD] I; lam-2(qy20[lam-2p::LAM-2::mNeonGreen]) X; lag-2(bmd202[lag-2p::LAG-2::P2A::H2B::mTurquoise2^lox511^ 2xHA]) V. | |
| Strain, strain background (C. elegans) | DQM853 | This paper | hlh-2(bmd90[hlh-2p::loxP::GFP::HLH-2]) I; stIs11476[nhr-67p::NHR-67::H1-wCherry+unc-119(+)]. | |
| Strain, strain background (C. elegans) | DQM957 | This paper | csh128[rpl-28p::TIR1::T2A::mCherry::his-11] II; qyIs225[cdh-3p:: mCherry::moeABD] V; qyIs7[laminin::GFP] X. | |
| Strain, strain background (C. elegans) | DQM958 | This paper | csh140[rpl-28p::TIR1(F79G)::T2A::mCherry::his-11] II; qyIs225[cdh-3p:: mCherry::moeABD] V; qyIs7[laminin::GFP] X. | |
| Strain, strain background (C. elegans) | DQM971 | This paper | pop-1(he335[pop-1p::eGFP::loxP::POP-1]) I; qyIs225[cdh-3p::mCherry::moeABD] V; qyIs7[laminin::GFP] X. | |
| Strain, strain background (C. elegans) | DQM989 | This paper | unc-37(devKi218[unc-37p::mNeonGreen::UNC-37]) I; qyIs225[cdh-3p::mCherry::moeABD] V; qyIs7[laminin::GFP] X. | |
| Strain, strain background (C. elegans) | DQM990 | This paper | unc-37(e262wd26) I; qyIs225[cdh-3p::mCherry::moeABD] V; qyIs7[laminin::GFP] X. | |
| Strain, strain background (C. elegans) | DQM1003 | This paper | nhr-67(syb509[nhr-67p::NHR-67::GFP]) IV; bmd168[rps-27p::DHB::2x-mKate2] II. | |
| Strain, strain background (C. elegans) | DQM1006 | This paper | LSY-22(bmd275[lsy-22p::loxP::mNeonGreen::AID::LSY-22]) I; qyIs225[cdh-3p::mCherry::moeABD] V; qyIs7[laminin::GFP] X. | |
| Strain, strain background (C. elegans) | DQM1008 | This paper | pop-1(he335[pop-1p::eGFP::loxP::POP-1]) I; bmd277[loxP::egl-43p::GFP-nanobody::P2A::HIS-58::mCherry] I; qyIs225[cdh-3p::mCherry::moeABD] V; qyIs7[laminin::GFP] X. | |
| Strain, strain background (C. elegans) | DQM1009 | This paper | unc-37(devKi218[unc-37p::mNeonGreen::UNC-37]) I; nhr-67(wy1633[nhr-67p::NHR-67::mScarlet-I::AID*::3xFLAG]) IV. | |
| Strain, strain background (C. elegans) | DQM1010 | This paper | hpl-2(ot860[hpl-2p::HPL-2::mKate2::HPL-2]) III; nhr-67(syb509[nhr-67p::NHR-67::GFP]) IV. | |
| Strain, strain background (C. elegans) | DQM1011 | This paper | hpl-1(ot841[hpl-1p::HPL-1::mKate2::HPL-1]) X; nhr-67(syb509[nhr-67p::NHR-67::GFP]) IV. | |
| Strain, strain background (C. elegans) | DQM1012 | This paper | LSY-22(bmd214[lsy-22p::lox2272::TagRFP-T::AID::LSY-22]) I; nhr-67(syb509[nhr-67p::NHR-67::GFP]) IV. | |
| Strain, strain background (C. elegans) | DQM1013 | This paper | pop-1(he335[pop-1p::eGFP::loxP::POP-1]) I; nhr-67(syb509[nhr-67p::NHR-67::GFP]) IV. | |
| Strain, strain background (C. elegans) | DQM1014 | This paper | unc-37(wd17wd22)/hT2[bli-4(e937) let-?(q782) qIs48] (I, III); qyIs225[cdh-3p::mCherry::moeABD] V; qyIs7[laminin::GFP] X. | |
| Strain, strain background (C. elegans) | DQM1017 | This paper | ama-1(ers49[ama-1p::AMA-1::AID::GFP]) IV; nhr-67(wy1633[nhr-67p::NHR-67::mScarlet-I::AID*::3xFLAG]) IV. | |
| Strain, strain background (C. elegans) | DQM1051 | This paper | lin-12(ljf31[lin-12::mNeonGreen[C1]^loxP^3xFlag]) III; lag-2(bmd202[lag-2p::LAG-2::P2A::H2B::mTurquoise2^lox511^ 2xHA]) V. | |
| Strain, strain background (C. elegans) | DQM1081 | This paper | bmd168[rps-27p::DHB::2x-mKate2] II; egl-13(devKi199[egl-13p::EGL-13::mNeonGreen]) X; lag-2(bmd202[lag-2p::LAG-2::P2A::H2B::mTurquoise2]) V. | |
| Strain, strain background (C. elegans) | DQM1101 | This paper | lsy-22(bmd275[lsy-22p::^loxP^mNeonGreen::AID::LSY-22]) I; csh128[rpl-28p::TIR1::P2A::mCherry::his-11] II; qyIs225[cdh-3p:: mCherry::moeABD] V; qyIs7[laminin::GFP] X. | |
| Strain, strain background (C. elegans) | DQM1115 | This paper | unc-37(miz36[unc-37p::UNC-37::AID::BFP]) I; csh128[rpl-28p::TIR1::P2A::mCherry::his-11] II; qyIs225[cdh-3p:: mCherry::moeABD] V; qyIs7[laminin::GFP] X. | |
| Strain, strain background (C. elegans) | DQM1127 | This paper | nhr-67(syb509[nhr-67p::NHR-67::GFP]) IV; stIs11476[nhr-67p::NHR-67::H1-wCherry+unc-119(+)]. | |
| Strain, strain background (C. elegans) | DQM1129 | This paper | bmd143[hsp::HLH-2::2xBFP] I; nhr-67(syb509[nhr-67p::NHR-67::GFP]) IV. | |
| Strain, strain background (C. elegans) | DQM1135 | This paper | qyIs227[cdh-3p::mCherry::moeABD] I; lam-2(qy20[lam-2p::LAM-2::mNeonGreen]) X; lag-2(bmd202[lag-2p::LAG-2::P2A::H2B::mTurquoise2^lox511^ 2xHA]) V; lag-1(devKi208[lag-1::mNeonGreen]) IV. | |
| Strain, strain background (C. elegans) | JK3791 | Phillips et al., 2007 | qIs95[sys-1p::Venus::SYS-1+pttx-3::DsRed] | |
| Strain, strain background (C. elegans) | NK1034 | Matus et al., 2015 | qyIs225[cdh-3p::mCherry::moeABD] V; qyIs7[laminin::GFP] X. | |
| Strain, strain background (C. elegans) | PHX509 | Medwig-Kinney et al., 2020 | nhr-67(syb509[nhr-67p::NHR-67::GFP]) IV. | |
| Strain, strain background (C. elegans) | PS5332 | Green et al., 2008 | syIs187[pes-10::7XTCF-mCherry-let-858(3’UTR)+unc-119(+)] | |
| Strain, strain background (C. elegans) | RW11476 | Gerstein et al., 2010 | unc-119(tm4063) III; stIs11476[nhr-67::H1-wCherry+unc-119(+)]. | |
| Strain, strain background (C. elegans) | SV2114 | van der Horst et al., 2019 | pop-1(he335[eGFP::loxP::pop-1]) I. | |
| Strain, strain background (C. elegans) | TV27467 | This paper | nhr-67(wy1632[nhr-67p::NHR-67::mNeonGreen::AID*::3xFLAG]) IV. | |
| Strain, strain background (C. elegans) | TV27468 | This paper | nhr-67(wy1633[nhr-67p::NHR-67::mScarlet-I::AID*::3xFLAG]) IV. | |
| Recombinant DNA reagent | Plasmid: pTNM087 | This paper | LSY-22 sgRNA plasmid (AAACGAAGTGGATCAGCCAG) | |
| Recombinant DNA reagent | Plasmid: pTNM088 | This paper | LSY-22^SEC^TagRFP-T::AID repair plasmid | |
| Recombinant DNA reagent | Plasmid: pTNM140 | This paper | LSY-22^SEC^mNeonGreen::AID repair plasmid | |
| Chemical compound, drug | 1-Naphthaleneacetic acid, potassium salt (K-NAA) | PhytoTech Labs | N610 | |
| Chemical compound, drug | Hygromycin B | Omega Scientific, Inc. | HG-80 | |
| Chemical compound, drug | Levamisole hydrochloride | Sigma-Aldrich | 31742 | |
| Chemical compound, drug | Sodium azide | Sigma-Aldrich | S2002 | |
| Software, algorithm | Adobe Illustrator | Adobe | Version 26.0.2 | |
| Software, algorithm | Alpha Fold | Jumper et al., 2021; Varadi et al., 2022 | Version 2 | |
| Software, algorithm | ApE – A Plasmid Editor | Wayne Davis | Version 2.0.61 | |
| Software, algorithm | Fiji/ImageJ | Schindelin et al., 2012 | Version 2.0.0-rc-69/1.53e | |
| Software, algorithm | ggplot2 | Tidyverse | Version 3.3.5 | |
| Software, algorithm | Exon-Intron Graphic Maker | Nikhil Bhatla | Version 4 | |
| Software, algorithm | JACoP (Just Another Colocalization Plugin) | Bolte and Cordelières, 2006 | Version 2.1.1 | |
| Software, algorithm | Metamorph | Molecular Devices | Version 7.10.3.279 | |
| Software, algorithm | Rstudio | R | Version 1.4.1717 |
Additional files
-
MDAR checklist
- https://cdn.elifesciences.org/articles/84355/elife-84355-mdarchecklist1-v1.pdf
-
Supplementary file 1
Sequences used in this study.
- https://cdn.elifesciences.org/articles/84355/elife-84355-supp1-v1.xlsx






