Structural dynamics of the active HER4 and HER2/HER4 complexes is finely tuned by different growth factors and glycosylation
Figures
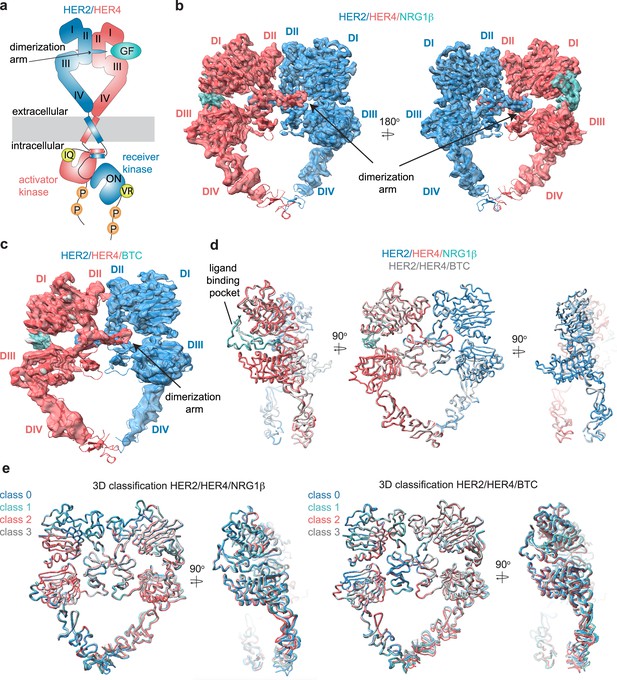
Structures of the HER2/HER4 heterodimers bound to NRG1β or BTC.
(a) Cartoon schematic of the HER2/HER4/NRG1β heterodimer depicts the assembly of a ‘heart-shaped’ ectodomain dimer upon binding of a ligand/growth factor (GF) to HER4. Individual domains of the HER ectodomains are annotated as domain (D) I – IV. The intracellular kinase domains assemble into an asymmetric dimer in which HER2 adopts the receiver (activated) and HER4 the activator (inactive) positions, enforced by the interface mutations: HER2-V956R and HER4-I712Q, respectively. (b–c) Structures of the near full-length HER2-V956R/HER4-I712Q complex (labeled HER2/HER4) bound to NRG1β or BTC. The ectodomain models are shown in cartoon representation fitted into the cryo-EM density. Only density for the ectodomain modules was observed. Domains I-IV are labeled DI-DIV. (d) Overlay of HER2/HER4 heterodimers bound to NRG1β and BTC aligned on the HER2 chain (RMSD 0.835 Å). (e) 3D classification analysis of HER2/HER4 heterodimers bound to NRG1β or BTC. Overlay of models in ribbon resulting from the 3D classification of particles into four classes are shown (HER2/HER4/NRG1β 289,192 particles, HER2/HER4/BTC 148,541 particles). Models were aligned on the HER2 chain.
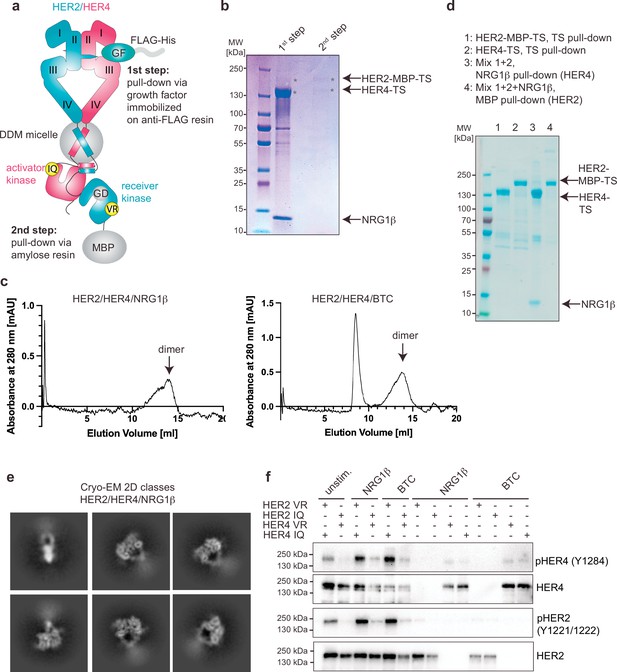
Purification and the functional analysis of the HER2/HER4 heterodimers.
(a) Overview of the HER2/HER4 purification strategy. HER2 features a G778D (GD) mutation to mediate Hsp90-independence. VR corresponds to HER2-V956R (receiver) and IQ to HER4-I712Q (activator). The mutant complex was used for all purification and structure determination steps shown in this figure (panels b-e) and is referred to as HER2/HER4. (b) Coomassie-stained SDS-PAGE gel analysis of the samples from the HER2/HER4 purification after ligand-mediated pulldown (1st step) and MBP pulldown (second step). (c) Representative Size Exclusion Chromatography (SEC) profiles for liganded HER2/HER4 heterocomplexes. (d) Coomassie-stained SDS-PAGE gel analysis of indicated HER2 and HER4 pulldown experiments. Lanes 1 and 2 show HER2-MBP-TS and HER4-TS TS (Twin-Strep) pulldown eluates. Eluates from lanes 1 and 2 were mixed and NRG1β-mediated (lane 3) or MBP pulldowns (amylose resin, lane 4) were performed (e) Representative 2D cryo-EM class averages of liganded HER2/HER4/NRG1β heterocomplexes. Box size is 321 Å. (f) Western blot showing that activation of HER2/HER4 heterodimers requires HER2 to adopt the kinase receiver function (HER2-VR) and HER4 to adopt the kinase activator (HER4-IQ) function in the heterodimer. Full-length constructs were co-transfected into COS7 cells, starved overnight, and stimulated with 10 nM ligand for 10 min at 37 °C. The HER2 constructs used in this experiment do not feature the G778D mutation. The blot is representative of three independent experiments.
-
Figure 1—figure supplement 1—source data 1
Original scan for Coomassie-stained gel in Figure 1—figure supplement 1b.
- https://cdn.elifesciences.org/articles/92873/elife-92873-fig1-figsupp1-data1-v1.zip
-
Figure 1—figure supplement 1—source data 2
Original scan for Coomassie-stained gel in Figure 1—figure supplement 1b with all labels and cropped areas shown.
- https://cdn.elifesciences.org/articles/92873/elife-92873-fig1-figsupp1-data2-v1.zip
-
Figure 1—figure supplement 1—source data 3
Original scan for Coomassie-stained gel in Figure 1—figure supplement 1d.
- https://cdn.elifesciences.org/articles/92873/elife-92873-fig1-figsupp1-data3-v1.zip
-
Figure 1—figure supplement 1—source data 4
Original scan for Coomassie-stained gel in Figure 1—figure supplement 1d with all labels and cropped areas shown.
- https://cdn.elifesciences.org/articles/92873/elife-92873-fig1-figsupp1-data4-v1.zip
-
Figure 1—figure supplement 1—source data 5
Original scan for western blot in Figure 1—figure supplement 1f.
- https://cdn.elifesciences.org/articles/92873/elife-92873-fig1-figsupp1-data5-v1.zip
-
Figure 1—figure supplement 1—source data 6
Original scan for western blot in Figure 1—figure supplement 1f with all labels and cropped areas shown.
- https://cdn.elifesciences.org/articles/92873/elife-92873-fig1-figsupp1-data6-v1.zip
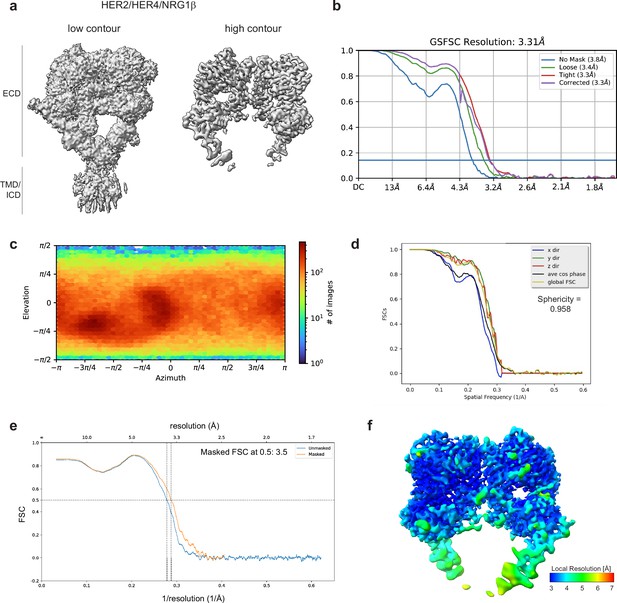
Cryo-EM density maps of HER2/HER4 bound to NRG1β.
(a) Cryo-EM map at different contour levels. (b) CryoSPARC GSFSC plots. (c) CryoSPARC Euler angle plots. (d) 3DFSC plots. (e) Model-Map-FSC curves from Phenix Validation (f) Local resolution map of HER2/HER4/NRG1βcreated using cryoSPARC v4.
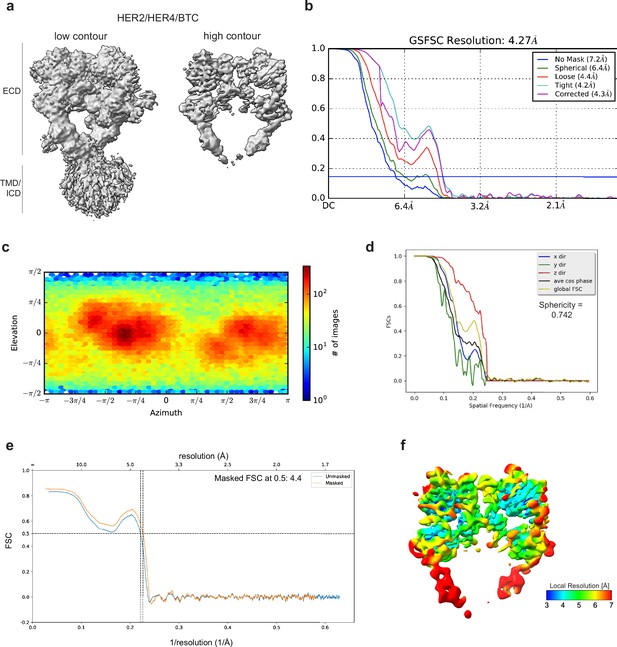
Cryo-EM density maps of HER2/HER4 bound to BTC.
(a) Cryo-EM map at different contour levels. (b) CryoSPARC GSFSC plots. (c) CryoSPARC Euler angle plots. (d) 3DFSC plots. (e) Model-Map-FSC curves from Phenix Validation (f) Local resolution map of HER2/HER4/BTCcreated using cryoSPARC v4.
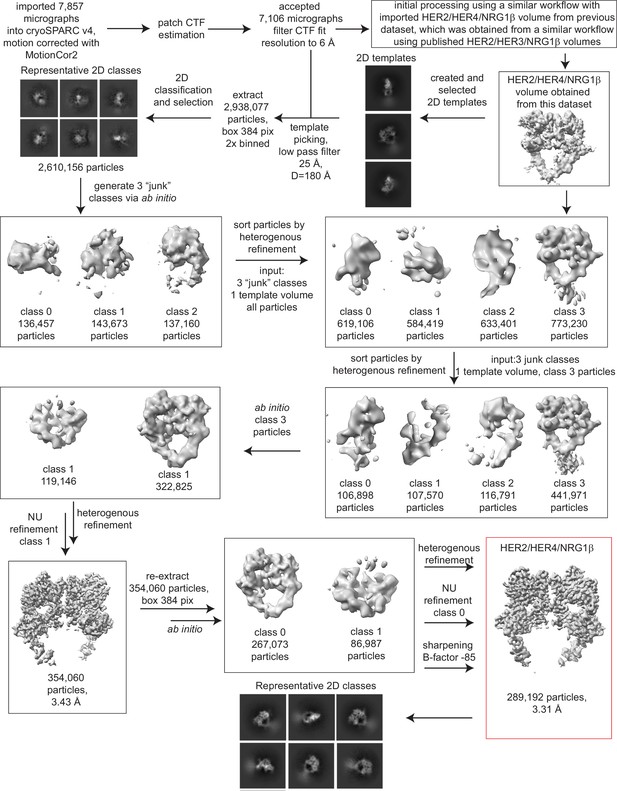
Processing workflow for the HER2/HER4/NRG1β structure.
Data were processed in cryoSPARC v4 using a strategy in which particles are picked generously using a template picker, selected by 2D classification to remove bad picks (<10% of particles), and then sorted via two rounds of heterogeneous refinement into a HER receptor dimer template volume and three ‘junk’ classes created from the impure particle stack. Picked particles were subjected to ab initio reconstruction to eliminate bias and further processed as shown.
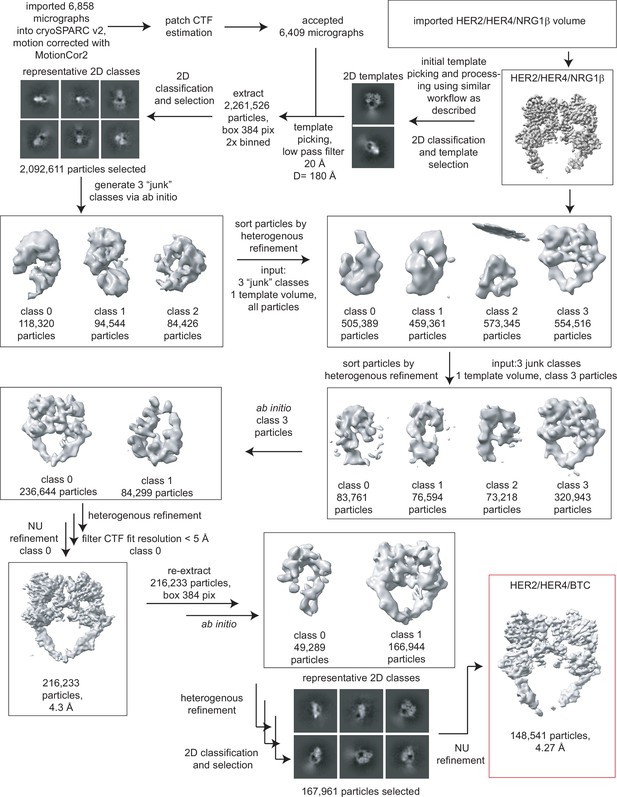
Processing workflow for the HER2/HER4/BTC structure.
Data were processed in cryoSPARC v2 using a strategy in which particles are picked generously using a template picker, selected by 2D classification to remove bad picks (<10% of particles), and then sorted via two rounds of heterogeneous refinement into a HER receptor dimer template volume and three ‘junk’ classes created from the impure particle stack. Picked particles were subjected to ab initio reconstruction to eliminate bias and further processed as shown.
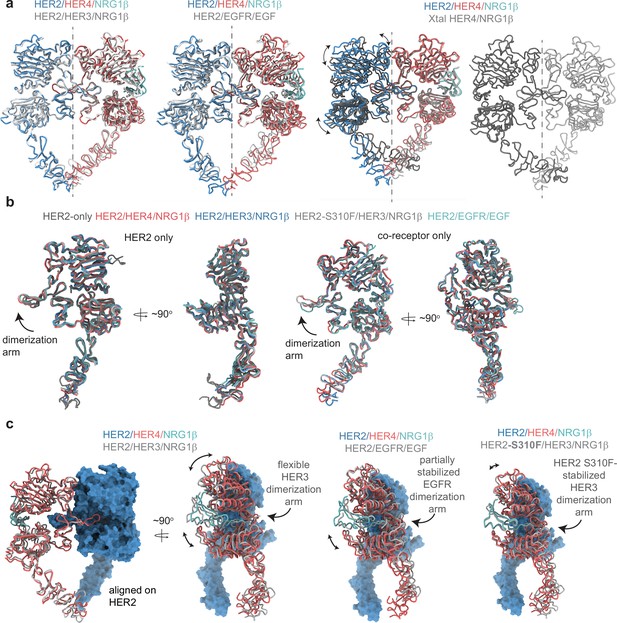
Comparison between the HER2 and HER4 homo- and heterodimeric ectodomain structures.
(a) Overlays of indicated homo- and heterodimers. Heterodimer alignments were performed using the HER2 chain, and alignments with HER4 homodimers were performed using the HER4 chain. The dotted line represents a C2 symmetry axis highlighting the asymmetry of heterodimers compared to near-perfect C2 symmetry observed for HER4/NRG1β homodimers. (b) Individual receptors from the HER2-containing heterodimers were aligned using the HER2 chain or its co-receptor chain, as indicated. HER2-only is cryo-EM structure of the HER2 ECD with Pertuzumab and Trastuzumab Fab bound (PDB: 6OGE; Fabs not shown), HER2/HER3/NRG1β (PDB: 7MN5), HER2-S310F/HER3/NRG1β (PDB: 7MN6), HER2/EGFR/EGF (PDB: 8HGO). (c) Comparison between indicated HER2 heterodimer structures. Structural models are overlayed on HER2 to highlight nuances with which HER2 engages its co-receptors. The same PDB codes were used as in (b).
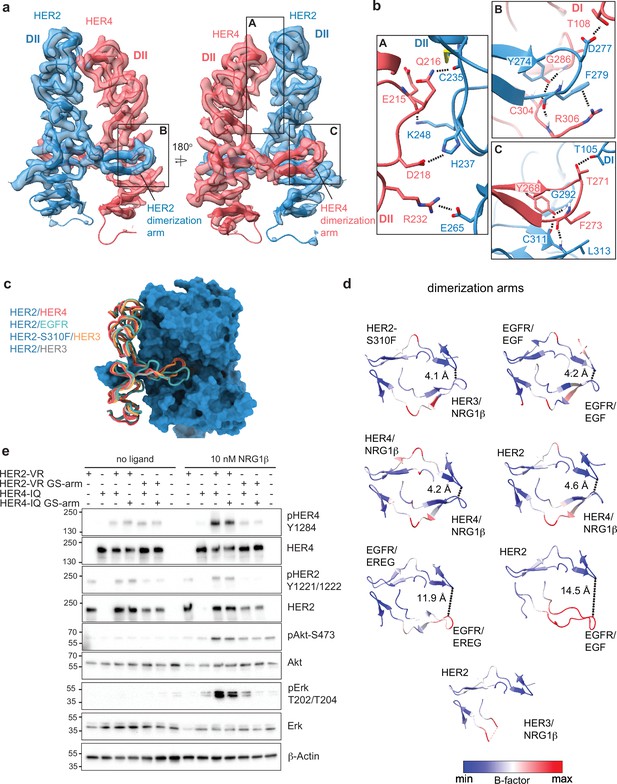
Near symmetric engagement of the HER2 and HER4 dimerization arms at the dimerization interface.
(a) Cryo-EM density and model of the HER2/HER4/NRG1β domain II at two different orientations highlight two equally well-resolved dimerization arms. (b) Hydrogen-bonds, cation-π interactions, and salt bridges are depicted at the dimer interface, with other residues omitted for clarity. The HER2 and HER4 dimerization arms engage in the same set of polar interactions (insets B and C), except for a cation-π interaction between HER2 F279 with HER4 R306 (A) due to a substitution of the equivalent of HER4 R306 to L313 in HER2. Residues labeled ‘DI’ are in receptor domain I while all others are in domain II (DII). Interface residues and hydrogen bonds were determined using UCSF ChimeraX. (c) Known HER2 heterodimers are aligned using the HER2 chain to highlight the positioning of the dimerization arms. (d) Dimerization arm regions of selected HER receptor dimers are shown colored by B-factors. B-factor colors were scaled to represent max and min B-factor values within each structure corresponding to different absolute values across structures due to variability in their resolution. Distance measurements at fixed points highlight a correlation between asymmetrically distributed B-factors and asymmetrically engaged dimerization arms. (e) Western blot analysis of NR6 cell lysates transduced with indicated HER2 and HER4 constructs. Cells were starved for 4 h prior to stimulation with 10 nM NRG1β at 37 °C for 10 min. Molecular weight markers (in kDa) are indicated next to each blot.
-
Figure 2—source data 1
Original files for western blot analysis in Figure 2e.
- https://cdn.elifesciences.org/articles/92873/elife-92873-fig2-data1-v1.zip
-
Figure 2—source data 2
Original files for western blot analysis in Figure 2e with all labels and cropped areas shown.
- https://cdn.elifesciences.org/articles/92873/elife-92873-fig2-data2-v1.zip
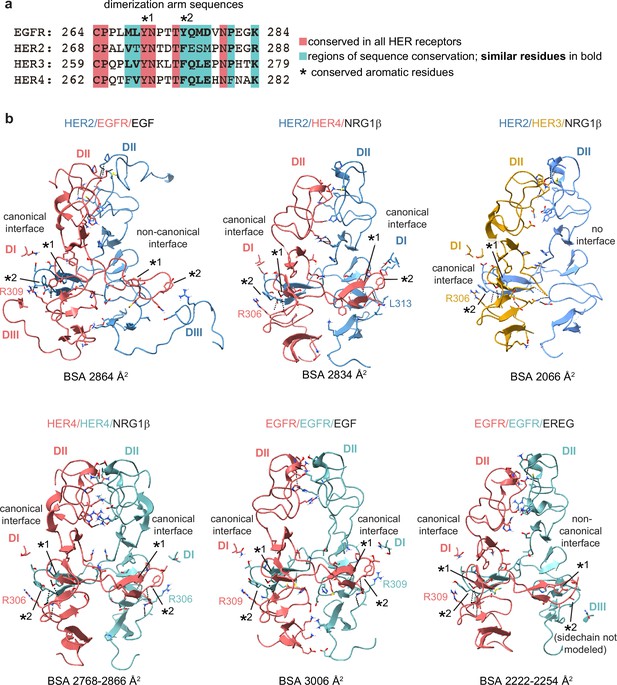
Detailed view of the dimer interfaces of HER receptor homo- and heterodimers.
(a) Sequence alignment of HER receptor dimerization arm regions with conserved residues highlighted in red. Two aromatic residues that are known to engage in hydrogen bonding with the partner receptor are marked with (*). (b) Full domains II (DII) for selected receptor dimers are shown in the cartoon and all the interface residues between two receptors within domains I and III (DI-DIII) are shown as sticks. Hydrogen bonds are indicated with dotted lines. Analysis was performed using UCSF ChimeraX. Domains IV are not resolved in most structures and are not included in this analysis. Canonical dimerization arm interactions involve domains DI and DII, while non-canonical interfaces, as seen for EGFR in the HER2/EGFR/EGF dimer and one EGFR/EREG monomer in the EGFR/EREG homodimer, engage DIII instead of DI. The buried surface area (BSA) at each interface is indicated. If more than one crystallographic dimer was observed in an asymmetric unit, the range of BSA values for all observed dimers is indicated. The following PDB codes were used: HER2/HER3/NRG1β (PDB: 7MN5), HER2/EGFR/EGF (PDB: 8HGO), EGFR/EGF (PDB: 3NJP), HER4/NRG1β (PDB: 3U7U), and EGFR/EREG (PDB: 5WB7).
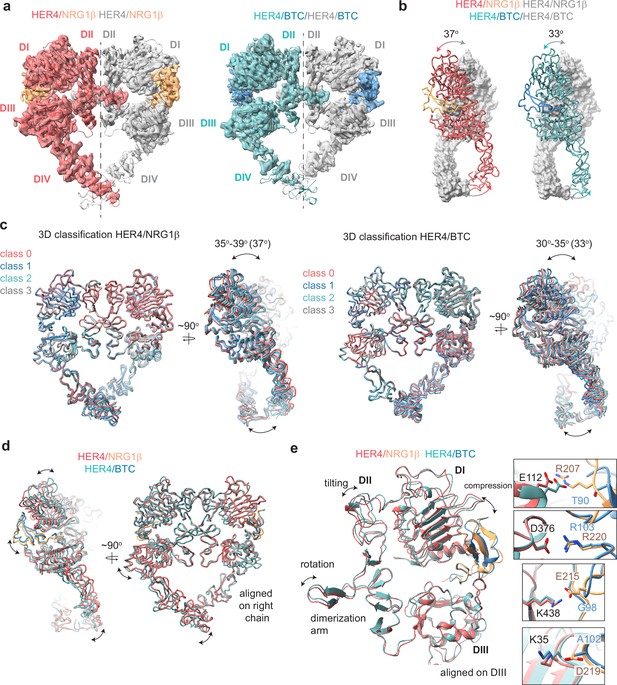
Structures of HER4 homodimers bound to NRG1β or BTC reveal ligand-specific conformational heterogeneity.
(a) Structures of full-length HER4 homodimers bound to either NRG1β or BTC. Only density for the ectodomain modules was observed in both structures, shown here as a cartoon representation fitted into the cryo-EM density. (b) Comparison between the NRG1β− and BTC-bound HER4 dimers. Angle measurements were derived using UCSF ChimeraX by defining an axis through each receptor in a dimer and measuring the angle between the two axes. (c) Overlays of ribbon models obtained by 3D classification of particles into four distinct classes are shown for HER4 homodimers bound to NRG1β or BTC (205,726 particles HER4/NRG1β and ~274,540 particles HER4/BTC). Classification was performed in cryoSPARC using the heterogeneous refinement job with four identical start volumes and particles from final reconstructions are shown in (a). (d-e) Overlays of HER4 receptor homodimers bound to NRG1β or BTC show differences in the ligand binding pockets and how receptors assemble into dimers. Receptors were aligned as indicated in the panels. The HER4-NRG1β engages 4 salt bridges in the binding pocket, three of which are not present in HER4-BTC (shown in boxes). The salt bridge involving HER4 K35 can only be confidently observed in cryo-EM maps of one monomer (chain A).
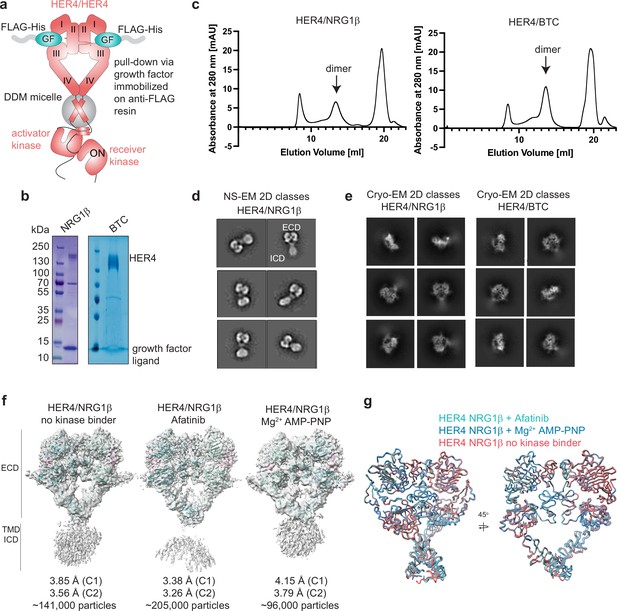
Purification of HER4 homodimers bound to NRG1β or BTC and structural analysis.
(a) Overview of the HER4 purification strategy. Untagged, full-length HER4 was purified by growth factor (GF)-coated anti-FLAG resin. (b) Coomassie-stained SDS-PAGE gel showing receptor samples after ligand-mediated receptor pulldown. (c) Size Exclusion Chromatography (SEC) profiles of samples after ligand-mediated receptor pulldown using a Superose 6 increase 10/300 GL column. Elution fractions are consistent with receptor dimers were used for negative-stain EM (NS-EM) and cryo-EM analyses. (d) HER4/NRG1β NS-EM 2D class averages show receptor dimers with ‘heart’-shaped ectodomains and additional density for intracellular kinase domains. (e) HER4/NRG1β and HER4/BTC cryo-EM 2D class averages show receptor dimers with ‘heart’-shaped extracellular domains without density for intracellular kinase domains. (f) Cryo-EM volumes of HER4/NRG1β obtained from HER4/NRG1β preparations in an apo form, with afatinib, or with Mg2+AMP-PNP bound. 10 mM afatinib was added to the culture medium during expression, 1 mM Mg2+AMP-PNP was added prior to crosslinking with glutaraldehyde (after FLAG elution). Receptors were subjected to SEC and 1 mM Mg2+AMP-PNP was again added prior to cryo-EM grid preparation. (g) Overlay of models for volumes in (f) shows all three volumes are identical.
-
Figure 3—figure supplement 1—source data 1
Original scans for Coomassie-stained gels in Figure 3—figure supplement 1b.
- https://cdn.elifesciences.org/articles/92873/elife-92873-fig3-figsupp1-data1-v1.zip
-
Figure 3—figure supplement 1—source data 2
Original scans for Coomassie-stained gels in Figure 3—figure supplement 1b with all labels and cropped areas shown.
- https://cdn.elifesciences.org/articles/92873/elife-92873-fig3-figsupp1-data2-v1.zip
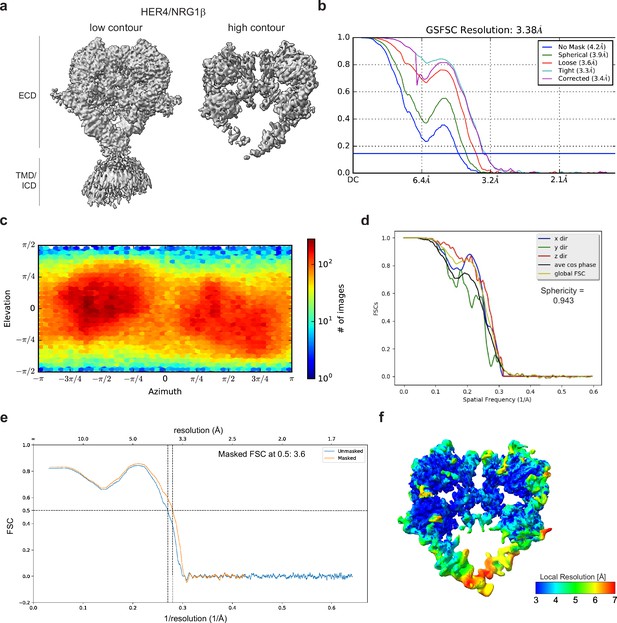
Cryo-EM density maps of HER4 bound to NRG1β processed without symmetry applied.
(a) Cryo-EM map at different contour levels. (b) CryoSPARC GSFSC plots. (c) CryoSPARC Euler angle plots. (d) 3DFSC plots. (e) Model-Map-FSC curves from Phenix Validation (f) Local resolution map of HER4/NRG1βcreated using cryoSPARC v4.
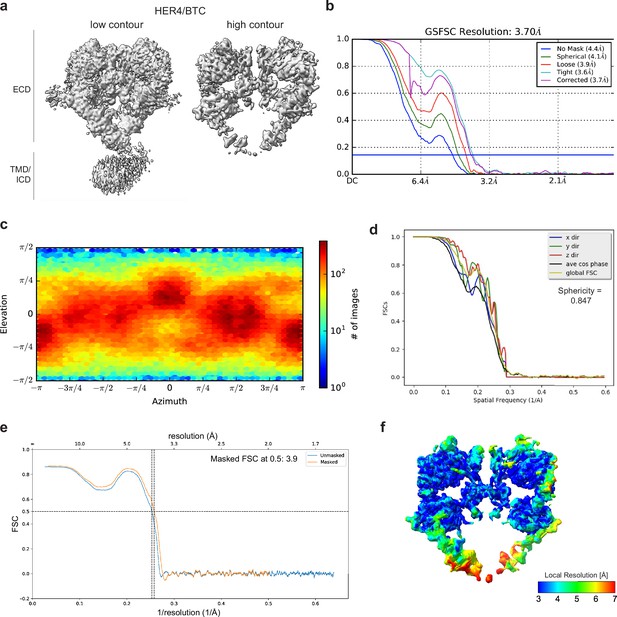
Cryo-EM density maps of HER4 bound to BTC processed without symmetry applied.
(a) Cryo-EM map at different contour levels. (b) CryoSPARC GSFSC plots. (c) CryoSPARC Euler angle plots. (d) 3DFSC plots. (e) Model-Map-FSC curves from Phenix Validation. (f) Local resolution map of HER4/BTC created using cryoSPARC v4.
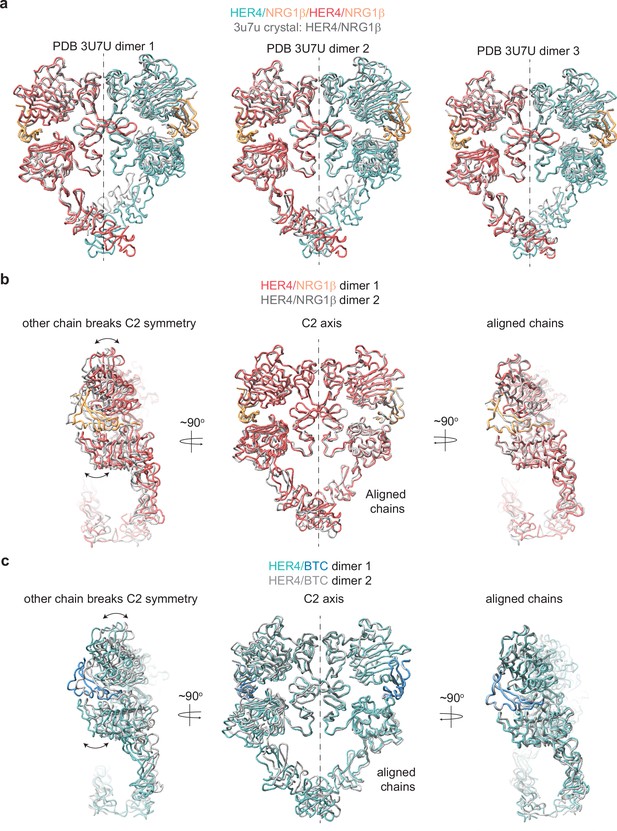
HER4 homodimers do not show ideal C2 symmetry.
(a) Overlay of three HER4/NRG1β ectodomain homodimers found in the asymmetric unit of the crystal structure (PDB: 3U7U) with the cryo-EM structure of full-length HER4/NRG1β. The crystal structure models are shown in gray. RMSDs for overlay of full dimers with the cryo-EM HER4/NRG1β dimer are 5.438 Å, 5.435 Å, and 3.662 Å, respectively. (b) HER4/NRG1β model built into C1 refined cryo-EM map was aligned across chains (chain A in one model aligned to chain B in another model). While the aligned chain showed a near-perfect match (RMSD 1.42 Å), the other chain showed a breaking of C2 symmetry. (c) HER4/BTC model built into C1 refined cryo-EM map was aligned across chains (chain A in one model aligned to chain B in another model). While the aligned chain showed a near-perfect match (RMSD 1.58 Å), the other chain showed a breaking of C2 symmetry.
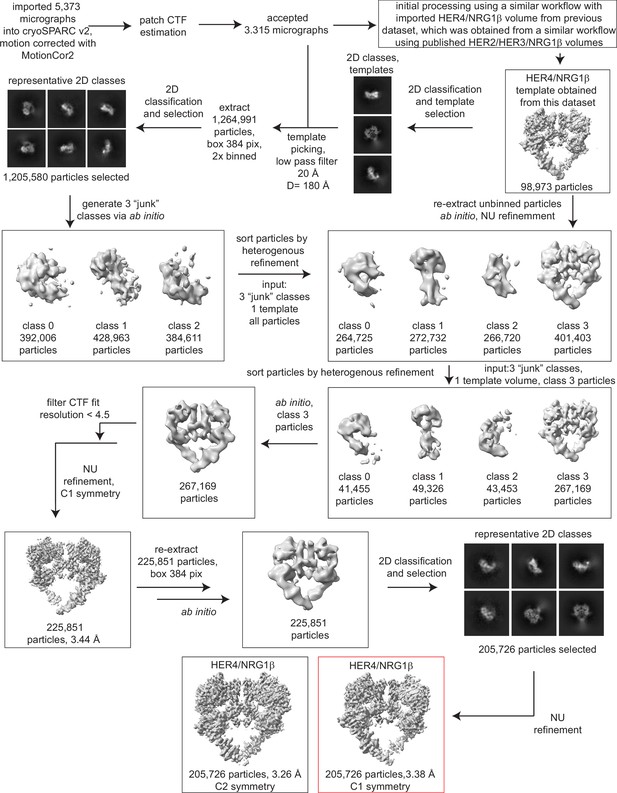
Processing workflow and data statistics for the HER4/NRG1β homodimer.
Data were processed in cryoSPARC v2 using a strategy in which particles are picked generously using a template picker, selected by 2D classification to remove bad picks (<10% of particles), and then sorted via two rounds of heterogeneous refinement into a HER receptor dimer template volume and three ‘junk’ classes created from the impure particle stack. Picked particles were subjected to ab initio reconstruction to eliminate bias and further processed as shown.
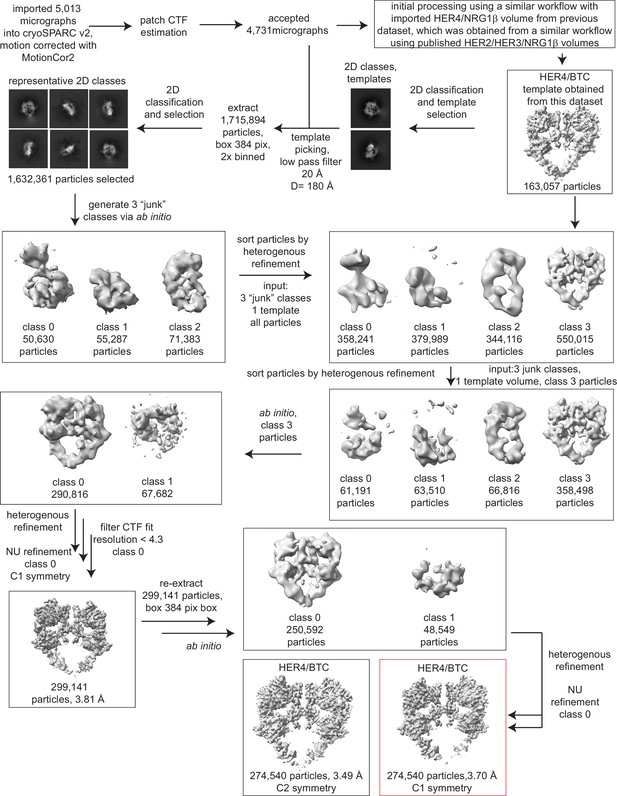
Processing workflow and data statistics for the HER4/BTC homodimer.
Data were processed in cryoSPARC v2 using a strategy in which particles are picked generously using a template picker, selected by 2D classification to remove bad picks (<10% of particles), and then sorted via two rounds of heterogeneous refinement into a HER receptor dimer template volume and three ‘junk’ classes created from the impure particle stack. Picked particles were subjected to ab initio reconstruction to eliminate bias and further processed as shown.
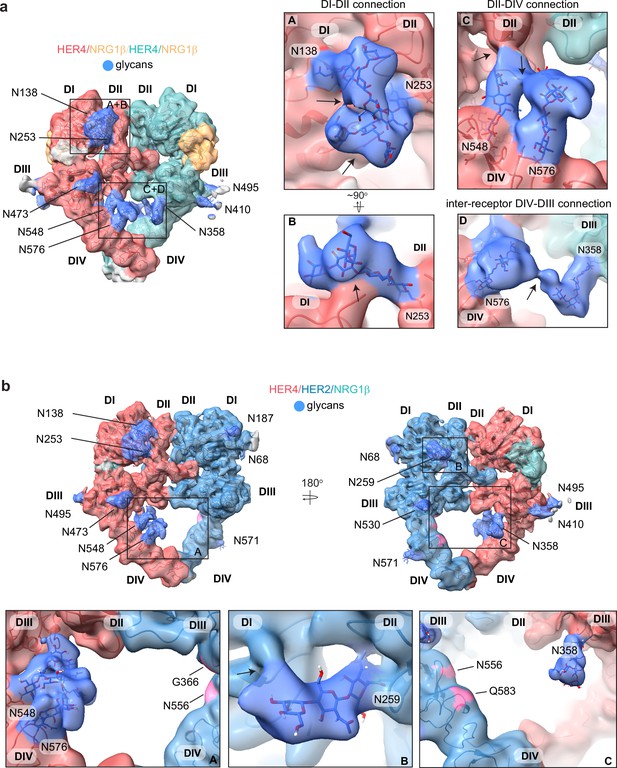
HER4 homodimers are stabilized via inter-receptor glycans.
(a) Model of the HER4/NRG1β homodimer fitted into the cryo-EM density, lowpass-filtered to 6 Å, reveals multiple glycans that mediate intra- and interreceptor connections. Glycans are shown in blue. Insets (A and B) are close-up views of glycans connected to N138 and N253, and are shown at a higher volume contour than the central heterodimer. Insets (C and D) are close-up views of glycans connected to N548, N576, and N358. (D) shows continuous glycan density originating from N576 of one receptor and connecting to N358 of the dimerization partner. Maps are shown at lower contour than in the central heterodimer. Various contour levels are shown in Figure 3—figure supplement 4a for reference. Arrows indicate regions in which the cryo-EM map from one glycan merges with the density of glycans or polypeptide chains from different HER receptor sub-domains. (b) Model of HER2/HER4/NRG1β fitted into cryo-EM density, lowpass-filtered to 6 Å, reveals intra-receptor glycosylation only. Insets (A) shows HER4 glycosylation on N548 and N576 pointing from HER4 domain IV to domain II, but less pronounced as observed in HER4 homodimers. Glycan connections between domain I and II in HER4, via N138 and N253-linked glycans, are comparable to the ones seen in HER4 homodimer shown in inset (A). Inset (B) shows the equivalent glycan connections in domain I and II of HER2. Inset (C) reveals missing glycosylation sites at equivalent positions in HER2; G366, N556, and Q583 (pink).
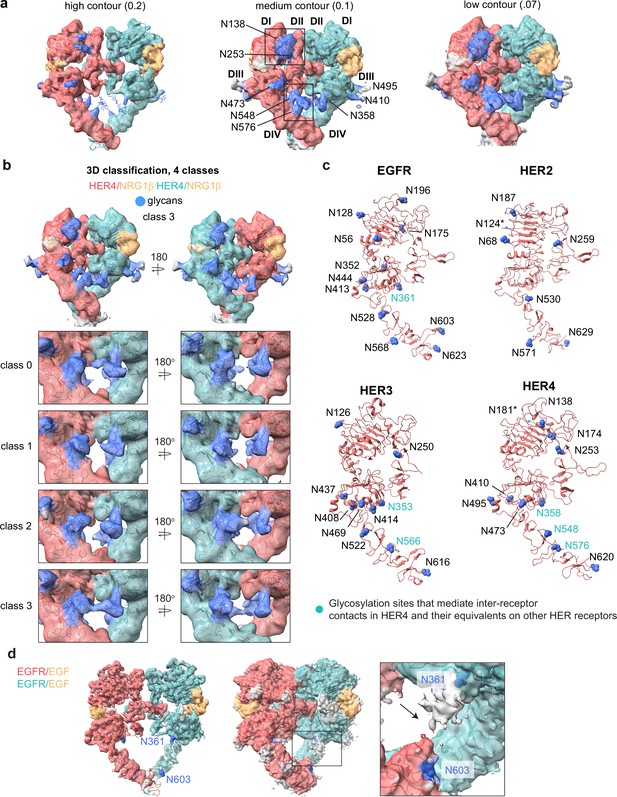
Glycosylation in the cryo-EM structures of HER4/NRG1β homodimer and comparison to other HER receptors.
(a) Model of the HER4/NRG1β homodimer fitted into the cryo-EM density, lowpass-filtered to 6 Å, at various contour levels. Glycans are shown in blue. (b) 3D classification of the HER4/NRG1β particles reveals strong continuous glycan density between two receptors within the dimer for class 3. (c) Glycosylation site asparagines in EGFR, HER2, HER3, and HER4 are marked in blue, and shown in sphere representation. Glycosylation site asparagines involved in inter-receptor contacts in our HER4 structures, and the equivalent residues in other HER receptors are indicated by teal labels. (d) Analysis of the cryo-EM map of the EGFR/EGF homodimer structure (PDB: 7SYD) at various contour levels suggests the presence of an inter-receptor glycan connection between N353 and N603, shown in blue.
Tables
Cryo-EM data collection, refinement, and validation statistics.
| HER2/HER4/NRG1β(EMDB: EMD-41886)(PDB: 8U4L) | HER2/HER4/BTC(EMDB: EMD-41885)(PDB: 8U4K) | HER4/NRG1β(EMDB: EMD-41883)(PDB: 8U4I) | HER4/BTC(EMDB: EMD:41884)(PDB: 8U4J) | |
|---|---|---|---|---|
| Data collection and processing | ||||
| Magnification | 105000 x | 105000 x | 105000 x | 105000 x |
| Voltage (kV) | 300 | 300 | 300 | 300 |
| Electron exposure (e–/Å2) | 45.8 | 45.8 | 68.7 | 45.8 |
| Defocus range (μm) | 0.9–2.0 | 0.9–2.0 | 0.9–2.0 | 0.9–2.0 |
| Pixel size (Å) | 0.835 | 0.835 | 0.835 | 0.835 |
| Symmetry imposed | C1 | C1 | C1 | C1 |
| Initial particle images (no.) | 2938077 | 2261526 | 1264991 | 1715894 |
| Final particle images (no.) | 289192 | 148541 | 205726 | 274540 |
| Map resolution (Å) FSC threshold | 3.3 0.143 | 4.3 0.143 | 3.4 0.143 | 3.7 0.143 |
| Map resolution range (Å) | 3–7 | 3–7 | 3–7 | 3–7 |
| Refinement | ||||
| Initial model used (PDB code) | 7MN5, 3U7U | 7MN5, 3U7U, AF-P35070-F1 | 3U7U | 3U7U, AF-P35070-F1 |
| Model resolution (Å) FSC threshold | 3.5 0.5 | 4.4 0.5 | 3.6 0.5 | 3.9 0.5 |
| Map sharpening B factor (Å2) | –85 | –162.4 | –105.5 | –135.8 |
| Model composition Non-hydrogen atoms Protein residues Ligands | 9989 1230 35 | 9989 1228 35 | 10842 1301 57 | 10851 1297 58 |
| B factors (Å2) Protein Ligand | 70.49 150.01 | 239.03 398.81 | 70.81 140.13 | 166.67 330.96 |
| R.m.s. deviations Bond lengths (Å) Bond angles (°) | 0.012 1.614 | 0.012 1.697 | 0.012 1.635 | 0.012 1.598 |
| Validation MolProbity score Clash score Poor rotamers (%) | 0.78 0.93 0.09 | 1.04 0.93 0.19 | 0.71 0.62 0.35 | 0.81 0.81 0.17 |
| Ramachandran plot Favored (%) Allowed (%) Disallowed (%) | 98.02 1.98 0 | 96.20 3.80 0 | 97.97 2.03 0 | 97.74 2.26 0 |





