The effects of caloric restriction on adipose tissue and metabolic health are sex- and age-dependent
Figures
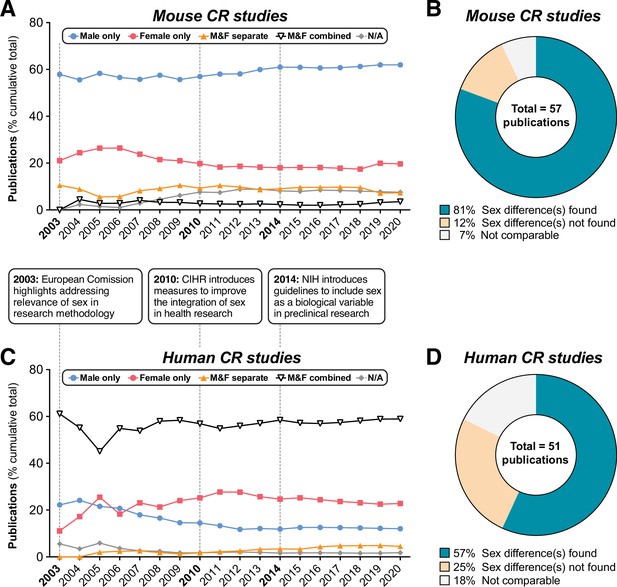
Summary of sex differences in mouse and human CR research.
PubMed was searched using MeSH terms to identify research articles that studied caloric restriction in vivo in mice (A,B) or humans (C,D), published between 2003 and 2021. Search results were then classified into the following groups depending on the sexes included in each study: ‘Male only’=male subjects used exclusively; ‘Female only’=female subjects used exclusively; ‘Male & Female separate’=male and female subjects used with data reported respective to each sex, allowing sex differences to be addressed; ‘Male & female combined’=male and female subjects used with data from both sexes combined either in part or in full; ‘N/A’=no sex data available. (A,C) Cumulative publications for studies within each group. The boxes between (A) and (C) highlight the dates of funders’ policies highlighting the importance of sex as a biological variable. (B,D) Pie charts of those studies in the ‘Male & Female separate’ group that considered sex a biological variable, and the proportion of these that did or did not detect sex differences. Source data are provided as a Source Data file. See also Figure 1—figure supplement 1.
-
Figure 1—source data 1
Literature search to identify sex differences in mouse and human CR research.
Values for the data presented in Figure 1A–D are shown in the file named ‘Figure 1_Source_Data.xlsx’. Raw data for the literature searches for mice and humans are contained in the files named ‘Literature_Search_Mice.xlsx’ and ‘Literature_Search_Humans.xlsx’.
- https://cdn.elifesciences.org/articles/88080/elife-88080-fig1-data1-v2.zip
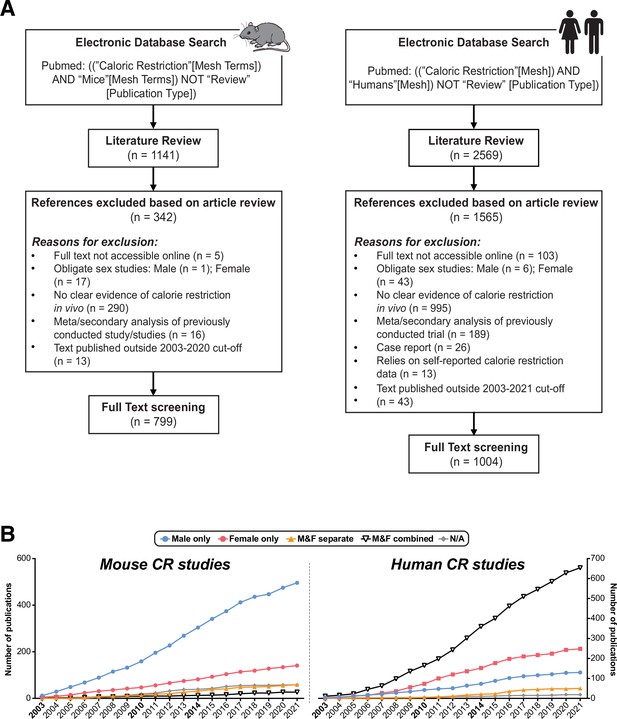
Methods used and additional outcomes relating to the comprehensive review of the CR literature.
The NIH PubMed database was searched using MeSH terms to identify research articles that studied CR in vivo in mice or humans, published between 2003 and 2021. (A) Method of database search presented as a flow diagram, including search terms, reasons for exclusion, and number of results in each category. (B) Percentage of mouse or human publications for each year, with publications classified into each group as described for Figure 1. Source data for (B) are provided as a Source Data file.
-
Figure 1—figure supplement 1—source data 1
Additional outcomes relating to the comprehensive review of the CR literature.
Values for the data presented in Figure 1—figure supplement 1B are shown in the file named ‘Figure 1-figure supplement 1_Source_Data.xlsx’.
- https://cdn.elifesciences.org/articles/88080/elife-88080-fig1-figsupp1-data1-v2.zip
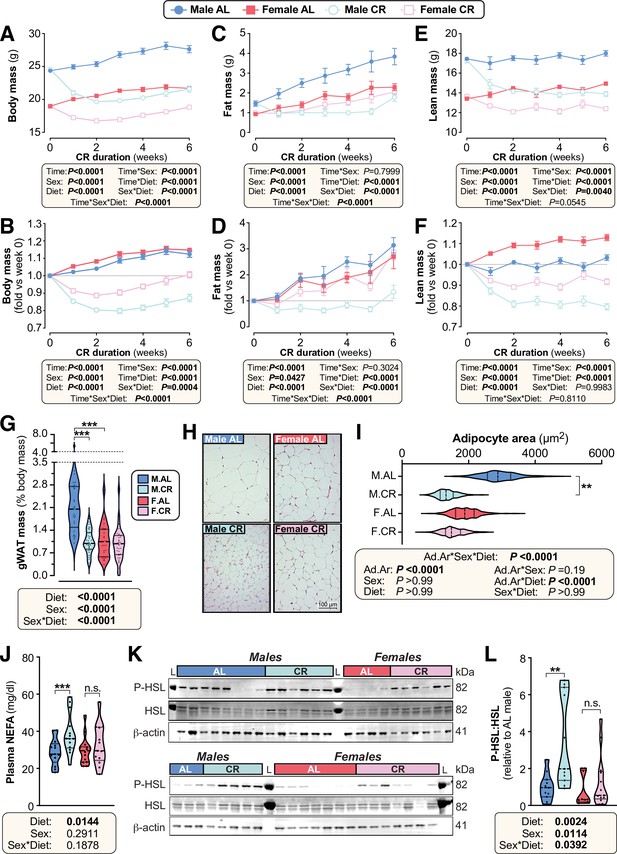
Female mice resist weight loss, fat loss, and lipolysis during CR.
Male and female mice on a C57BL/6NCrl or C57BL/6 J background were fed ad libitum (AL) or a 30% CR diet from 9 to 15 weeks of age (0–6 weeks of CR). (A–F) Each week mice were weighed (A,B) and body composition was determined by TD-NMR (C–F). Body mass, fat mass, and lean mass are shown as absolute masses (A,C,E) or fold-change relative to baseline (B,D,F). (G) The mass of gWAT (gonadal WAT) was recorded at necropsy and is shown as % body mass. (H,I) Micrographs of H&E-stained sections of gWAT (H) were used for histomorphometric analysis of adipocyte area (I); in (H), scale bar = 100 µm. (J) Plasma was sampled at 15 weeks of age and the concentration of non-esterified fatty acids (NEFA) was assayed. (K,L) At 10 weeks of age, during the period of maximal weight and fat loss, a separate cohort of mice were culled and iWAT was collected for analysis of the indicated proteins by Western blotting. Fluorescent Western blots (K) were quantified using LICOR software (L); L=protein ladder. Data in (H) and (K) show representative micrographs (H) and immunoblots (K). Data in (A–F) are shown as mean ± SEM of the following numbers of mice per group: male AL, n=42; female AL, n=43; male CR, n=44; female CR, n=52. Data in (G), (J) and (L) are shown as violin plots of the following numbers of mice per group: male AL, n=29(G), 13 (J) or 11 (L); female AL, n=28(G), 14 (J) or 10 (L); male CR, n=33 (G), 14 (J) or 11 (L); female CR, n=34 (G) or 13 (J,L). Data in (H–I) are shown as representative micrographs or violin plots from 5 (male AL) or 6 (female AL, male CR, female CR) mice per group. For (A–F), significant effects of diet, sex and/or time, and interactions thereof, were determined by three-way ANOVA or a mixed-effects model. For (G), (J) and (L), significant effects of diet and/or sex were determined by two-way ANOVA with Tukey’s or Šídák’s multiple comparisons tests. For (I), significant effects of diet and/or sex on adipocyte area (Ad.Ar) were determined using a mixed-effects model, while significant differences in median Ad.Ar between AL and CR mice were determined two-way ANOVA with Šídák’s multiple comparisons test. p Values from ANOVA or mixed models are shown beneath the graphs, as indicated. For (G), (I), (J) and (L), significant differences between comparable groups are indicated by * (p<0.05), ** (p<0.01), or *** (p<0.001). Source data are provided as a Source Data file. See also Figure 2—figure supplements 1–4.
-
Figure 2—source data 1
Female mice resist weight loss, fat loss, and lipolysis during CR.
Values for the data presented in Figure 2A–G1–J and L are shown in the file named ‘Figure 2_Source_Data.xlsx’. Raw uncropped images of immunoblots in Figure 2K are shown in the files named ‘Figure 2 blots lower beta-actin.tif’, ‘Figure 2 blots lower HSL.png’, ‘Figure 2 blots lower P-HSL.png’, ‘Figure 2 blots upper beta-actin.tif’, ‘Figure 2 blots upper HSL.png’, and ‘Figure 2 blots upper P-HSL.png’. Annotated images of uncropped immunoblots from Figure 2K are shown in the file named ‘Figure_2 K_Uncropped_blots.eps’.
- https://cdn.elifesciences.org/articles/88080/elife-88080-fig2-data1-v2.zip
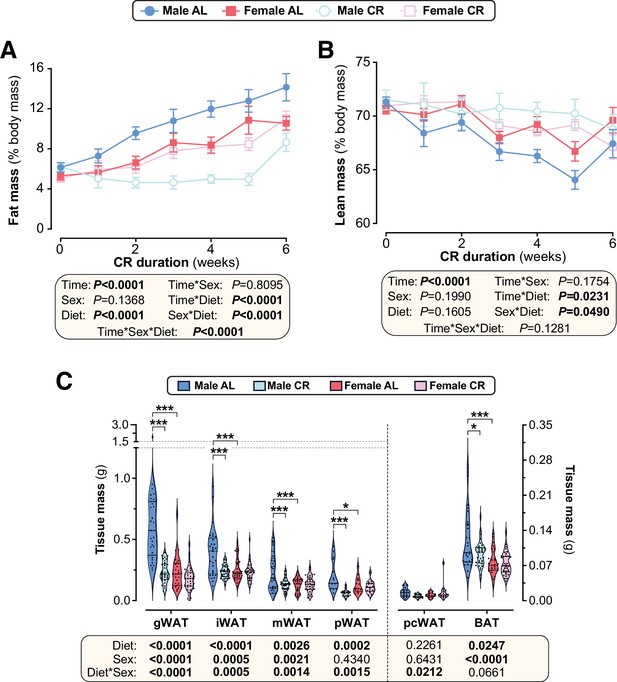
The effect of CR on percent fat and lean mass differs between male and female mice.
Male and female mice were fed AL or CR diets and body composition was assessed weekly, as described for Figure 2. (A,B) Fat mass (A) and lean mass (B) were determined by TD-NMR and calculated as % body mass. (C) Masses of iWAT (inguinal WAT), mWAT (mesenteric WAT), pWAT (perirenal WAT), pcWAT (pericardial WAT) and BAT (brown adipose tissue) were recorded at necropsy. Data in (A–B) are shown as mean ± SEM of the following numbers of mice per group: male AL, n=42; female AL, n=43; male CR, n=44; female CR, n=52. Data in (C) are shown as violin plots of the following numbers of mice per group and tissue type: male AL, n=29 (iWAT, mWAT, BAT), 15 (pWAT) or 14 (pcWAT); female AL, n=28 (iWAT, mWAT, BAT), 14 (pWAT) or 13 (pcWAT); male CR, n=33 (iWAT, mWAT, BAT), 16 (pWAT) or 15 (pcWAT); female CR, n=34 (iWAT, mWAT, BAT), 15 (pWAT), or 14 (pcWAT). For (A–B), significant effects of diet, sex and/or duration were determined by three-way ANOVA (A) or a mixed-effects model (B). For (C), significant effects of diet and/or sex within each tissue were determined by two-way ANOVA with Tukey’s multiple comparisons tests; overall ANOVA p values are shown beneath each graph. Source data are provided as a Source Data file.
-
Figure 2—figure supplement 1—source data 1
The effect of CR on percent fat and lean mass differs between male and female mice.
Values for the data presented in Figure 2—figure supplement 1A–C are shown in the file named ‘Figure 2-figure supplement 1_Source_Data.xlsx’.
- https://cdn.elifesciences.org/articles/88080/elife-88080-fig2-figsupp1-data1-v2.zip
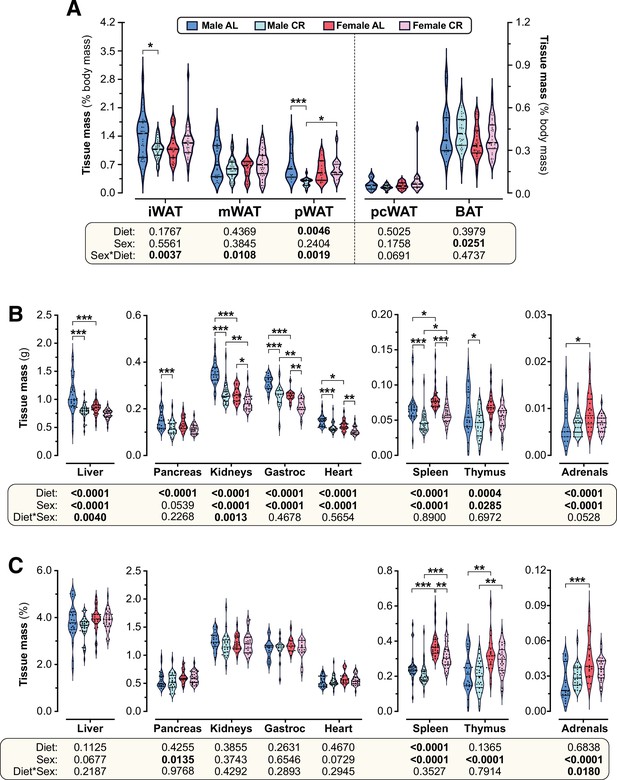
CR and sex effects on the masses of adipose and non-adipose tissues.
Male and female mice were fed AL or CR diets, as described for Figure 2. After six weeks of CR (15 weeks of age) tissues were sampled and weighed. (A–C) Violin plots, overlaid with individual data points, showing the % body mass (A,C) or absolute mass (B) of each tissue; Gastroc = gastrocnemius muscle. For (A), the numbers of mice per group and tissue type are as reported for Figure 2G and Figure 2—figure supplement 1C. For (B) and (C), the following numbers of mice were used for each group and tissue type: male AL, n=29 (liver, spleen), 23 (pancreas, kidneys, heart, thymus), 22 (adrenals) or 16 (gastroc.); female AL, n=33 (liver, spleen), 27 (pancreas, kidneys, heart, thymus, adrenals) or 16 (gastroc.); male CR, n=28 (liver, spleen), 22 (pancreas, kidneys, heart, thymus, adrenals) or 14 (gastroc.); female CR, n=34 (liver, spleen), 28 (pancreas, kidneys, heart, thymus, adrenals) or 15 (gastroc.). For each tissue, significant effects of diet and/or sex were determined by two-way ANOVA with Tukey’s multiple comparisons tests and are reported as described for Figure 2. Source data are provided as a Source Data file.
-
Figure 2—figure supplement 2—source data 1
CR and sex effects on the masses of adipose and non-adipose tissues.
Values for the data presented in Figure 2—figure supplement 2A–C are shown in the file named ‘Figure 2-figure supplement 2_Source_Data.xlsx’. This file also includes data for gonad masses and gut length, which are not included in the figures.
- https://cdn.elifesciences.org/articles/88080/elife-88080-fig2-figsupp2-data1-v2.zip
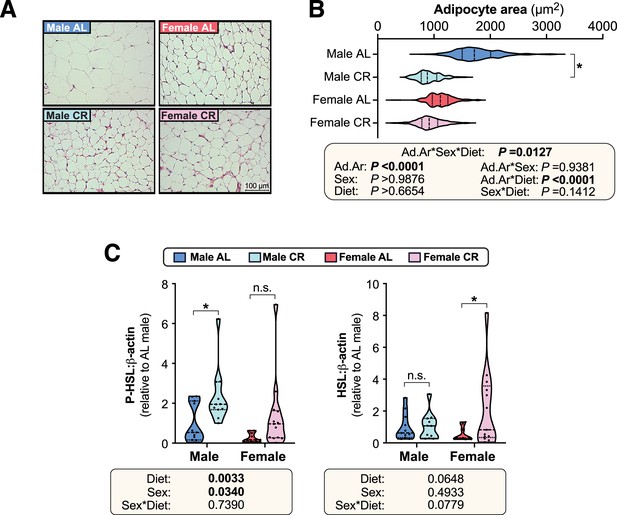
CR decreases adipocyte size and stimulates lipolysis in male but not in female mice.
Male and female mice were fed AL or CR diets as described for Figure 2. (A,B) After six weeks of CR (15 weeks of age) mWAT was sampled and processed for histological analysis. Micrographs of H&E-stained sections of mWAT (A) were used for histomorphometric analysis of adipocyte area (B); in (A), scale bar = 100 µm. (C) At 10 weeks of age, during the period of maximal weight and fat loss, a separate cohort of mice were culled and iWAT was collected for Western Blot analysis, as described for Figure 2. Expression of phosphorylated HSL (P-HSL) and total HSL was quantified using LICOR software and are shown normalised to β-actin expression. Data in (A) show representative micrographs. Data in (B) and (C) are shown as violin plots from the following number of mice per group: male AL, n=5 (B) or 11 (C); female AL, n=6 (B) or 10 (C); male CR, n=6 (B) or 11 (C); female CR, n=6 (B) or 13 (C). For (B), significant effects of diet and/or sex on adipocyte area (Ad.Ar) were determined by three-way ANOVA, with p values shown beneath the graph. Significant differences in median Ad.Ar between AL and CR mice were further determined two-way ANOVA with Šídák’s multiple comparisons test; *=p < 0.05. For (C), significant differences were determined by two-way ANOVA with Šídák’s multiple comparisons test and are reported as described for Figure 2. Source data are provided as a Source Data file.
-
Figure 2—figure supplement 3—source data 1
CR decreases adipocyte size and stimulates lipolysis in male but not in female mice.
Values for the data presented in Figure 2—figure supplement 3B–C are shown in the file named ‘Figure 2-figure supplement 3_Source_Data.xlsx’.
- https://cdn.elifesciences.org/articles/88080/elife-88080-fig2-figsupp3-data1-v2.zip
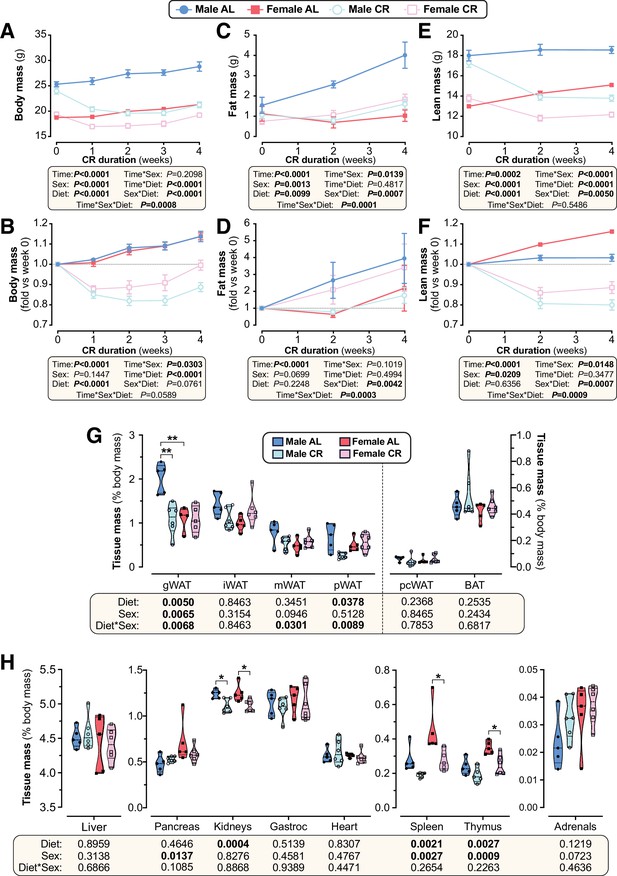
Sex differences in the effects of CR on body mass and composition persist when CR mice are fed in the evening.
Male and female C57BL6/NCrl mice were single housed and fed AL or CR diet (70% of daily AL intake) from 9 to 13 weeks of age, with CR mice receiving their daily diet ration between 18:00 and 19:00. (A–F) Each week mice were weighed (A,B) and body composition was determined by TD-NMR (C–F). Body mass, fat mass and lean mass are shown as absolute masses (A,C,E) or fold-change relative to baseline (B,D,F). (G,H) Masses of the indicated tissues were recorded at necropsy and are shown as % body mass. Data in (A–H) represent 5 (male AL, female AL) or 6 (male CR, female CR) mice per group and are shown as mean ± SEM (A–F) or as truncated volcano plots overlaid with individual data points (G,H). For (A–F), significant effects of diet, sex or time, and interactions thereof, were determined by three-way ANOVA. For (G–H), significant effects of sex, diet, and sex-diet interaction were assessed using two-way ANOVA with Tukey’s multiple comparisons test. Overall p values for each variable, and their interactions, are shown beneath each graph. For (G–H), statistically significant differences between comparable groups are indicated by ** (p<0.01). Source data are provided as a Source Data file.
-
Figure 2—figure supplement 4—source data 1
Sex differences in the effects of CR on body mass and composition persist when CR mice are fed in the evening.
Values for the data presented in Figure 2—figure supplement 4A–H are shown in the file named ‘Figure 2-figure supplement 4_Source_Data.xlsx’.
- https://cdn.elifesciences.org/articles/88080/elife-88080-fig2-figsupp4-data1-v2.zip
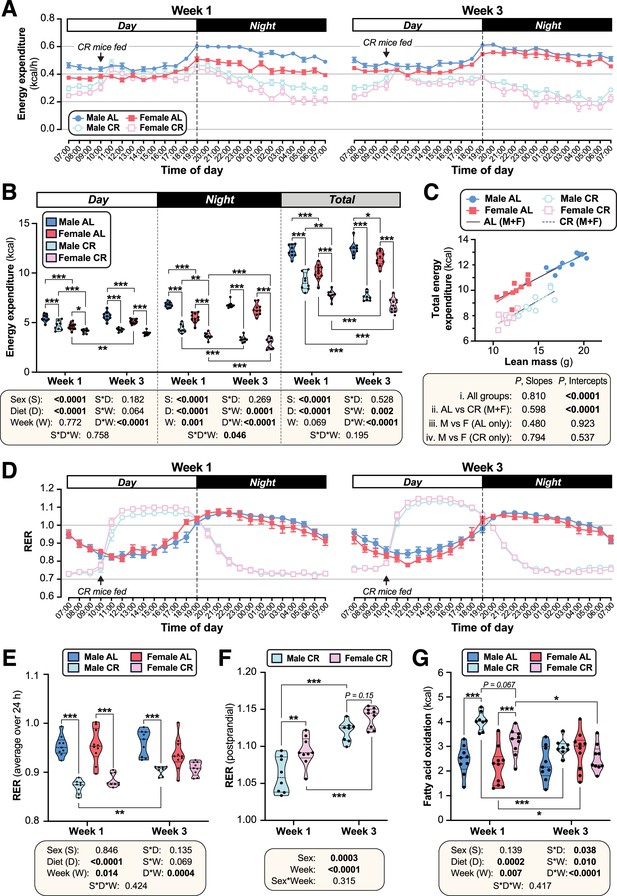
CR decreases energy expenditure and stimulates postprandial lipogenesis more in female than in male mice.
Male and female mice were fed AL or CR diets, as described for Figure 2. In week 1 and week 3 after beginning AL or CR diets, mice were housed for 4 days in Promethion CORE System cages for indirect calorimetry. Energy expenditure (A–C) and respiratory exchange ratio (RER; D–F) was recorded every minute throughout the 4 days. (A,D) Average energy expenditure (A) (kcal) or RER (D) per hour over the 24 hr light (Day) and dark (Night) periods, based on the average for days 2–4 of Promethion housing, for Week 1 (left) and Week 3 (right). (B) Overall energy expenditure (kcal) during the day, night, or day +night (Total) for Week 1 and Week 3. (C) Linear regression of lean mass vs total energy expenditure (kcal/24 h) during Week 1. (E) Average total RER (day +night) in Weeks 1 and 3. (F) Average RER during the postprandial period, from 12.00 to 17.00, for CR mice. (G) Absolute FA oxidation was determined based on energy expenditure and RER as described (Bruss et al., 2010). Data are from 10 (female AL), 9 (female CR, male AL) or 8 (male CR) mice per group. In (A) and (D), data are shown as mean ± SEM. In (B) and (E–G), data are shown as violin plots overlaid with individual data points; within each time period (day, night, or total), significant effects of sex, diet, week, and interactions thereof, were determined by three-way (B,E,G) or two-way ANOVA (F), with p values shown beneath each graph. Statistically significant differences between comparable groups were further assessed by Šídák’s (B,E,G) or Tukey’s (F) multiple comparisons tests and are indicated by * (p<0.05), ** (p<0.01), or *** (p<0.001). For linear regression in (C), ANCOVA was used to test if the relationship between lean mass and total energy expenditure differs significantly across all of the individual diet-sex groups (i. All mice); between AL and CR mice, irrespective of sex (ii. AL vs CR (M+F)); and between males and females fed AL diet (iii) or CR diet (iv) only. ANCOVA p values for differences in slope and intercept are reported beneath the graph. See also Figure 3—figure supplements 1–2.
-
Figure 3—source data 1
CR decreases energy expenditure and stimulates postprandial lipogenesis more in female than in male mice.
Values for the data presented in Figure 3A–G are shown in the file named ‘Figure 3_Source_Data.xlsx’.
- https://cdn.elifesciences.org/articles/88080/elife-88080-fig3-data1-v2.zip
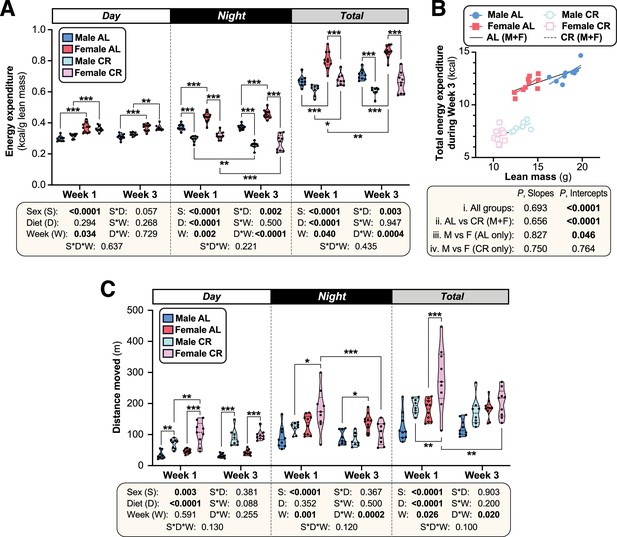
Effects of CR on energy expenditure and physical activity.
Male and female AL and CR mice underwent indirect calorimetry as described for Figure 3. (A) Overall energy expenditure relative to lean mass (kcal/g) during the day, night, or day +night (Total) for Week 1 and Week 3. (B) Linear regression of lean mass vs Total energy expenditure (kcal/24 hr) during Week 3. (C) Distance moved by each mouse during the day, night, or day +night (total) for Week 1 and Week 3. Data presentation and statistical analysis are as described for Figure 3. Source data are provided as a Source Data file.
-
Figure 3—figure supplement 1—source data 1
Effects of CR on energy expenditure and physical activity.
Values for the data presented in Figure 3—figure supplement 1A–C are shown in the file named “Figure 3-figure supplement 1_Source_Data.xlsx”.
- https://cdn.elifesciences.org/articles/88080/elife-88080-fig3-figsupp1-data1-v2.zip
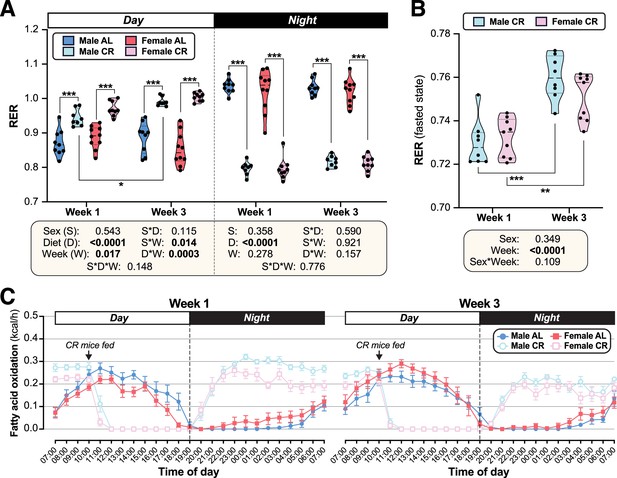
Effects of CR on Respiratory exchange ratio and FA oxidation.
Male and female AL and CR mice underwent indirect calorimetry as described for Figure 3. (A) Average RER during the day or night for Weeks 1 and 3. (B) Average RER during the fasted period, from 04.00 to 09.00, for Weeks 1 and 3. (C) Absolute FA oxidation was determined from energy expenditure and RER (Bruss et al., 2010) and is shown as kcal/h over the 24 hr day night periods, based on the average for days 2–4 of Promethion housing, for Week 1 (left) and Week 3 (right). Data presentation and statistical analysis are as described for Figure 3. Source data are provided as a Source Data file.
-
Figure 3—figure supplement 2—source data 1
Effects of CR on Respiratory exchange ratio and FA oxidation.
Values for the data presented in Figure 3—figure supplement 2A–C are shown in the file named ‘Figure 3-figure supplement 2_Source_Data.xlsx’.
- https://cdn.elifesciences.org/articles/88080/elife-88080-fig3-figsupp2-data1-v2.zip
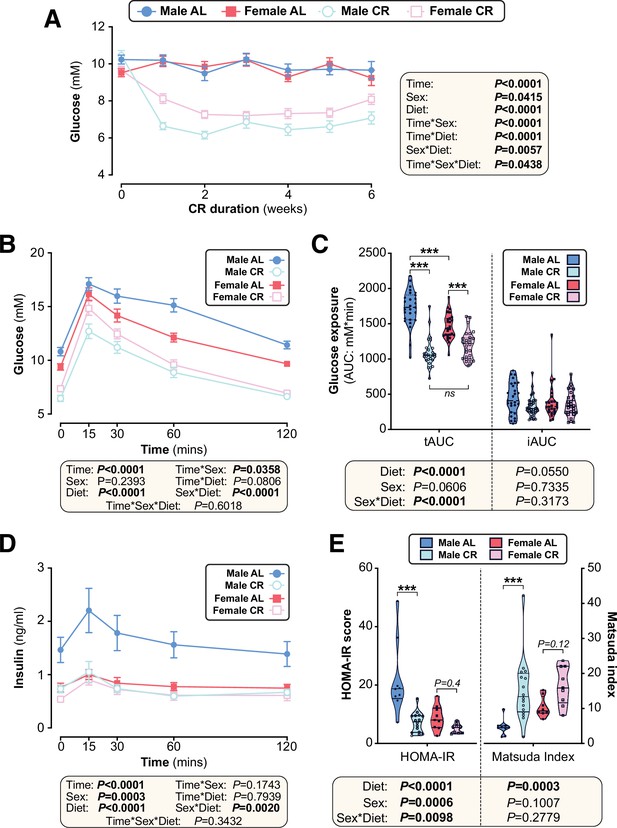
The effects of CR on glucose homeostasis differ between young male and female mice.
Male and female C57BL6/NCrl mice were fed AL or CR diet from 9 to 15 weeks of age, as described for Figure 2. (A) Random-fed blood glucose was recorded each week. (B–D) At 13 weeks of age, mice underwent an oral glucose tolerance test (OGTT). (B) Blood glucose readings during the OGTT. (C) Area under the curve (AUC) during the OGTT was determined relative to 0 mmol/L (total AUC: tAUC) and relative to baseline (incremental AUC: iAUC). (D) Glucose-stimulated insulin secretion in mice during OGTT was assessed using an insulin ELISA. (E) HOMA-IR and Matsuda indices of mice calculated from glucose and insulin concentrations during the OGTT. Data in (A), (B), and (D) are presented as mean ± SEM. Data in (C) and (E) are presented as violin plots overlaid with individual data points. For each group and timepoint, the following numbers of mice were used: (A): male AL, n=42 (Wk 0), 36 (Wk2, Wk 2), 34 (Wk 3), 29 (Wk 4), 26 (Wk 5), or 22 (Wk 6); female AL, n=43 (Wk 0), 28 (Wk 1), 35 (Wk 2), 31 (Wk 3), 27 (Wk 4), 26 (Wk 5), or 21 (Wk 6); male CR, n=44 (Wk 0), 40 (Wk 1), 38 (Wk 2), 35 (Wk 3), 31 (Wk 4), 29 (Wk 5), or 26 (Wk 6); female CR, n=51 (Wk 0), 44 (Wk 1, Wk 2), 41 (Wk 4), 35 (Wk 4), 33 (Wk 5), or 27 (Wk 6). (B): male AL, n=27 (T0, T15, T30, T60, T120); female AL, n=26 (T15, T30, T120), or 25 (T0, T60); male CR, n=27 (T0, T15, T30, T60, T120); female CR, n=28 (T0, T15, T30, T60, T120). (C): male AL, n=27; female AL, n=26; male CR, n=27; female CR, n=28. (D): male AL, n=9 (T0, T15, T30, T60, T120); female AL, n=11 (T0, T15), 10 (T30, T60), or 8 (T120); male CR, n=17 (T0), 16 (T30), 15 (T60), 13 (T15), or 11 (T120); female CR, n=11 (T15, T30, T60,), or 10 (T0, T120). (E): male AL, n=9; female AL, n=9; male CR, n=16; female CR, n=9. In (A), (B), and (D), significant effects of diet, sex and/or time, and interactions thereof, were determined using a mixed-effects model. In (C) and (E), significant effects of sex, diet, and sex-diet interaction for tAUC, iAUC, HOMA-IR, and Matsuda Index were assessed using two-way ANOVA with Tukey’s multiple comparisons test. Overall p values for each variable, and their interactions, are shown with each graph. For (C) and (E), statistically significant differences between comparable groups are indicated by *** (p<0.001) or with p values shown. Source data are provided as a Source Data file. See also Figure 4—figure supplement 1.
-
Figure 4—source data 1
The effects of CR on glucose homeostasis differ between young male and female mice.
Values for the data presented in Figure 4A–E are shown in the file named ‘Figure 4_Source_Data.xlsx’.
- https://cdn.elifesciences.org/articles/88080/elife-88080-fig4-data1-v2.zip
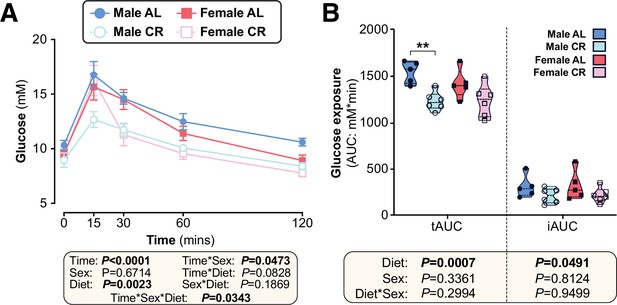
Sex differences in the effects of CR on glucose homeostasis persist when CR mice are fed in the evening.
Male and female C57BL6/NCrl mice were fed AL or CR diet from 9 to 15 weeks of age, as described for Figure 2—figure supplement 4. (A,B) At 13 weeks of age mice underwent OGTTs. Blood glucose was recorded at each timepoint (A) and the tAUC and iAUC was determined (B). Data represent 5 (male AL, female AL) or 6 (male CR, female CR) mice per group and are shown as mean ± SEM (A) or as truncated volcano plots overlaid with individual data points (B). In (A), significant effects of diet, sex or time, and interactions thereof, were determined by three-way ANOVA. In (B), significant effects of sex, diet, and sex-diet interaction were assessed using two-way ANOVA with Tukey’s multiple comparisons test. Overall p values for each variable, and their interactions, are shown beneath each graph. For (B), statistically significant differences between comparable groups are indicated by ** (p<0.01). Source data are provided as a Source Data file.
-
Figure 4—figure supplement 1—source data 1
Sex differences in the effects of CR on glucose homeostasis persist when CR mice are fed in the evening.
Values for the data presented in Figure 4—figure supplement 1A–B are shown in the file named ‘Figure 4-figure supplement 1_Source_Data.xlsx’.
- https://cdn.elifesciences.org/articles/88080/elife-88080-fig4-figsupp1-data1-v2.zip
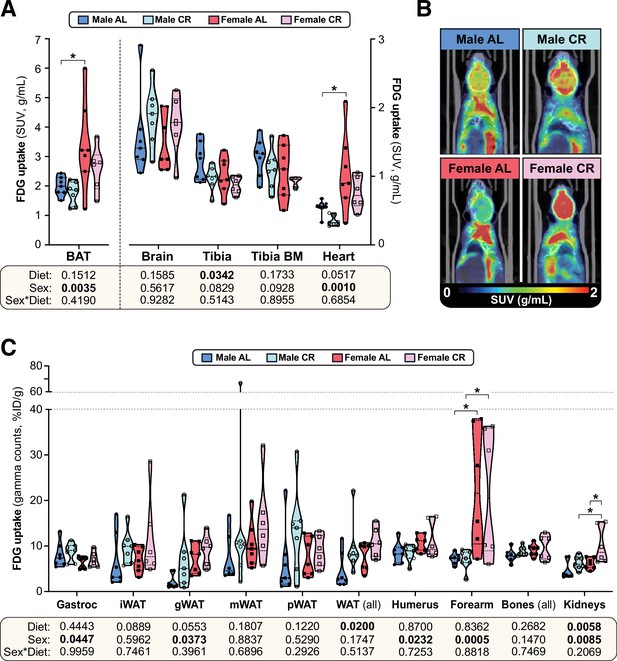
The effects of CR on glucose uptake do not differ between male and female mice.
Male and female C57BL6/NCrl mice were fed AL or CR diet from 9 to 15 weeks of age, as described for Figure 2. At 15 weeks of age, glucose uptake was assessed by PET/CT with 18F-fluorodeoxyglucose (18F-FDG). (A–C) 18F-FDG uptake in the indicated tissues was determined as SUVs from PET/CT scans (A) or as % injected dose per gram of tissue (%ID/g) from gamma counting of dissected whole tissues (C). (B) PET/CT images confirm that 18F-FDG uptake in interscapular BAT is greater in females than in males, regardless of diet. Data are shown as violin plots (A,C) or representative images (B) of 7 (male AL, female AL, male CR) or 6 (female CR) mice per group. For each tissue in (A) and (C), significant effects of sex, diet, and sex-diet interaction were assessed by two-way ANOVA; overall p values for each variable, and their interactions, are shown beneath each graph. Significant differences between each group, as determined using Tukey’s multiple comparisons test, are indicated by * (p<0.05). Source data are provided as a Source Data file. See also Figure 5—figure supplement 1.
-
Figure 5—source data 1
The effects of CR on glucose uptake do not differ between male and female mice.
Values for the data presented in Figure 5A and C are shown in the file named ‘Figure 5_Source_Data.xlsx’.
- https://cdn.elifesciences.org/articles/88080/elife-88080-fig5-data1-v2.zip
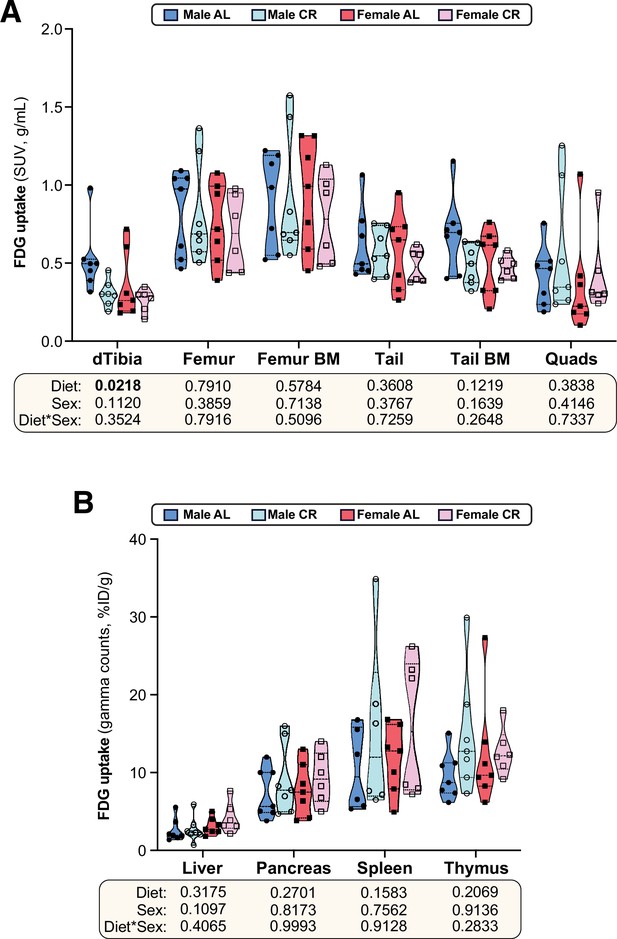
The effects of CR on glucose uptake in peripheral tissues.
Male and female C57BL6/NCrl mice were fed AL or CR diet and glucose uptake was assessed by 18F-FDG PET/CT, as described for Figure 5. (A,B)18F-FDG uptake in the indicated tissues was determined by PMOD analysis of PET/CT scans (A) or gamma counting of dissected whole tissues (B). Data in (A) and (B) are presented as truncated violin plots, overlaid with individual data points, from 7 (male AL, female AL, male CR) or 6 (female CR) mice per group; in (B), data for spleen are from 6 mice per group only. Statistical significance was determined as described for Figure 5. Source data are provided as a Source Data file.
-
Figure 5—figure supplement 1—source data 1
The effects of CR on glucose uptake in peripheral tissues.
Values for the data presented in Figure 5—figure supplement 1A–B are shown in the file named ‘Figure 5-figure supplement 1_Source_Data.xlsx’.
- https://cdn.elifesciences.org/articles/88080/elife-88080-fig5-figsupp1-data1-v2.zip
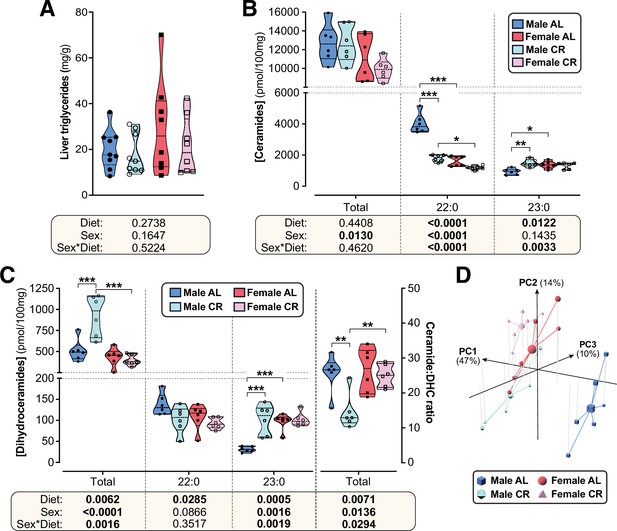
CR exerts sexually dimorphic effects on hepatic sphingolipid content.
Male and female C57BL6/NCrl mice were fed AL or CR diet from 9 to 15 weeks of age, as described for Figure 2. Livers were sampled at necropsy (15 weeks) and used to assess triglyceride, ceramide, and dihydroceramide. (A) Hepatic triglyceride content. (B,C) LC-MS analysis of total, 22:0 and 23:0 ceramides (B), dihydroceramides (C, left side) and the ceramide:DHC ratio (C, right side). (D) Principal component analysis of ceramide and dihydroceramide content based on data in (B–C) and Figure 6—figure supplement 1. PC1, PC2, and PC3 account for 47.4%, 14.1%, and 10.5% of the variance, respectively. Data in (A), (B), and (C) are presented as truncated violin plots overlaid with individual data points. Data represent the following numbers of mice per group. (A): male AL, n=9; female AL, n=8; male CR, n=9; female CR, n=8. (B–D): 6 mice per group. For (A), (B), and (C), significant effects of sex, diet, and sex-diet interaction were assessed using two-way ANOVA; overall p values for each variable, and their interactions, are shown beneath each graph. Significant differences between comparable groups were assess using Tukey’s multiple comparison test and are indicated by * (p<0.05), ** (p<0.01), or *** (p<0.001). Source data are provided as a Source Data file. See also Figure 6—figure supplement 1.
-
Figure 6—source data 1
CR exerts sexually dimorphic effects on hepatic sphingolipid content.
Values for the data presented in Figure 6A–D are shown in the file named ‘Figure 6_Source_Data.xlsx’. The code used for Principal Component Analysis in Figure 6D is in the file named ‘R code for Principal Component Analysis from ceramide data.r’.
- https://cdn.elifesciences.org/articles/88080/elife-88080-fig6-data1-v2.zip
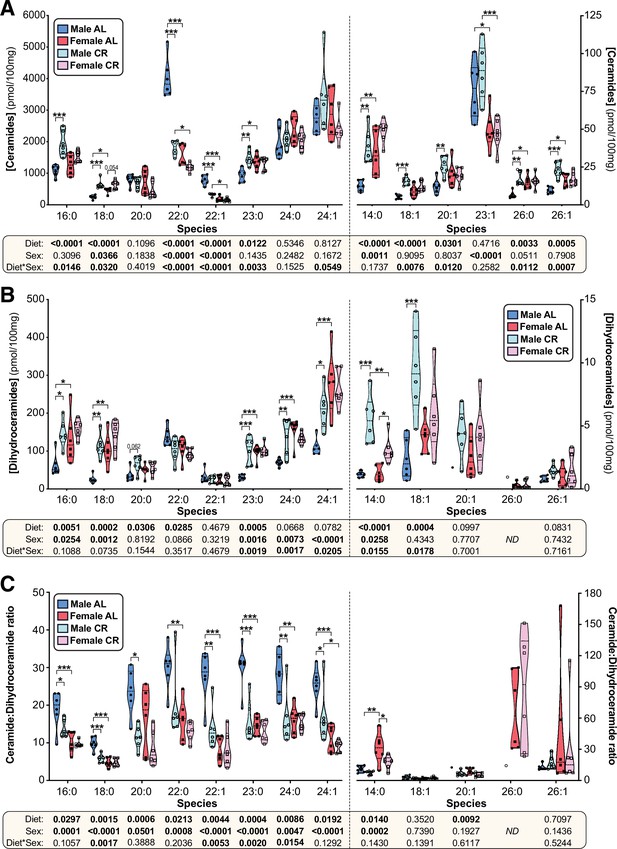
CR exerts sexually dimorphic effects on hepatic sphingolipid content.
Male and female mice were fed AL or CR diets as described for Figure 2. (A–C) At necropsy, livers were sampled for analysis of ceramide and dihydroceramide content by LC-MS. Data show the concentration of each species of ceramide (A), dihydroceramide (B), or dihydroceramide:ceramide ratio (C) and are presented as truncated violin plots showing all points. The following numbers of mice were used per group and species: male AL, n=6 (all except: DHC 20:1 & 26:0, Cer:DHC 20:1 & 26:0), 1 (DHC & Cer:DHC 20:1), or 0 (DHC & Cer:DHC 26:0); female AL, n=6; male CR, n=6 (all except DHC & Cer:DHC 26:0), or 1 (DHC & Cer:DHC 26:0); female CR, n=6. For each species, significant effects of sex, diet, and sex-diet interaction were assessed using two-way ANOVA; overall p values for each variable, and their interactions, are shown beneath each graph. Significant differences between comparable groups were assess using Tukey’s multiple comparison test and are indicated by * (p<0.05), ** (p<0.01), or *** (p<0.001). Source data are provided as a Source Data file.
-
Figure 6—figure supplement 1—source data 1
CR exerts sexually dimorphic effects on hepatic sphingolipid content.
Values for the data presented in Figure 6—figure supplement 1A–C are shown in the file named “Figure 6-figure supplement 1_Source_Data.xlsx”.
- https://cdn.elifesciences.org/articles/88080/elife-88080-fig6-figsupp1-data1-v2.zip
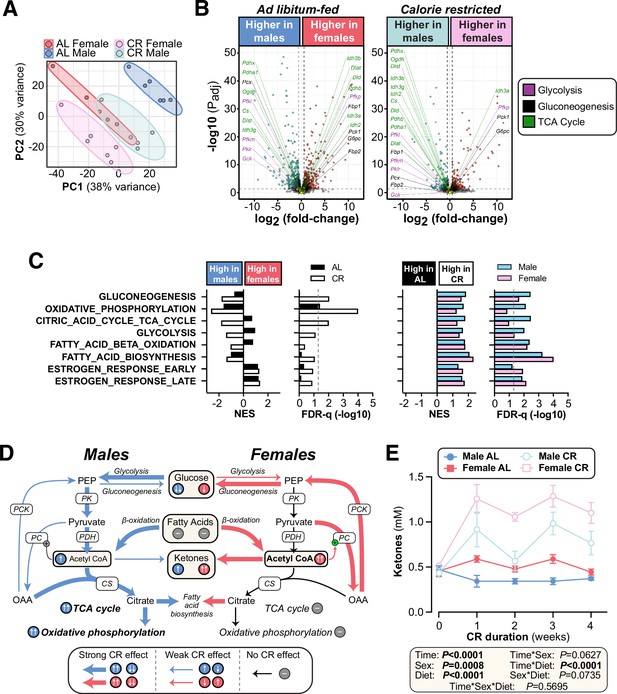
CR exerts sexually dimorphic effects on hepatic gene expression and blood ketone levels.
Male and female C57BL6/NCrl mice were fed AL or CR diet from 9 to 13 weeks of age, as described for Figure 2. Blood ketone concentrations were assessed weekly. Livers were sampled at necropsy (13 weeks) and analysed by RNA-seq. (A) Principal component analysis of all samples. PC1 and PC2 account for 38% and 30% of the variance, respectively. (B) Volcano plots showing differentially expressed genes between AL males and AL females (left) or between CR males and CR females (right). Transcripts encoding key proteins in gluconeogenesis, glycolysis, or the TCA cycle are shown on each graph. The names of genes with positive fold-change values are shown on the right side and those with negative fold-change values are shown on the left side of the volcano plots; none of these genes had absolute fold-change >1.5 and Padj <0.05. (C) GSEA results for gene sets indicative of relevant pathways. Significant sex differences (within AL or CR diets) are shown on the left; significant diet differences (within males or females) are shown on the right; NES = normalised enrichment score. (D) Schema showing the proposed sex differences in liver metabolism during CR, based on the RNA-seq data. PK, pyruvate kinase; PDH, pyruvate dehydrogenase; CS, citrate synthase; PC, pyruvate carboxylase; PCK, phosphoenolpyruvate carboxykinase. (E) Blood ketone concentrations. Data represent the following numbers of mice per group: (A–C) – male AL, n=6; female AL, n=5; male CR, n=5; female CR, n=6. (E) – male AL, n=7; female AL, n=9; male CR, n=6; female CR, n=7. For (A) and (B), R script are provided as a Source Data file. For (C), gene set collection files are prepared with the 8 gene sets shown for GSEA. For (E), significant effects of sex, diet, and sex-diet interaction were assessed using three-way ANOVA; overall p values for each variable, and their interactions, are shown beneath each graph. Source data are provided as a Source Data file. See also Figure 7—figure supplement 1.
-
Figure 7—source data 1
CR exerts sexually dimorphic effects on hepatic gene expression and blood ketone levels.
Values for the data presented in Figure 7A, C and E are shown in the file named ‘Figure 7_Source_Data.xlsx. The code used for Principal Component Analysis in Figure 7A and generating volcano plots in Figure 7B is in the file named ‘R code for Principal Component Analysis and Volcano plots from RNAseq data.r’.
- https://cdn.elifesciences.org/articles/88080/elife-88080-fig7-data1-v2.zip
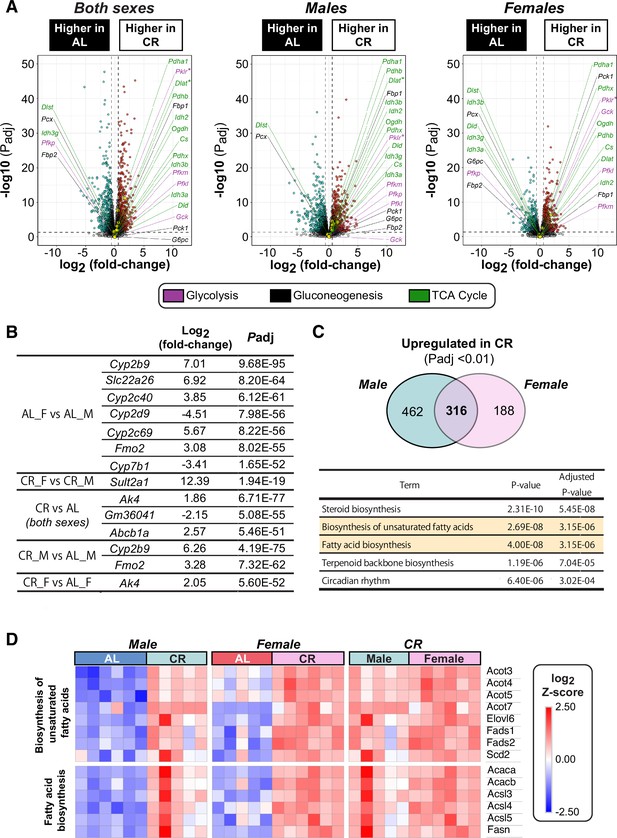
Effects of sex and diet on the hepatic transcriptome.
Male and female C57BL6/NCrl mice were fed AL or CR diet from 9 to 13 weeks of age, as described for Figure 2. Livers were sampled at necropsy (13 weeks) and analysed by RNA-seq. (A) Volcano plots showing differentially expressed genes between AL and CR mice of both sexes (left), AL males vs CR males (middle), and AL females vs CR females (right). Transcripts encoding key proteins in gluconeogenesis, glycolysis, or the TCA cycle are shown on each graph. The names of genes with positive fold-change values are shown on the right side and those with negative fold-change values are shown on the left side of the volcano plots. Genes with absolute fold-change >1.5 and Padj <0.05 are marked with an asterisk next to the gene name. (B) List of genes not shown in the volcano plots of Figure 7B and Figure 7—figure supplement 1A because their higher values of absolute fold-change and/or Padj do not fit on the scale of the axes. (C) The number of genes highly expressed in CR male compared to AL male (Padj <0.01, left circle) and CR female compared to AL female (Padj <0.01, right circle), with the overlap representing the 316 CR-induced genes common to both sexes. GO analysis (KEGG_2019_Mouse) were conducted with the overlapping gene list and the top 5 GO pathways are shown in the table. (D) A heatmap showing log2 Z-scores for genes linked to the GO terms of ‘biosynthesis of unsaturated fatty acids’ or ‘fatty acid biosynthesis’ among the 316 genes whose expression was higher in CR than in AL in both males and females (Padj <0.05). Data represent the following numbers of mice per group: male AL, n=6; female AL, n=5; male CR, n=5; female CR, n=6. For (A), R scripts are provided as a Source Data file.
-
Figure 7—figure supplement 1—source data 1
Effects of sex and diet on the hepatic transcriptome.
Values for the data presented in Figure 7—figure supplement 1D are shown in the file named ‘Figure 7—figure supplement 1_Source_Data.xlsx’. The code used to generate volcano plots in Figure 7—figure supplement 1A, and for the analyses in Figure 7—figure supplement 1B–C, is in the file named ‘R code for Principal Component Analysis and Volcano plots from RNAseq data.r’.
- https://cdn.elifesciences.org/articles/88080/elife-88080-fig7-figsupp1-data1-v2.zip
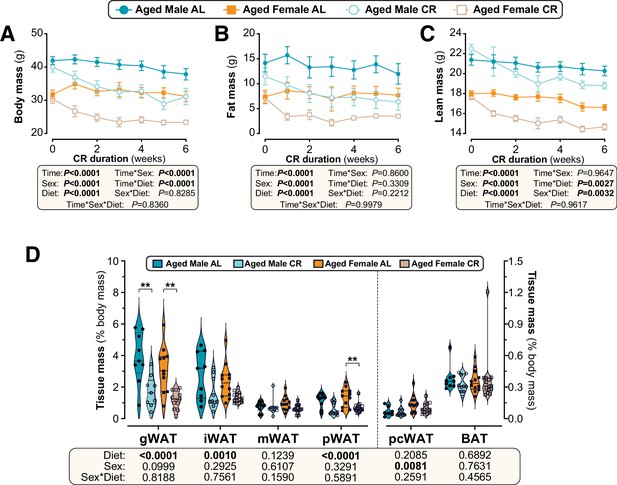
Sex differences in CR-induced weight loss and fat loss are absent when CR is initiated in aged mice.
Male and female C57BL6/NCrl mice were single housed and fed AL or a CR diet (70% of daily AL intake) from 78 to 84 weeks of age. (A–F) Each week mice were weighed (A,B) and body composition was determined by TD-NMR (C–F). Body mass, fat mass and lean mass are shown as absolute masses (A,C,E) or fold-change relative to baseline (B,D,F). (G) Masses of gWAT, iWAT, mWAT, pWAT, pcWAT, and BAT were recorded at necropsy and are shown as % body mass. Data in (A–F) are shown as mean ± SEM. Data in (G) are shown as violin plots overlaid with individual data points. For each group and timepoint, the following numbers of mice were used: (A,B): male AL, n=9 (Wk 0, Wk 2, Wk3, Wk 4), 8 (Wk 1), or 6 (Wk 5, Wk 6); female AL, n=13 (Wk 0, Wk 2), 12 (Wk 4, Wk 5), 11 (Wk 6), 9 (Wk 1), or 8 (Wk 3); male CR, n=10 (Wk 0, Wk 2–4), 9 (Wk 1), 7 (Wk 5), or 6 (Wk 6); female CR, n=14 (Wk 0, Wk 4, Wk 5), 13 (Wk 2, Wk 6), 10 (Wk 3), or 9 (Wk 1). (C–F): male AL, n=9 (Wk 0, Wk 2), 8 (Wk 3, Wk 4, Wk 6), or 7 (Wk 1, Wk 5); female AL, n=13 (Wk 0, Wk 2), 9 (Wk 4, Wk 6), 8 (Wk 5), 6 (Wk 1), or 5 (Wk 3); male CR, n=10 (Wk 0, Wk 2), 9 (Wk3-6), or 8 (Wk 1); female CR, n=14 (Wk 1, Wk 2), 11 (Wk 4–6), 7 (Wk 3), or 6 (Wk 1). (G): male AL, n=9; female AL, n=12 (iWAT, mWAT) or 11 (gWAT, pWAT); male CR, n=10 (iWAT, pWAT), or 9 (gWAT, mWAT); female CR, n=14 (mWAT, pWAT), 13 (iWAT), or 12 (gWAT). For (A–F), significant effects of diet, sex and/or time, and interactions thereof, were determined by three-way ANOVA or a mixed-effects model. For (G), significant effects of sex, diet, and sex-diet interaction were assessed using two-way ANOVA with Tukey’s multiple comparisons test. Overall p values for each variable, and their interactions, are shown beneath each graph. For (G), significant differences between comparable groups are indicated by ** (p<0.01) or *** (p<0.001). Source data are provided as a Source Data file. See also Figure 8—figure supplement 1.
-
Figure 8—source data 1
Sex differences in CR-induced weight loss and fat loss are absent when CR is initiated in aged mice.
Values for the data presented in Figure 8A–D are shown in the file named ‘Figure 8_Source_Data.xlsx’.
- https://cdn.elifesciences.org/articles/88080/elife-88080-fig8-data1-v2.zip
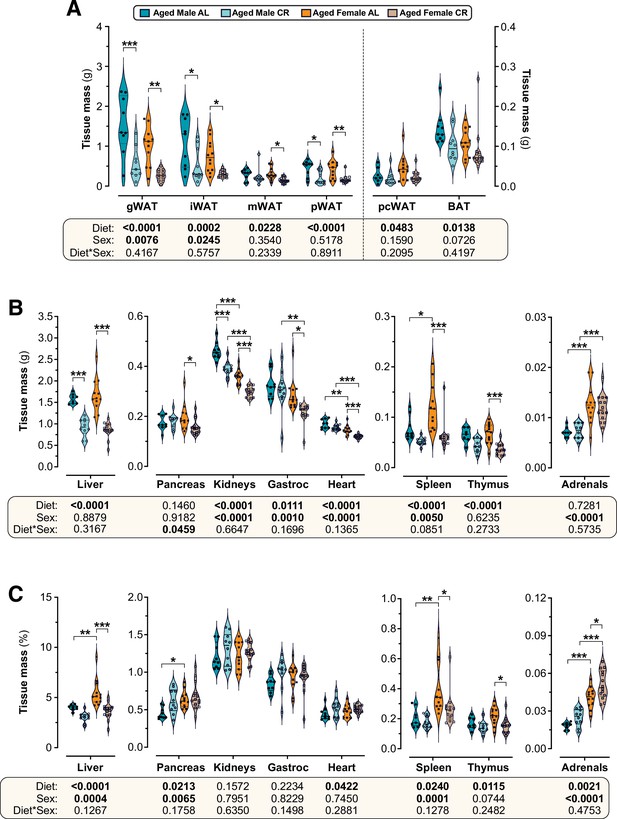
CR and sex effects on the masses of adipose and non-adipose tissues in aged mice.
Male and female mice were fed AL or CR diets from 78 to 84 weeks of age, as described for Figure 7. After six weeks of CR (84 weeks of age) tissues were sampled and weighed. (A–C) Violin plots, overlaid with individual data points, showing the absolute mass (A,B) or % body mass (C) of each tissue; Gastroc = gastrocnemius muscle. The numbers of mice per group and tissue type are as reported for Figure 7G. For each tissue, significant effects of diet and/or sex were determined by two-way ANOVA with Tukey’s multiple comparisons test and are reported as described for Figure 8. Source data are provided as a Source Data file.
-
Figure 8—figure supplement 1—source data 1
CR and sex effects on the masses of adipose and non-adipose tissues in aged mice.
Values for the data presented in Figure 8—figure supplement 1A–C are shown in the file named “Figure 8-figure supplement 1_Source_Data.xlsx”.
- https://cdn.elifesciences.org/articles/88080/elife-88080-fig8-figsupp1-data1-v2.zip
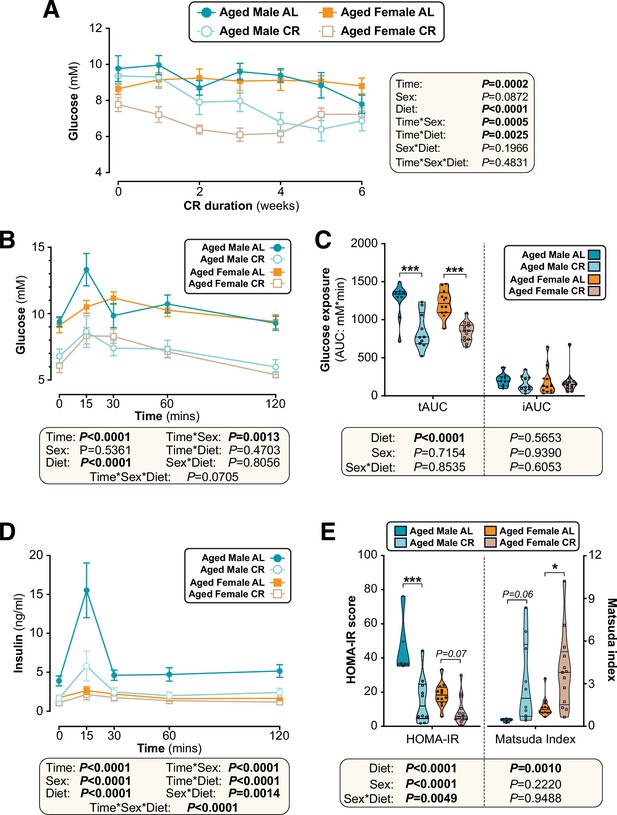
Sex differences in CR’s effects on glucose homeostasis are largely absent when CR is initiated in aged mice.
Male and female C57BL6/NCrl mice were fed AL or CR diet from 78 to 84 weeks of age, as described for Figure 7. (A) Random-fed blood glucose was recorded each week. (B–D) At 82 weeks of age mice underwent OGTTs. Blood glucose was recorded at each timepoint (B) and the tAUC and iAUC was determined (C). (D) Glucose-stimulated insulin secretion in mice during OGTT was assessed using an insulin ELISA. (E) HOMA-IR and Matsuda indices of mice calculated from glucose and insulin concentrations during the OGTT. Data in (A), (B), and (D) are presented as mean ± SEM. Data in (C) and (E) are presented as violin plots overlaid with individual data points. For each group and timepoint the following numbers of mice were used: (A): male AL, n=9 (Wk 0, Wk 2–4, Wk 6), 8 (Wk 1), or 5 (Wk 5); female AL, n=12 (Wk 0, Wk2, Wk4), 8 (Wk 1, Wk 3), or 11 (Wk 5, Wk 6); male CR, n=10 (Wk 0, Wk 2–4, Wk 6), 9 (Wk 1), or 7 (Wk 5); female CR, n=14 (Wk 0, Wk 2, Wk 4–6), 9 (Wk 1) or 10 (Wk 3). (B–E): male AL, n=9 (B–C) or 5 (D–E); female AL, n=12; male CR, n=10; female CR, n=14. In (A), (B), and (D), significant effects of diet, sex and/or time, and interactions thereof, were determined using a mixed-effects model or three-way ANOVA. In (C) and (E), significant effects of sex, diet, and sex-diet interaction for tAUC, iAUC, HOMA-IR, and Matsuda Index were assessed using two-way ANOVA with Tukey’s multiple comparisons test. Overall p values for each variable, and their interactions, are shown with each graph. For (C) and (E), statistically significant differences between comparable groups are indicated by * (p<0.05) or *** (p<0.001), or with p values shown. Source data are provided as a Source Data file.
-
Figure 9—source data 1
Sex differences in CR’s effects on glucose homeostasis are largely absent when CR is initiated in aged mice.
Values for the data presented in Figure 9A–E are shown in the file named ‘Figure 9_Source_Data.xlsx’.
- https://cdn.elifesciences.org/articles/88080/elife-88080-fig9-data1-v2.zip
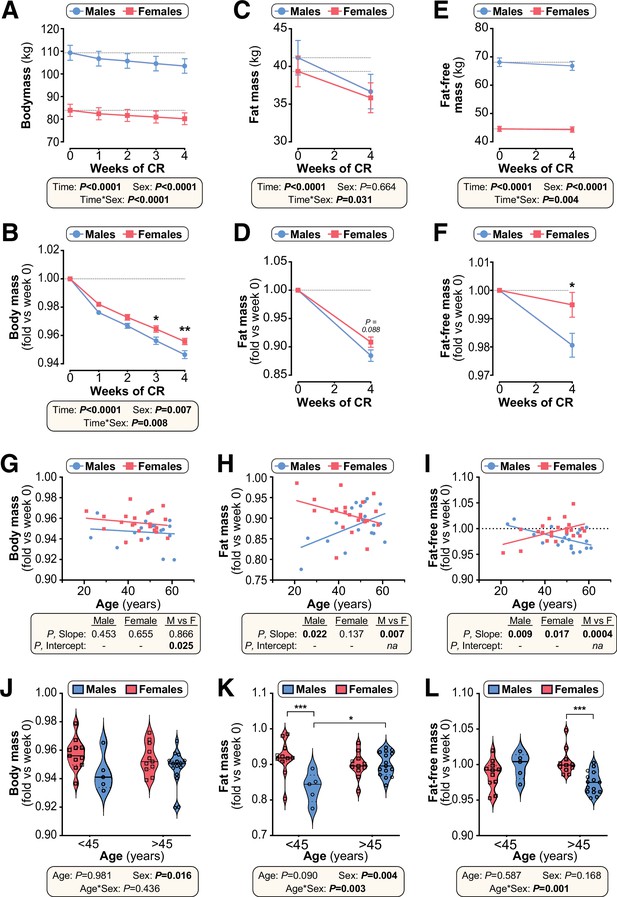
Effects of CR on body mass and body composition in humans are sex- and age-dependent.
Twenty male and 22 female volunteers participated in a weight loss study involving a 4-week dietary intervention. (A–F) Body mass was recorded each week (A,B). Fat mass (FM) and fat-free mass (FFM) were measured by air-displacement whole-body plethysmography at weeks 0 and 4 (C–F). Body mass, fat mass, and fat-free mass are shown as absolute masses (A,C,E) or fold-change relative to baseline (B,D,F). Data are presented as mean ± SEM. For (A–C) and (E), significant effects of time, sex, and time*sex interaction were assessed using two-way ANOVA. In (B), (D) and (F), significant differences between males and females at each time point were determined by Šídák’s multiple comparisons test (B) or unpaired T-test (D,F) and are indicated by * (p<0.05) or ** (p<0.01). (G–I) Simple linear regression of age vs fold-change (week 4 vs week 0) in body mass (G), fat mass (H) and fat-free mass (I). For each sex, significant associations between age and outcome (fold-change) are indicated beneath each graph as ‘P, Slope’. ANCOVA was further used to test if the age-outcome relationship differs significantly between males and females. ANCOVA results are reported beneath each graph as ‘P, Slope’ and ‘P, Intercept’ for males vs females (M vs F). In (G), similar slopes but different intercepts show that sex significantly influences weight loss, but the influence of age does not differ between the sexes. In (H,I) the slopes differ significantly, indicating that the age-outcome relationship differs between the sexes. (J–L) Fold-change (week 4 vs week 0) in body mass (J), fat mass (K) and fat-free mass (L) for males vs females separated into younger (<45 years) and older (>45 years) groups. Data are presented as violin plots overlaid with individual data points. Significant effects of age, sex, and age*sex interaction were assessed using two-way ANOVA with Tukey’s multiple comparisons test. Overall p values for each variable, and their interactions, are shown beneath each graph. Significant differences between comparable groups are indicated by * (p<0.05) or *** (p<0.001). Source data are provided as a Source Data file. See also Table 2 and Figure 10—figure supplements 1–3.
-
Figure 10—source data 1
Effects of CR on body mass and body composition in humans are sex- and age-dependent.
Values for the data presented in Figure 10A–L are shown in the file named ‘Figure 10_Source_Data.xlsx’.
- https://cdn.elifesciences.org/articles/88080/elife-88080-fig10-data1-v2.zip
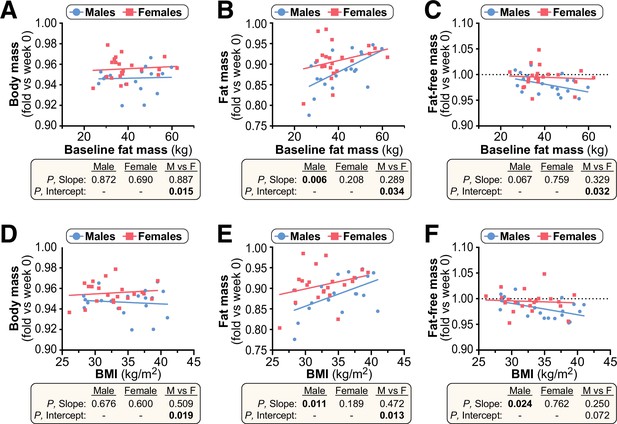
Baseline fat mass or BMI do not influence sex differences in the effects of CR on body mass or body composition.
Twenty male and twenty-two female volunteers participated in a weight loss study involving a 4-week dietary intervention, as described for Figure 10. (A–F) Simple linear regression of baseline fat mass (A–C) or baseline BMI (D–F) vs fold-change (week 4 vs week 0) in body mass (A,D), fat mass (B,E) and fat-free mass (C,F). For each sex, significant associations between age and outcome (fold-change) are indicated beneath each graph as ‘P, Slope’. ANCOVA was further used to test if the age-outcome relationship differs significantly between males and females. ANCOVA results are reported beneath each graph as ‘P, Slope’ and ‘P, Intercept’ for males vs females (M vs F). Similar slopes but different intercepts show that sex significantly influences weight loss, but the influence of baseline fat mass or BMI does not differ between the sexes. See also Table 2. Source data are provided as a Source Data file.
-
Figure 10—figure supplement 1—source data 1
Baseline fat mass or BMI do not influence sex differences in the effects of CR on body mass or body composition.
Values for the data presented in Figure 10—figure supplement 1A–F, and data used to calculate these values, are shown in the file named ‘Figure 10-figure supplement 1_Source_Data.xlsx’.
- https://cdn.elifesciences.org/articles/88080/elife-88080-fig10-figsupp1-data1-v2.zip
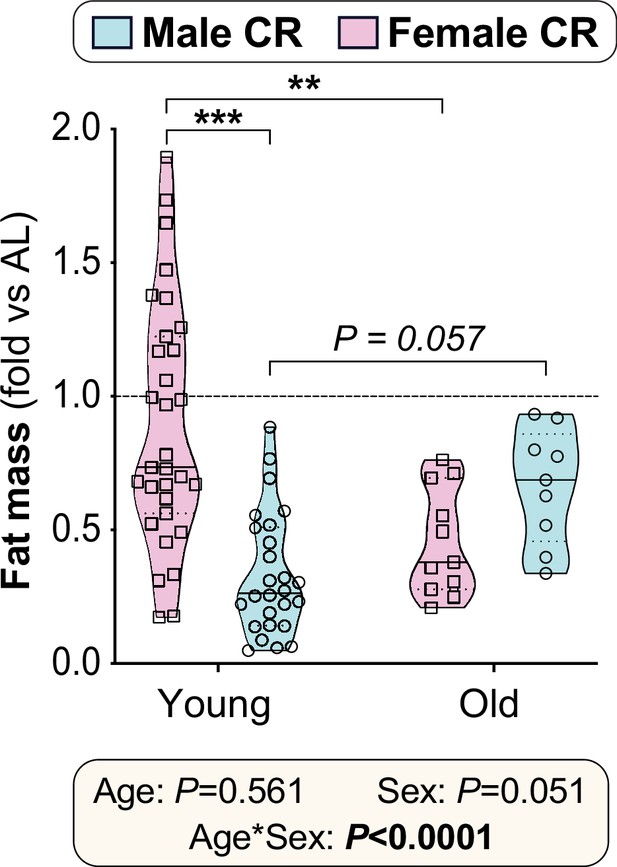
CR-induced fat loss in mice is age- and sex-dependent.
Male and female mice were fed AL or CR diets from 9 to 15 or 78–84 weeks of age and fat mass was determined each week, as described for Figures 2 and 7. For each mouse (CR and AL diets), the ratio of absolute fat masses (g) was calculated for week 4 vs week 0 (i.e. 13 vs 9 weeks of age for young mice; 82 vs 78 weeks for aged mice); week 4 was selected for consistency with the 4-week duration of our human CR study (Figure 9). For the CR mice, the fold-change in fat mass, relative to AL mice, was then determined as follows: (i) the mean week 4:week 0 ratio was calculated for AL mice in each age group; (ii) within each sex and age group, the week 4:week 0 ratio for each CR mouse was calculated relative to the mean week 4:week 0 ratio for the corresponding AL group. The rationale for showing these ‘fold changes of fold changes’ is that the AL mice gain fat mass over the course of the study and therefore we must determine how much the fat mass of each CR mouse has changed compared to where we would expect it to be if it had continued on AL diet. Data in are shown as violin plots of the following numbers of mice per group: young female, n=31; young male, n=26; old female, n=11; old male, n=9. Significant effects of diet and/or sex were determined by two-way ANOVA with Tukey’s multiple comparisons test and are reported as described for Figure 2—figure supplement 2. Source data are provided as a Source Data file.
-
Figure 10—figure supplement 2—source data 1
CR-induced fat loss in mice is age- and sex-dependent.
Values for the data presented in Figure 10—figure supplement 2, and data used to calculate these values, are shown in the file named ‘Figure 10-figure supplement 2_Source_Data.xlsx’.
- https://cdn.elifesciences.org/articles/88080/elife-88080-fig10-figsupp2-data1-v2.zip
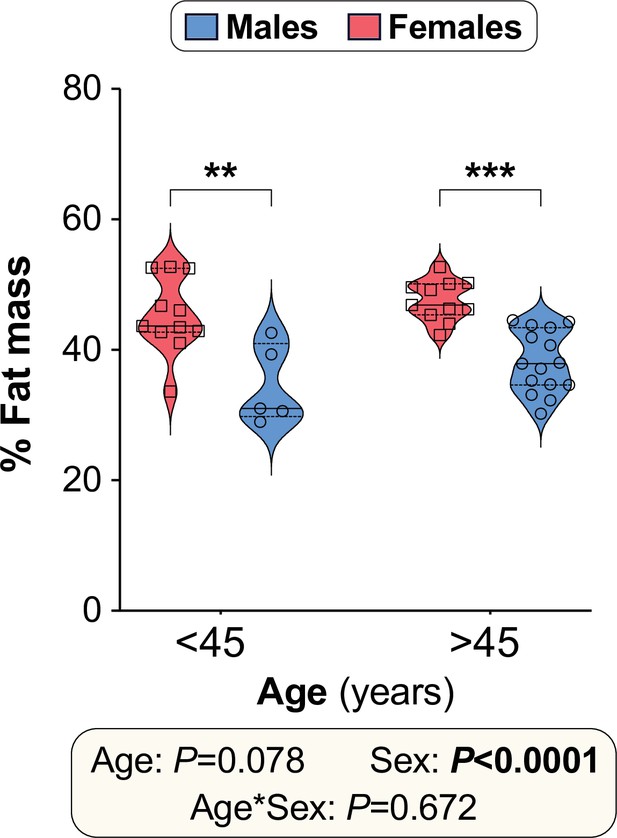
Body fat percentage in human CR participants.
Twenty male and twenty-two female volunteers participated in a weight loss study involving a 4-week dietary intervention, as described for Figure 10. Total % fat mass at week 0 is shown for males vs females separated into younger (<45 years) and older (>45 years) groups. Data are presented as violin plots overlaid with individual data points. Significant effects of age, sex, and age*sex interaction were assessed using two-way ANOVA with Tukey’s multiple comparisons test. Overall p values for each variable, and their interactions, are shown beneath the graph. Significant differences between comparable groups are indicated by ** (p<0.01) or *** (p<0.001). See also Table 2. Source data are provided as a Source Data file.
-
Figure 10—figure supplement 3—source data 1
Body fat percentage in human CR participants.
Values for the data presented in Figure 10—figure supplement 3, and data used to calculate these values, are shown in the file named “Figure 10-figure supplement 3_Source_Data.xlsx”.
- https://cdn.elifesciences.org/articles/88080/elife-88080-fig10-figsupp3-data1-v2.zip
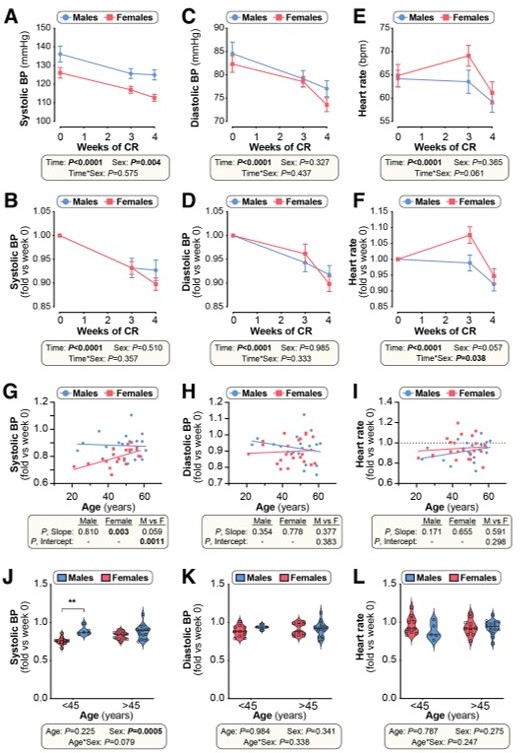
Effects of CR on cardiovascular parameters in humans (related to Figure 10).
Twenty male and twenty-two female volunteers participated in a weight loss study involving a 4-week dietary intervention, as described for Figure 9. (A-F) Systolic blood pressure (BP), diastolic BP and heart rate were recorded at weeks 0, 3 and 4. Data are shown as absolute values (A,C,E) or fold-change relative to baseline (B,D,F). Data are presented as mean ± SEM. Significant effects of time, sex, and time*sex interaction were assessed using 2-way ANOVA. (G-I) Simple linear regression of age vs fold-change (week 4 vs week 0) in systolic BP (G), diastolic BP (H) and heart rate (I). For each sex, significant associations between age and outcome (fold-change) are indicated beneath each graph as ‘P, Slope’. ANCOVA was further used to test if the age-outcome relationship differs significantly between males and females. ANCOVA results are reported beneath each graph as ‘P, Slope’ and ‘P, Intercept’ for males vs females (M vs F). In (G), similar slopes but different intercepts show that sex significantly influences changes in systolic BP, but the influence of age does not differ between the sexes. In (H,I) neither slopes nor intercepts differ significantly between males and females, indicating that the age-outcome relationship is similar between the sexes. (J-L) Fold-change (week 4 vs week 0) in systolic BP (J), diastolic BP (K) and heart rate (L) for males vs females separated into younger (<45 years) and older (>45 years) groups. Data are presented as violin plots overlaid with individual data points. Significant effects of age, sex, and age*sex interaction were assessed using 2-way ANOVA with Tukey’s multiple comparisons test. Overall P values for each variable, and their interactions, are shown beneath each graph. Significant differences between comparable groups are indicated by ** (P<0.01).
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Chemical compound, drug | 18F-Fluoro-deoxyglucose (18F-FDG) (radionuclide) | Edinburgh Clinical Research Imaging Centre (Edinburgh, UK) | N/A | |
| Chemical compound, drug | Ribozol | Amresco (USA) | N580 | |
| Commercial assay or kit | NEFA assay reagents | FUJIFILM Wako Chemicals Europe GmbH (Neuss, Germany) | 434–91795 436–91995 270–77000 | |
| Antibody | anti-HSL (Rabbit polyclonal) | Cell Signaling Technology (Danvers, MA, USA) | #4107 RRID:AB_2296900 | (1:1000) |
| Antibody | Anti-Phospho-HSL (Ser563) (Rabbit polyclonal) | Cell Signaling Technology (Danvers, MA, USA) | #4139 RRID:AB_2135495 | (1:1000) |
| Antibody | Anti-beta actin (Rabbit polyclonal) | Abcam (Cambridge, UK) | #ab8227 RRID:AB_2305186 | (1:1000) |
| Antibody | IRDye 800CW Goat anti-Rabbit IgG Secondary Antibody | LI-COR (Lincoln, NE, USA) | #926–32211 | (1:5000) |
| Strain, strain background (Mus musculus) | C57BL/6 J or C57BL/6NCrl | Charles River | 027 RRID:IMSR_CRL:027 | |
| Software, algorithm | Prism | GraphPad Software, LLC | v9.5.1 | |
| Software, algorithm | PMOD | PMOD Technologies LLC (Zurich, Switzerland) | v3.806 | |
| Software, algorithm | TrimGalore | https://www.bioinformatics.babraham.ac.uk/projects/trim_galore/ | V0.6.6 | |
| Software, algorithm | FastQC | https://www.bioinformatics.babraham.ac.uk/projects/fastqc/ | v0.11.7 | |
| Software, algorithm | STAR | Dobin et al., 2013 | v2.7.10a | |
| Software, algorithm | Subread | Liao et al., 2014 | v1.5.2 | |
| Software, algorithm | R | https://www.r-project.org/ | v4.2.0 | |
| Software, algorithm | RStudio | https://posit.co/download/rstudio-desktop/ | v2022.12.0+353 | |
| Software, algorithm | DESeq2 | Love et al., 2014 | v1.36.0 | |
| Software, algorithm | GSEA | Mootha et al., 2003; Subramanian et al., 2005 | V4.3.2 | |
| Software, algorithm | Morpheus | https://software.broadinstitute.org/morpheus | NA | |
| Software, algorithm | Enricher | Xie et al., 2021; Kuleshov et al., 2016; Chen et al., 2013 | NA | |
| Software, algorithm | appyters | Clarke et al., 2021 | NA | |
| Software, algorithm | Image Studio | LI-COR Biosciences (USA) | v5.2 | |
| Software, algorithm | ExpeData | Sable Systems International (Las Vegas, USA) | v1.9.27 |
Summary of CR protocol in each group of mice.
Because mice are singly housed, each mouse represents an independent experimental unit.
| Young | Aged | Evening-fed | |
|---|---|---|---|
| Age at single housing | 8 weeks | 77 weeks | 8 weeks |
| Age at start of CR | 9 weeks | 78 weeks | 9 weeks |
| Time of feeding | 0900–1000 | 0900–1000 | 1800–1900 |
| Duration of CR | 6 weeks | 6 weeks | 4 weeks |
| Related data | Figures 2—7, Figure 2—figure supplements 1–3; Figure 3—figure supplements 1–2; Figure 5—figure supplement 1; Figure 6—figure supplement 1; Figure 7—figure supplement 1; Figure 10—figure supplement 2 | Figures 8–9; Figure 8—figure supplement 1; Figure 10—figure supplement 2 | Figure 2—figure supplement 4; Figure 4—figure supplement 1 |
| Experimental unit | Single mouse | ||
| Groups compared | AL vs CR (within sex); Male vs Female (within diet); and diet*sex interactions | ||
Human subject characteristics at baseline.
Age, BMI, glucose and daily caloric intake (Diet) are mean ± SD. ND = not determined.
| Sex | Number of subjects | Age (years) | BMI (kg/m2) | Glucose (mM) | Diet (kcal/day) | Diabetic |
|---|---|---|---|---|---|---|
| Male | 20 | 48.61±10.7 | 34.7±4.2 | 5.6±0.7 | 1616±214 | 0% |
| Female | 22 | 44.7±9.7 | 32.8±3.7 | 5.2±0.3 | 1325±62 | 0% |
Gene sets used for GSEA.
Further details can be found by searching for the Standard Gene Set name at http://www.gsea-msigdb.org/gsea/msigdb/mouse/genesets.jsp.
| Gene Set name (Standard) | Systematic Name | Gene set name (shown in Figure 7C) |
|---|---|---|
| REACTOME_GLUCONEOGENESIS | MM15392 | GLUCONEOGENESIS |
| HALLMARK_OXIDATIVE_PHOSPHORYLATION | MM3893 | OXIDATIVE_PHOSPHORYLATION |
| REACTOME_CITRIC_ACID_CYCLE_TCA_CYCLE | MM15407 | CITRIC_ACID_CYCLE_TCA_CYCLE |
| HALLMARK_GLYCOLYSIS | MM3894 | GLYCOLYSIS |
| REACTOME_MITOCHONDRIAL_FATTY_ACID_BETA_OXIDATION | MM15462 | FATTY_ACID_BETA_OXIDATION |
| WP_FATTY_ACID_BIOSYNTHESIS | MM15885 | FATTY_ACID_BIOSYNTHESIS |
| HALLMARK_ESTROGEN_RESPONSE_EARLY | MM3872 | ESTROGEN_RESPONSE_EARLY |
| HALLMARK_ESTROGEN_RESPONSE_LATE | MM3873 | ESTROGEN_RESPONSE_LATE |





