Prdm1 positively regulates liver Group 1 ILCs cancer immune surveillance and preserves functional heterogeneity
eLife assessment
The authors investigated the requirement and function of Blimp1/Prdm1 in murine natural killer (NK) cells and the ILC1 lineage of innate lymphoid cells, using a conditional knockout model. The single-cell mRNA-seq data provided here represent a valuable resource for the community, but the lack of mechanistic investigations leaves the study partially incomplete. The work will be of interest to the fields of innate lymphoid cell biology and tissue immunology.
https://doi.org/10.7554/eLife.92948.3.sa0Valuable: Findings that have theoretical or practical implications for a subfield
- Landmark
- Fundamental
- Important
- Valuable
- Useful
Incomplete: Main claims are only partially supported
- Exceptional
- Compelling
- Convincing
- Solid
- Incomplete
- Inadequate
During the peer-review process the editor and reviewers write an eLife Assessment that summarises the significance of the findings reported in the article (on a scale ranging from landmark to useful) and the strength of the evidence (on a scale ranging from exceptional to inadequate). Learn more about eLife Assessments
Abstract
Group 1 innate lymphoid cells (ILCs) comprise conventional natural killer (cNK) cells and type 1 innate lymphoid cells (ILC1s). The main functions of liver cNK cells and ILC1s not only include directly killing target cells but also regulating local immune microenvironment of the liver through the secretion of cytokines. Uncovering the intricate mechanisms by which transcriptional factors regulate and influence the functions of liver cNK cells and ILC1s, particularly within the context of liver tumors, presents a significant opportunity to amplify the effectiveness of immunotherapies against liver malignancies. Using Ncr1-drived conditional knockout mouse model, our study reveals the regulatory role of Prdm1 in shaping the composition and maturation of cNK cells. Although Prdm1 did not affect the killing function of cNK cells in an in vivo cytotoxicity model, a significant increase in cancer metastasis was observed in Prdm1 knockout mice. Interferon-gamma (IFN-γ), granzyme B, and perforin secretion decreased significantly in Prdm1-deficient cNK cells and liver ILC1s. Single-cell RNA sequencing (scRNA-seq) data also provided evidences that Prdm1 maintains functional subsets of cNK cells and liver ILC1s and facilitates communications between cNK cells, liver ILC1s, and macrophages. The present study unveiled a novel regulatory mechanism of Prdm1 in cNK cells and liver ILC1s, showing promising potential for developing innovative immune therapy strategies against liver cancer.
Introduction
Group 1 ILCs consist of cNK cells and ILC1s (Spits et al., 2013; Peng et al., 2013), with distinct developmental trajectories and effect molecules (Bai et al., 2021). Both cNK cells and ILC1s play indispensable roles in combatting viral infections (Weizman et al., 2017), maintaining local immune homeostasis (Schuster et al., 2014), eradicating malignant transformed cells (Ducimetière et al., 2021), and fostering cross-talk with adaptive immunity (Fumagalli et al., 2022). cNK cells demonstrate potent cellular cytotoxicity, facilitating direct elimination of target cells (Kiessling et al., 1975). On the contrary, a defining attribute of ILC1s is their predominant cytokine-mediated functions, with limited cellular killing capacity (Nabekura et al., 2020). In a state of homeostasis, liver group 1 ILCs (CD45+CD3-NK1.1+NKp46+) can be discriminated into cNK cells and ILC1s by the differential expression of CD49a and CD49b (Peng et al., 2013): cNK cells are marked by the expression of CD49b, while liver ILC1s exhibit a distinctive positivity for CD49a. Tumor Necrosis Factor Related Apoptosis Inducing Ligand (TRAIL) is also expressed on liver ILC1s, but not on cNK cells (Takeda et al., 2001). Transcriptional factors (TFs) such as T-bet, Nfil3, PLZF, and ID2 are required for cNK cells and liver ILC1s development or generation of their progenitors (Townsend et al., 2004; Yokota et al., 1999; Gascoyne et al., 2009; Constantinides et al., 2014). Eomes is considered to be necessary for NK cell maturation, but not for ILC1s (Gordon et al., 2012). Liver environment facilitated T-bet expression in the early stage of NK cells development, which results in Eomes repression. The repression of T-bet is required for Eomes+ NK cells (Daussy et al., 2014). On the contrary, deficiency of Hobit results in the depletion of ILC1s, while only leaving little impact on liver NK cells (Mackay et al., 2016). These studies suggest that some TFs may, or usually, play different roles in NK cells and ILC1s.
During the development of liver cancers, the functions of immune cells are often inhibited, resulting in the formation of an immunosuppressive tumor microenvironment, and sometimes even systemic immune suppression (Lee et al., 2022). Transforming a poorly immunogenic tumor into a immunologically ‘hot’ tumor with a stronger immune response is one of the goals of many cancer immunotherapies (Galon and Bruni, 2019). Research targeting the immune system is showing increasing clinical promise in liver cancer treatment (Ruff et al., 2022). One of our recent studies also found that Toll-like receptor agonists can significantly enhance the anti-tumor effect of Sorafenib by reconstructing the tumor immune microenvironment and reshaping the vascular system (He et al., 2023). The anti-tumor activity of cNK cells in the liver is relatively well-established, whereas the relationship between liver ILC1 and tumors is still a topic of controversy. Clinical data showed that liver tumors with a higher infiltration of NK cells are associated with a better prognosis (Nersesian et al., 2021). Decreased NK cell activity is closely correlated with the malignancy of liver tumors and represents a significant risk factor for recurrence (Lee et al., 2021). The tumor microenvironment orchestrates the transformation of cNK cells into ILC1s in a TGF-β-dependent manner, resulting in their diminished capacity to control tumor growth and metastasis. This process ultimately promotes tumor immunoevasion (Gao et al., 2017). However, researches also revealed that, distinct from directly inhibiting tumor growth, the primary function of ILC1 is to suppress the seeding of metastatic tumor cells in liver tissue (Ducimetière et al., 2021). Comprehensive research in this field is essential to harness the precision of group 1 ILCs targeted immunotherapy for liver cancers.
The transcription factor network governs the function of group 1 ILCs and the balance between cNK and ILC1. Our research team, along with others, has observed that one of the transcription factor in TGF-β pathway, Smad4, promotes the shift in balance from cNK cells towards ILC1 in a TGF-beta-independent pathway, and simultaneously, it positively regulates the expression of another transcription factor, PR domain 1 (Prdm1/Blimp1; Wang et al., 2018; Cortez et al., 2017). Prdm1 plays a crucial role in the differentiation of B cells into plasma cells and the homeostasis of T cells (Keller and Maniatis, 1991; Kallies et al., 2006; Martins et al., 2006). The function and regulatory network of Prdm1 in NK cells is distinct from B cells and T cells, for its expression is independent of B-cell lymphoma 6 (Bcl-6) and Interferon Regulatory actor 4 (IRF4) but relies on T-bet. In the study that identified Prdm1 as an essential transcriptional factor for NK cell maturation (Kallies et al., 2011), no significant difference was observed in IFN-γ production or cytotoxicity between Prdm1-deficient and wild-type NK cells. Although Prdm1 expression is dependent on IL-15 in immature NK cells and can be further upregulated by IL-12 or IL-21, it plays a role in downregulating the expression of certain cytokine receptors, such as CD25 (IL-2Rα), consequently diminishing the responsiveness of NK cells to IL-2 (Akman et al., 2021). Notably, some studies even suggested that Prdm1 suppressed the secretion of IFN-γ in NK cells (Smith et al., 2010). These findings imply that, although Prdm1 promotes NK cell development, it may function as a negative regulator of their activity. However, direct evidence supporting the impact of Prdm1 on NK cell anti-tumor capabilities is still lacking. Furthermore, there has been no investigation into its influence on the homeostasis of cNK cells and ILC1s in the liver, nor has there been an exploration of the underlying mechanisms.
In the current study, we found that the deletion of Prdm1 in Ncr1+ cells resulted in an imbalance in the homeostasis of liver group 1 ILCs, with a shift towards cNK cells. The data also support the essential role of Prdm1 in the cancer surveillance mediated by cNK cells. While Prdm1 positively regulates genes associated with cellular cytotoxicity, it concurrently exerts inhibitory control over certain positive regulators of NK cell development and functionality, such as JunB. Using scRNA-seq, we have identified a subset of cNK cells within the liver characterized by elevated JunB expression. These cells exhibit decreased expression of genes associated with cellular cytotoxicity, and their abundance significantly increases following Prdm1 knockout. Furthermore, our data also support that Prdm1 promotes the cross-talk between group 1 ILCs and macrophages.
Results
Prdm1 promotes group 1 ILCs homeostasis and terminal maturation
Examination of 363 liver hepatocellular carcinoma (LIHC) patient samples from The Cancer Genome Atlas (TCGA) revealed a positive correlation between the expression of NK-cell-associated genes (Cursons et al., 2019) (NCR1, KLRB1, CD160, PRF1, etc.) and PRDM1 expression (Figure 1A). The patients are ordered from highest to lowest based on the expression of NK-PRDM1 for survival analysis (Figure 1B). Notably, patients exhibiting higher levels of NK-PRDM1 expression (above the median) experienced better survival outcomes compared to those with lower levels of NK-PRDM1 expression (below the median; Figure 1C). Similar results were also found in skin cutaneous melanoma (SKCM, n=454) and lung adenocarcinoma (LUAD, n=497) patients (Figure 1—figure supplement 1A-F). Patients within the highest quartile of NK-PRDM1 signature expression demonstrated enhanced overall survival, a result that achieved statistical significance in LUAD and SKCM patients (Figure 1—figure supplement 1G-I). These data suggested that PRDM1 in NK cells might be essential for immune surveillance in solid tumors, including liver cancer, and prompted us to investigate the function and mechanism of PRDM1 in NK cells and ILC1 within the context of liver cancer.
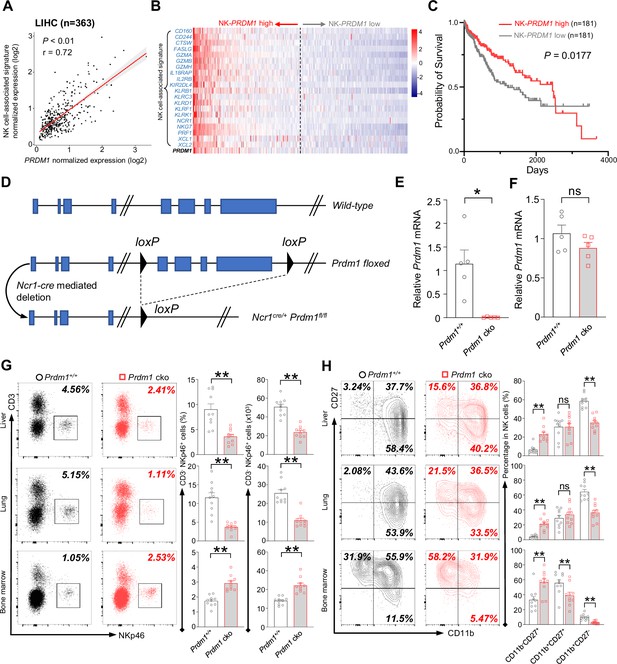
Prdm1 promotes NK cell homeostasis and terminal maturation.
(A) Correlation between the average expression of NK cell-associated signature and PRDM1 in LIHC (Liver Hepatocellular Carcinoma; n=363) patients sourced from TCGA datasets. (B) Heatmap of the ordered, z-score normalized expression values for PRDM1 and NK cell-associated genes in liver cancer patients. High and low expression of NK-PRDM1 signature are indicated. (C) Prognostic value of the NK-PRDM1 signature for overall survival of liver cancer patients comparing high and low samples with a median cutoff. (D) Schematic representation of the Prdm1-conditional knockout mouse model. Targeted exon 6–8 of Prdm1 (top) is flanked with loxP sites (middle). Ncr1-expressed Cre recombinase was used to generate the Prdm1 cko allele (bottom). (E and F) Real-time RT-PCR quantification of Prdm1 expression in NKp46+ cells (E) and splenocytes (F) to determine the presence of Prdm1 (n=5). (G) Representative flow cytometric plots (left) and quantification (right) of the proportion and absolute number of CD45+CD3-NKp46+ cells among lymphocytes in liver, lung, and bone marrow (n=10). (H) Representative flow cytometric plots (left) of the CD11b and CD27 expression within CD3-NK1.1+NKp46+CD49b+ cells in liver, lung, and bone marrow (n=10). Right panel showed the percentage of distinct maturation stages of NK cells. Data are presented as the mean ± SEM and were analyzed by two-tailed, paired t-test. Differences were evaluated between littermates. Each circle and square on graphs represents an individual mouse; P, p-value; r, pearson correlation coefficient; *, p<0.05; **, p<0.01, ns, not significant.
Ncr1-Cre mice were crossed with Prdm1fl/fl mice to generate Ncr1-cre Prdm1fl/fl mice, which specifically knockout exons 6–8 of Prdm1 (Shapiro-Shelef et al., 2003) in NKp46-positive cells (Figure 1D). The mice carrying Ncr1Cre/+Prdm1fl/fl were referred to as Prdm1 conditional knockout (Prdm1 cko) mice, and the mice carrying Ncr1+/+Prdm1fl/fl were referred to as Prdm1+/+ mice. To further validate the deletion of Prdm1 in NKp46+ cells of Prdm1 cko mice, CD3-NK1.1+NKp46+ cells were sorted and the expression of Prdm1 was measured by real-time RT-PCR. The expression of Prdm1 was almost undetectable in CD3-NK1.1+NKp46+ cells of Prdm1 cko mice, while was similar between total splenocytes in Prdm1+/+ and Prdm1 cko mice (Figure 1, E and F) indicating successful, and specifical knockout of Prdm1 in NKp46+ cells.
Proportion and absolute number of NK cells in blood, bone marrow, lung, liver, spleen, and lymph nodes were analyzed by flow cytometry. Compared with Prdm1+/+ mice, the percentage and absolute number of NK cells (CD45+CD3-NK1.1+NKp46+) among lymphocytes was decreased in all of these tissues, whereas increased number of NK cells were observed in bone marrow (Figure 1G; Figure 1—figure supplement 2A). NK cell terminal maturation can be divided into four stages according to the expression of CD11b and CD27 (Hayakawa and Smyth, 2006). Stage IV (CD11b+CD27-) NK cells show higher level of effector molecules than any other stages and are considered as the most mature NK cells (Chiossone et al., 2009). The maturation of cNK cells (gated by CD45+CD3-NK1.1+NKp46+CD49b+) from blood, bone marrow, lung, liver, spleen, and lymph nodes were assessed, based on the expression of CD11b and CD27. Compared with Prdm1+/+ mice, the proportion of the most mature CD11b+CD27- NK cells were significantly decreased in all of the analyzed tissues in Prdm1 cko mice (Figure 1H; Figure 1—figure supplement 2B). Killer cell lectin-like receptor subfamily G member 1 (KLRG1) is a lectin-like receptor which was considered as another marker of NK cell maturation (Huntington et al., 2007). In both Prdm1+/+ and Prdm1 cko mice, NK cells from the liver and lung had the highest expression of KLRG1, following by blood and spleen (Figure 1—figure supplement 2C). The lowest KLRG1 expression was observed in NK cells derived from lymph nodes and bone marrow, indicating the presence of the most immature NK cells in these tissues. Consistent with CD11b/CD27 based maturation analysis, a significant loss of KLRG1+ cells in NK cells was observed in Prdm1 cko mice (Figure 1—figure supplement 2C). Together, these data confirmed that Prdm1 is required for the terminal maturation of NK cells among various tissues.
Prdm1 is required for group 1 ILCs to control tumor metastasis
Two subpopulations of liver group 1 ILCs (gated by CD45+CD3-NK1.1+NKp46+) were further analyzed based on the expression of CD49a and CD49b (Figure 2A). Compared with Prdm1+/+ mice, the Prdm1 cko mice exhibited an increased percentage of cNK cells (CD49a-CD49b+) and reduced proportion of ILC1s (CD49a+CD49b-) (Figure 2A). Of note, the absolute number of both cNK cells and ILC1s were decreased in Prdm1 cko mice, with a more robustly reduction in ILC1s (Figure 2A), which underscored the crucial role of Prdm1 in maintaining the quantity of both liver cNK cells and ILC1s. Expression level of CD49b was slightly upregulated in Prdm1 cko cNK cells and NKp46+ cells in the liver and other tissues (Figure 1—figure supplement 2D and E). Increased CD49a expression was also observed in Prdm1 cko liver ILC1s, while it showed decreased expression in NKp46+ cells in the liver, bone marrow, and lymph nodes (Figure 1—figure supplement 2F and G). These results indicated the essential role of Prdm1 in maintaining the balance and hemostasis of cNK cells and ILC1s.
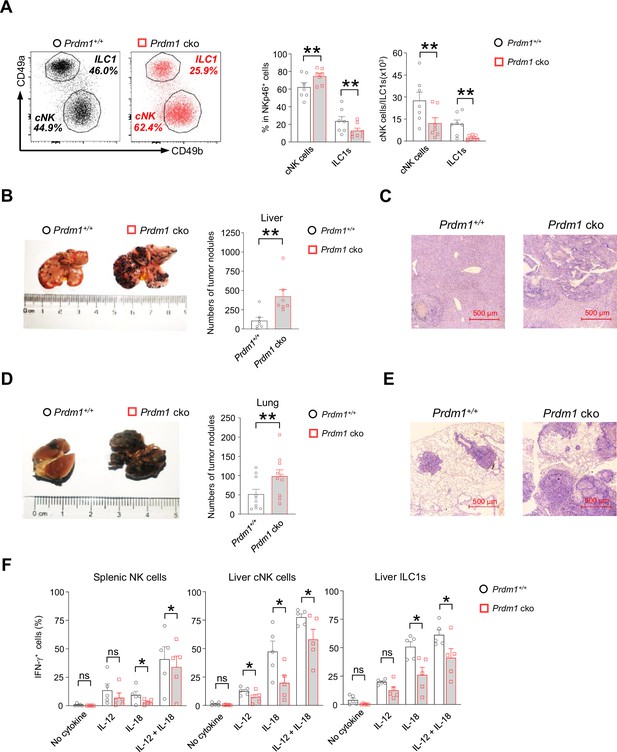
Prdm1 is required for group 1 ILCs to control tumor metastasis.
(A) Representative flow cytometric plots (left) of liver cNK cells (CD49a-CD49b+) and ILC1s (CD49a+CD49b-) from Prdm1+/+ and Prdm1 cko mice. The two bar graphs (right) quantitate the percentages and absolute numbers of cells respectively (n=7). (B and D) Representative image (left) and quantification (right) of tumor nodes on the livers (n=7) (B) and lungs (n=10) (D) of Prdm1+/+ and Prdm1 cko mice at day 14 or 21 after inoculation with B16F10 melanoma cells. (C and E) Representative histopathological images of liver (C) and lung (E) tissues stained by hematoxylin-eosin to detect tumor metastasis. Red bar indicates 500 μm distance under the microscope. (F) Liver cells and splenocytes were co-stimulated in the presence or absence of IL-12 and IL-18 for 12 hr. GolgiStop was added 4 hr before intracellular staining of IFN-γ. The graphs showed percentage of IFN-γ+ splenic cNK cells, liver cNK cells and ILC1s from Prdm1+/+ and Prdm1 cko mice (n=5). Data are presented as the mean ± SEM and were analyzed by two-tailed, paired t-test. Differences were evaluated between littermates. Each circle and square on graphs represents an individual mouse; P, p-value; *, p<0.05; **, p<0.01, ns, not significant.
The decreased quantity of NK cell homeostasis and maturation of cNK cells due to Prdm1 loss motivated us to further explore whether deficiency of Prdm1 impaired NK cell cytotoxicity. A B2M-deficient cell-based in vivo cytotoxicity assay was used to evaluate the effect of Prdm1 on the cytotoxicity of NK cells (Bix et al., 1991; Bern et al., 2019). B2M-deficient cells do not have detectable Major Histocompatibility Complex I (MHC-I) on the cell surface, making them the target of NK cells (Kärre et al., 1986). Healthy NK cells will reject B2M-deficient donor cells efficiently and the elimination was used to quantify the cytotoxicity of NK cells. Although significant impaired homeostasis and maturation of NK cells were observed in Prdm1 cko mice, no significant difference in the in vivo cytotoxicity assay were observed between Prdm1+/+ and Prdm1 cko mice (Figure 1—figure supplement 2H and I).
Besides their direct cytotoxic capabilities, NK cells' anti-tumor potential is also influenced by additional factors. These include their ability to counteract tumor-induced immune suppression and exhaustion, enhancing their effectiveness against cancer cells. Furthermore, NK cells can secrete cytokines that activate other immune cells, thereby orchestrating a broader immune response for the elimination of tumors. Moreover, the cytotoxicity assay, due to its relatively short duration, might not fully represent the anti-tumor activity of NK cells when continuously exposed to immune inhibitory signals in the tumor microenvironment. Therefore, we initiated an in vivo tumor model to further investigate the impact of Prdm1 on NK cell anti-tumor capability. B16F10 is a melanoma cell line with low expression of MHC-I, which was susceptible to NK cell killing and usually used to evaluate NK cell anti-tumor capacity (Viant et al., 2014; Cillo et al., 1987; Sathe et al., 2014). The B16F10 cells were intravenously (for lung metastasis) or intrasplenic (for liver metastasis) administrated in the mice. The melanoma nodes were quantified 3 (intravenous injection) or 2 (intrasplenic injection) weeks after tumor inoculation. Compared with Prdm1+/+ mice, deficiency of Prdm1 resulted in more metastasis nodules in both lung (~twofold) and liver (~fourfold) (Figure 2B and D). Histological analysis further confirmed the increased frequency of metastasis tumor foci in Prdm1 cko mice (Figure 2C and E). In agreement with the in vivo data, we also observed decreased IFN-γ secretion in Prdm1 cko mice-derived splenic cNK cells, liver cNK cells, and liver ILC1s when stimulated by IL-18 alone or IL-12/IL-18 (Figure 2F; Figure 2—figure supplement 1; Figure 7—figure supplement 1B), which indicated that Prdm1 is required for full activation of cNK cells and ILC1s in the context of IFN-γ production. These data implied that Prdm1 is indispensable for NK-cell-mediated tumor surveillance.
Bulk RNA-seq depicts Prdm1-mediated functions in cNK cells
Bulk RNA sequencing of splenic cNK cells (CD3-NK1.1+NKp46+) was conducted to uncover the molecular mechanisms by which Prdm1 regulates NK cell anti-tumor immunity (Figure 3A). Differentially expressed genes (DEGs) between Prdm1+/+ and Prdm1 cko mice were determined using a criterion of log2 (fold change)>0.5 and p<0.05. 445 DEGs were identified out of 17434 protein-coding genes, which consisted of 223 upregulated genes and 222 downregulated genes (Figure 3B).
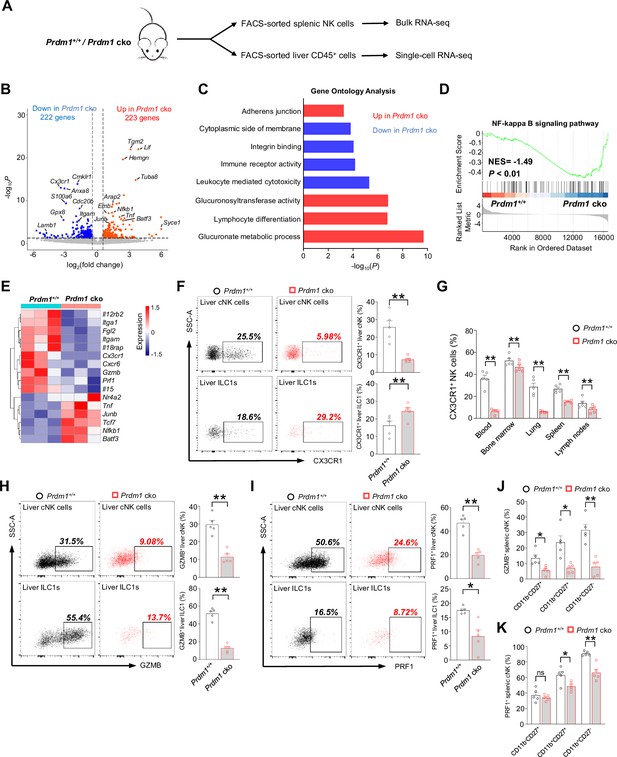
Bulk RNA-seq reveals Prdm1-mediated functions in splenic cNK cells.
(A) Splenic cNK cells and liver CD45+ cells were sorted from Prdm1+/+ and Prdm1 cko mice using flow cytometry, and prepared for bulk RNA-seq and single-cell RNA-seq analysis. (B) Volcano plot of the bulk RNA-seq differentially expressed genes (DEGs) (log2|fold change|>0.5; p<0.05) in splenic cNK cells between Prdm1+/+ and Prdm1 cko mice. Upregulated and downregulated genes in Prdm1 cko cells were highlighted in red and blue. (C) Enriched Gene Ontology (GO) terms of DEGs in Prdm1 cko cells compared Prdm1+/+ cells. The Enrichment gene set in upregulated (red) and downregulated (blue) genes were indicated in different colour. Bar length represents statistical significance. (D) Gene Set Enrichment Analysis (GSEA) showing the enrichment of NF-kappa B signaling pathway of DEGs in Prdm1 cko cells compared Prdm1+/+ cells. NES, normalized enrichment score. (E) Heatmap of selected genes from DEGs. Shown is z-score transformed expression of DEGs. (F) Representative flow cytometric plots (left) and cumulative data (right) of the percentage and absolute numbers of CX3CR1+ cells in liver cNK cells and ILC1s (n=5). (G) Quantification of CX3CR1+ cells in NK cells in blood, bone marrow, lung, liver, spleen, and lymph nodes (n=5). (H and I) Representative flow cytometric plots (left) and cumulative data (right) showing the proportion of GZMB+ (H) and PRF1+ (J) liver cNK cells and ILC1s from Prdm1+/+ and Prdm1 cko mice (n=5). (J and K) Proportion of GZMB+ (J) and PRF1+ (K) splenic NK cells at different maturation stages was analyzed by flow cytometry (n=5). Data are presented as the mean ± SEM and were analyzed by two-tailed, paired t-test. Differences were evaluated between littermates. Each circle and square on graphs represents an individual mouse; P, p-value; *, p<0.05; **, p<0.01, ns, not significant.
Gene Ontology (GO) analysis revealed the enrichment of glucuronate metabolism and lymphocyte differentiation in upregulated genes in Prdm1 cko mice derived NK cells (Figure 3C), both of which were associated with cellular growth and development. In contrast, leukocyte-mediated cytotoxicity, immune receptor activity, and integrin binding were enriched in the genes which decreased their expression level in in Prdm1 cko mice (Figure 3C). Gene Set Enrichment Analysis (GSEA) showed that NF-kappa B signaling pathway enriched in Prdm1-deficient cNK cells (Figure 3D), suggesting the potential targets by Prdm1 to regulate NK cell function. Increased expression of multiple TFs such as Junb, Batf3, Nfkb1, Tcf7, and Nr4a2 was observed in Prdm1 cko cNK cells, suggesting they might be suppressed by Prdm1 (Figure 3E). Downregulation of granzyme B (Gzmb), Perforin (Prf1) were observed in Prdm1-deficient NK cells (Figure 3E), implying decreased anti-tumor ability, which was consistent with increased melanoma metastasis in Prdm1 cko mice (Figure 2, B and D). CXCR6 and CX3CR1 was considered to play an important role in promoting the egress of NK cells from bone marrow. Decreased expression of Cxcr6 and Cx3cr1 in Prdm1 cko NK (Figure 3E) might be the reason for the increased quantity of NK cells in bone marrow and decreased number in peripheral tissues (Figure 1G). As a result of the reduced expression levels of Cxcr6 and Cx3cr1, NK cells may not be able to egress from the bone marrow and accumulated therein. Consistent with decreased production of IFN-γ after stimulated by IL-12/IL-18 (Figure 2F), decreased expression of Il18rap and Il12rb2 were observed in Prdm1 cko cNK cells (Figure 3E), implying impaired response to cytokine stimulation.
To confirm the result of RNA-sequencing, the expression of fractalkine receptor (CX3CR1), granzyme B and perforin were analyzed by flow cytometry. The percentage of CX3CR1+ cNK cells was significantly decreased in multiple tissues of Prdm1 cko mice, while the proportion of CX3CR1+ ILC1 was increased in the liver (Figure 3, F and G). Lower GZMB and PRF1 production was observed in Prdm1-deficient splenic cNK cells, liver cNK cells and ILC1s (Figure 3, H-K; Figure 3—figure supplement 1, A-I). Notably, the proportion of GZMB+ and PRF1+ cNK cells was decreased among almost all of the maturation stages of cNK cells (Figure 3, J and K). The relative mean fluorescent intensities (MFIs) of GZMB and PRF1 consistently show a reduction across all developmental stages in Prdm cko NK cells (Figure 3—figure supplement 1, H and I). Yet, no statistical difference of PRF1 was found within the CD11b-CD27+ and CD11b+CD27+ subsets, likely due to the relatively lower perforin levels in these populations (Figure 3—figure supplement 1I). These findings suggest that Prdm1 may directly influence cytotoxic molecule in NK cells, rather than impacting their anti-tumor abilities solely by affecting the maturation phenotype of Prdm1-deficient NK cells.
scRNA-seq reveals distinct properties of two clusters of liver group I ILCs following Prdm1 knockout
To further investigate the effect of Prdm1 in liver cNK cells and ILC1s and the changes in the hepatic immune microenvironment caused by the deficiency of Prdm1 in group 1 ILCs, single-cell RNA sequencing (scRNA-seq) was performed for liver CD45+ cells (Figure 4A; Figure 4—figure supplement 1A). Initial quality control revealed high-quality of cell purity, library assembly, and sequencing (Figure 4—figure supplement 1B). 10,978 cells passed the quality criteria and were selected for further analysis (6,161 from Prdm1+/+ mice, 4,817 from Prdm1 cko mice). Unsupervised clustering of all sequenced cells based on transcript signatures identified twelve distinct clusters (Figure 4—figure supplement 1C), including B cells, epithelial cells (ECs), CD4+ T cells, CD8+ T cells, NKT cells, ILC1s, cNK cells, dendritic cells (DCs), monocyte-derived macrophages (MDMs), Monocytes, Kupffer cells (KCs), Neutrophils, and a small number of undefined cells (Figure 4A). In Prdm1 cko mice, an increased proportion of MDMs, CD4+ T cells, CD8+ T cells, Monocytes, and DCs was observed, alongside a decreased proportion of ECs, cNK cells, ILC1s, and NKT cells (Figure 4—figure supplement 1D). Liver cNK cells and ILC1s were identified and discriminated based on the expression of surface markers and distinctive TFs (Figure 4—figure supplement 1, E and F). Compared with other clusters, cNK cells and ILC1s highly expressed Ncr1 and Klrb1c (NK1.1; Figure 4—figure supplement 1E). cNK cells expressed high levels of Itga2 (CD49b) and Eomes, while ILC1s had high levels expression of Itga1 (CD49a) and Tnfsf10 (Figure 4—figure supplement 1, F and G). Consistent with our flow cytometry data, both cNK cells and ILC1s have significant reduced proportion in Prdm1 cko mouse (Figure 4—figure supplement 1H). In group 1 ILCs from Prdm1 cko mice, there was an increase in the proportion of cNK cells accompanied by a decrease in ILC1s (Figure 4, B and C).
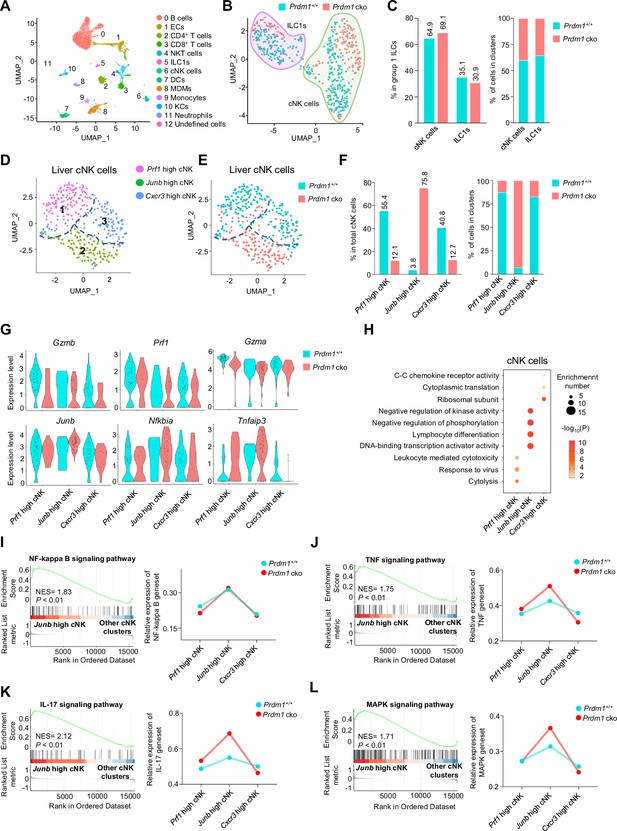
Different properties of cNK clusters following Prdm1 knockout.
(A) Uniform manifold approximation and projection (UMAP) visualization of liver CD45+ cells from Prdm1+/+ and Prdm1 cko mice. Twelve clusters were defined and indicated by distinct colours. Each dot represents a single cell. (B) UMAP visualization of liver cNK and ILC1 clusters. Cells were colored by genotypes (Prdm1+/+-blue; Prdm1 cko-red). (C) Percentages of cNK cells and ILC1s in total group 1 ILCs (left), and their distribution in each cluster (right). (D and E) UMAP visualization of three different liver cNK clusters from two mouse strains. (F) Proportions of cNK cells among total cNK cells (left; 211 cells in Prdm1+/+, and 141 cells in Prdm1 cko) and within clusters (right). (G) Violin plots showing the normalized expression of select genes in different cNK clusters. (H) Enriched GO term of marker genes in three cNK clusters. Dot size represents enriched gene number, and color intensity represents significance. (I–L) GSEA plots (left) depicting the enrichment of NF-kappa B (I), TNF (J), IL-17 (K), and MAPK (L) signaling pathway in Junb high cNK cluster compared with clusters of Prf1 high and Cxcr3 high cNK cells. Right panel showed dynamic relative expression of the given gene sets from cluster1 to cluster3 between Prdm1+/+ and Prdm1 cko. Dots represent the average expression of given gene set in each cell, which was calculated through the sum of normalized expression of each individual gene within the designated gene set in every single cell. NES, normalized enrichment score.
To better understand the specific function of Prdm1 in liver cNK cells and ILC1s, the two subpopulations of liver group 1 ILCs were further analyzed separately using unsupervised clustering and visualized by Uniform Manifold Approximation and Projection (UMAP; Figure 4, D and E). Based on the cluster specific gene expression signature (Figure 4—figure supplement 2A), the subpopulation of liver cNK cells were referred as ‘Prf1 high’, ‘Junb high’, and ‘Cxcr3 high’ cNK cells (Figure 4D), with different distribution in Prdm1 cko and Prdm1+/+ genotype mice (Figure 4F; Figure 4—figure supplement 2B).
The Prf1 high cNK cell cluster was defined by high expression of cytolysis-related genes, including Ncr1, Gzma, Gzmb, Prf1, and Fgl2 (Figure 4G; Figure 4—figure supplement 2, C and D), indicating the strong target-killing ability of this cluster. Although this cluster is present in both Prdm1 cko and Prdm1+/+ mice, there is a significant reduction in Prdm1 cko mice (Figure 4F), indicating the importance of Prdm1 in maintaining this group of cells. GO analysis further revealed the enrichment signatures of cytolysis, response to virus, and lymphocyte-mediated immunity in the genes upregulated in Prf1 high cNK cell cluster, further confirming the cytotoxic effects of this cluster (Figure 4H). These data underscore the crucial role of Prdm1 in maintaining NK cells with immune effector functions.
The Junb high liver cNK cell cluster distinguished themselves by higher expression of Junb compared to other clusters (Figure 4G; Figure 4—figure supplement 2, C and D). The predominant majority (92.98%) of Junb high liver cNK cells are derived from Prdm1 cko mice, with less than ten percent (7.02%) originating from Prdm1+/+ mice (Figure 4F). Many signal transduction elements, gene expression regulator, and transcriptional factors, such as Nfkbia, Tnfaip3, Nr4a1/2/3, Batf3, Fos, Fosb, Tcf7, and Kit were upregulated in the Junb high liver cNK cells (Figure 4G; Figure 4—figure supplement 2D). The expression of cytotoxicity related genes, such as Gzmb and Prf1, in Junb high cluster was lower than other cNK cell clusters (Figure 4G). GO analysis showed that genes upregulated in Junb high liver cNK cells enriched in cell differentiation, cell activation, and transcriptional regulation (Figure 4H). GSEA indicates that the NF-kappa B, IL-17, MAPK, and TNF signaling pathways were upregulated in this clusters (Figure 4, I-L). GSEA also showed that mitochondrial related pathways, such as mitochondrial protein, oxidative phosphorylation, and respiratory electron transport chain were suppressed in Junb high cNK cell cluster (Figure 4—figure supplement 2, E-G). Increased proportion of Junb high cluster in Prdm1 deficient cNK cells suggested impaired anti-tumor activity, which was consistent with more melanoma metastasis in Prdm1 cko mice and lower expression of cytotoxicity-related genes in splenic cNK cells based on bulk RNA-sequencing.
The Cxcr3 high cNK cell cluster was characterized with high expression of Cxcr3, Ccr2, and some genes encoding ribosomal subunits such as Rps7 (Figure 4—figure supplement 2A). Expression of tissue-resident markers Cd69 was also highly expressed in this clusters (Figure 4—figure supplement 2D). The enrichment of chemokine receptors in genes upregulated in the Cxcr3 high cluster implied a greater likelihood of this cluster being tissue-resident compared with other cNK cell clusters (Figure 4H). To further confirm tissue-resident properties of this clusters, we calculated the module score based on top30 DEGs in ILC1 versus cNK clusters, including Cxcr6, Itga1, Cd160, Cd226, etc. Cxcr3 high cNK clusters have the highest score among all cNK clusters (Figure 4—figure supplement 2H), indicating the similarity with liver ILC1s. In the tumor microenvironment, reports indicated that NK cells could transform into ILC1s (Gao et al., 2017). If this conversion of cNK cells into ILC1s also occurred under normal physiological conditions then Cxcr3 high cNK cell cluster might be the most susceptible to such transformation.
The significant enrichment of ribosomal subunits and cytoplasmic translation in Cxcr3 high cluster (Figure 4H) implied their distinct and active metabolic profile and the capability to mount immune responses. The remarkably decreased proportion of Cxcr3 high cNK cell cluster in Prdm1 cko mice (Figure 4F) emphasized the critical role of Prdm1 in maintaining this cluster of liver cNK cells, consistent with the flow cytometry result that showed an increase in the number of NK cells in the bone marrow and a decrease in NK cells in peripheral tissues (Figure 1G). This is consistent with flow cytometry data, which showed a decrease in the proportion of CX3CR1+ cNK cells across all tested organs following Prdm1 knockout. However, it is noteworthy that, in contrast to the trend in cNK cells, CX3CR1 expression was increased in liver ILC1s after Prdm1 knockout (Figure 3, F and G). This not only substantiates the involvement of Prdm1 in managing NK cell migration but also underscores its distinctive regulatory impacts on the chemokine receptor expressions within NK cells and ILC1 populations. Bulk RNA sequencing data also found that the expression of Cx3cr1 and Cxcr6 decreased in Prdm1 cko cNK cells (Figure 3E). These findings supported the hypothesis that Prdm1, through regulating chemokine receptor expression levels, influenced the distribution of NK cells in the bone marrow and peripheral tissues, particularly within the liver tissue.
Three clusters of ILC1s were identified from liver ILC1s (Figure 5A), comprising ‘Il7r high’, ‘Klra high’, and ‘Gzmb high’ ILC1s. Prdm1+/+ and Prdm1 cko ILC1s seemed to co-cluster largely and have minor difference within the proportion of clusters (Figure 5, B and C; Figure 5—figure supplement 1B). The first two clusters of ILC1s were characterized by higher expression of Il7r (CD127) and Klra5 separately, while the Gzmb high ILC1 cluster was identified by elevated expression of both Gzma and Gzmb (Figure 5D; Figure 5—figure supplement 1, A, C and D). Additionally, both Gzma and Gzmb expression were downregulated, and Junb was upregulated, in Prdm1 cko mice derived cNK cells and ILC1s compared to those from Prdm1+/+ mice (Figure 5E). The Il7r high ILC1s cluster was distinguishable from other ILC1 clusters by its high expression of Il7r, Il18r1 (IL18RA), and Ifng (IFN-γ) (Figure 5B; Figure 5—figure supplement 1C). The high expression of Il18r1 and Ifng in Il7r high ILC1s indicated this cluster of cells was highly responsive to IL-18 (Figure 5—figure supplement 1D). GSEA and GO analysis showed that IL-17, NF-kappa B, TNF, MAPK signaling pathway and T cell differentiation were activated in the Il7r high ILC1 cluster (Figure 5, F-H; Figure 5—figure supplement 1, E-G). Module scores, calculated based on the expression of feature genes within the Junb high cNK cell cluster, revealed a comparable Junb high signature expression pattern within the Il7r high ILC1 cluster (Figure 5I). Several ILC3 signature genes, such as Rora, Tmem176a, and Tmem176b (Robinette et al., 2015), highly expressed in this cluster (Figure 5—figure supplement 1D). Considering the close relationship between IL-17-mediated immunity response and ILC3 (Spits et al., 2013; Klose et al., 2013), it is plausible that Il7r high ILC1 cluster may be attributed, at least in part, to potential plasticity between ILC1 and ILC3 subsets.
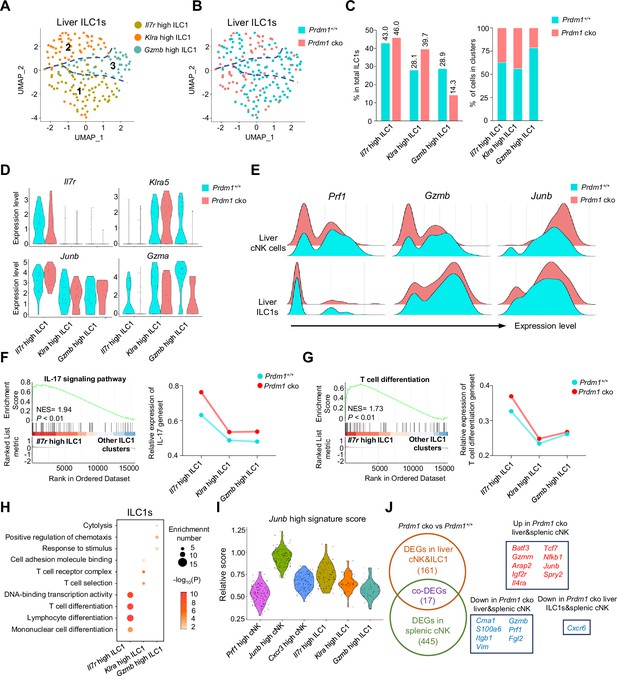
Different properties of ILC1 clusters following Prdm1 knockout.
(A and B) UMAP visualization of three different liver ILC1 clusters from two mouse strains. (C) Proportions of ILC1s among total ILC1s in different genotypes (left; 114 cells in Prdm1+/+, and 63 cells in Prdm1 cko) and within each cluster (right). (D) Violin plots showing the normalized expression of select genes in different ILC1 clusters. (E) Ridge plots showing the normalized expression of Gzmb, Prf1, and Junb in cNK and ILC1 clusters between Prdm1+/+ and Prdm1 cko cells. (F and G) GSEA plots (left) depicting the enrichment of IL-17 signaling pathway (F) and T cell differentiation (G) in Il7r high ILC1 cluster compared with clusters of Klra high and Gzmb high ILC1s. Right panel showed dynamic relative expression of the given gene sets from cluster1 to cluster3 between Prdm1+/+ and Prdm1 cko. Dots represent the average expression of given gene set in each cell, which was calculated through the sum of normalized expression of each individual gene within the designated gene set in every single cell. NES, normalized enrichment score. (H) Enriched GO term of marker genes in three ILC1 clusters. Dot size represents enriched gene number, and color intensity represents significance. (I) Violin plot showing the Junb high signature score for cNK cell and ILC1 clusters, calculated using the signature genes of Junb high cNK cluster. (J) Venn diagram showing overlapping and unique DEGs in comparisons within liver cNK cells, ILC1s and splenic cNK cells between Prdm1+/+ and Prdm1 cko(left), and 17 overlapped DEGs were shown at the right panel.
The second liver ILC1 cluster, characterized by high expression of Klra5 (Ly49E) and Klra7 (Ly49G), was designated as the Klra high ILC1 cluster (Figure 5D; Figure 5—figure supplement 1D). Notably, there was an elevated proportion of Klra high ILC1s in Prdm1 cko ILC1s (39.7%) compared to Prdm1+/+ ILC1 s (28.1%; Figure 5C). Liver Ly49E+ ILC1s have been identified as possessing greater cytotoxic potential and a more robust viral response compared to liver Ly49E- ILC1s (Chen et al., 2022). The Klra high cluster exhibited notably high expression of Ccl5 (Figure 5—figure supplement 1D). Previous research has underscored the pivotal role of CCL5, produced by both cNK cells and ILC1s, in facilitating the accumulation of DCs within the tumor microenvironment, thereby impeding tumor immune evasion, as highlighted in studies (Böttcher et al., 2018; Kirchhammer et al., 2022). The expression of Ccl5 was reduced in the Klra high cluster of Prdm1 cko ILC1s compared to Prdm1+/+ ILC1 s (Figure 5—figure supplement 1D), which could potentially have a detrimental impact on the ability of Klra high ILC1s to develop a connection between innate and adaptive immune responses.
The Gzmb high ILC1 cluster was identified according to the high expression of Gzma, Gzmb, and Fgl2 (Figure 5—figure supplement 1D) compared to other ILC1 clusters. GO analysis also revealed the enrichment in cytolysis and stimulus-response capacity (Figure 5H) of the Gzmb high ILC1 cluster. Consistent with the Prf1 high cNK cell cluster, the proportion of the Gzmb high cluster among liver ILC1s exhibited a considerable reduction in Prdm1 cko mice compared to Prdm1+/+ mice, and the expression of Gzma also downregulated in Prdm1 cko mice (Figure 5B). Previous reports showed that GzmA+ ILC1 constituted the main population of liver ILC1s at birth, with the potential target-killing ability (Di Censo et al., 2021; Friedrich et al., 2021). Within cNK cells, Il12rb2, Il18r1 and Il18rap was highly expressed in Prf1 high and Cxcr3 high cNK clusters (Figure 4—figure supplement 2I), indicating the IL-18 receptor expression correlated with the NK cell maturation. While in ILC1, these receptors mostly expressed on Il7r high and Gzmb high ILC1 clusters (Figure 5—figure supplement 1D). Significant decreased of Il18r1 expression in Prdm1 cko cNK cells and ILC1s may associated with the impaired ability to produce IFN-γ.
To investigate the universal transcriptional program between group 1 ILCs across liver and spleen, we have explored DEGs in Prdm1 cko liver cNK cells and ILC1s using our scRNA-seq data. Compared with liver ILC1s, more DEGs was observed in liver cNK cells, including Junb, Kit, Tcf7, Gzmb, Prf1, etc. (Figure 5—figure supplement 2, A and B). Through the integration of the bulk RNA-seq and scRNA-seq data, we identified 17 DEGs that are regulated by Prdm1 among liver cNK cells, splenic cNK cells, and liver ILC1s. Batf3, Junb, Tcf7, and Nfkb1 was upregulated, whereas Gzmb, Prf1, and Fgl2 downregulated, in both Prdm1 cko liver and splenic cNK cells (Figure 5J). Cxcr6 was downregulated in liver ILC1s and splenic cNK cells in Prdm1 cko mice (Figure 5J). Previous research found that spleen NK cells could be divided into three distinct groups based on their expression levels of CD27, CD62L, CD49a, and CD49b (Flommersfeld et al., 2021). CD27+CD62L- NK cells have remarkable high expression of Batf3, while it was only barely expressed in CD27+CD62L+ and CD27-CD62L+ NK cells (Flommersfeld et al., 2021). Based on the sequencing data published by Flommersfeld et al. (GSE180978), a notable negative correlation was observed between the expression levels of Prdm1 and Batf3 (Figure 5—figure supplement 2I). On top of that, our findings unveiled the negative regulatory influence of Prdm1 on Batf3 within both spleen and liver NK cells. This discovery highlights a potential upstream mechanism that may influence the hemostasis of the spleen NK cell subpopulations through Batf3.
We also compared the gene regulation patterns between Prdm1 and Hobit (homologue of Blimp1) based on two published scRNA-seq data (Friedrich et al., 2021; Yomogida et al., 2021). Following the knockout of Hobit, the DEGs were primarily identified within ILC1s. Conversely, after the knockout of Prdm1, a greater number of DEGs were observed in cNK cells. This indicates that Prdm1 likely possesses a broader range of target genes within cNK cells, whereas Hobit appears to have a more pronounced impact on gene expression within ILC1s (Figure 5—figure supplement 2, C-F). There are some overlaps between the downstream transcriptional profile of Prdm1 and Hobit in liver cNK cells and ILC1s (Figure 5—figure supplement 2, G and H). Specifically, genes such as Junb, Fosb, Tcf7, Kit, Gzmb, Prf1, and Cxcr6 were simultaneously upregulated or downregulated in both Prdm1 cko and HobitKO liver cNK cells or ILC1s, indicating the similar regulatory networks of Prdm1 and Hobit.
Prdm1 facilitates the intercellular communication between liver group 1 ILCs and macrophages
The reciprocal crosstalk between group 1 ILCs and macrophages plays a critical role in maintaining liver immune homeostasis and anti-cancer immune surveillance (Tu et al., 2008; Park et al., 2023). The scRNA sequencing analysis identified two well-established subpopulations of liver macrophages: the resident Kupffer Cells (KCs) and the Monocyte-Derived Macrophages (MDMs) (Figure 6, A-C; Figure 6—figure supplement 1A). When comparing the total proportion of macrophages within the immune cell population of the liver between Prdm1+/+ and Prdm1 cko mice, there is an increase in Prdm1 cko mice (Figure 6C). To confirm these findings, we utilized flow cytometry to define macrophages, including both KCs and MDMs, gating by CD45+Ly6G-F4/80+CD11b+ (Figure 6D). Our analysis showed that, following the deletion of Prdm1 in Group 1 ILCs, there is a significant increase in both the proportion and number of macrophages in the liver (Figure 6D).
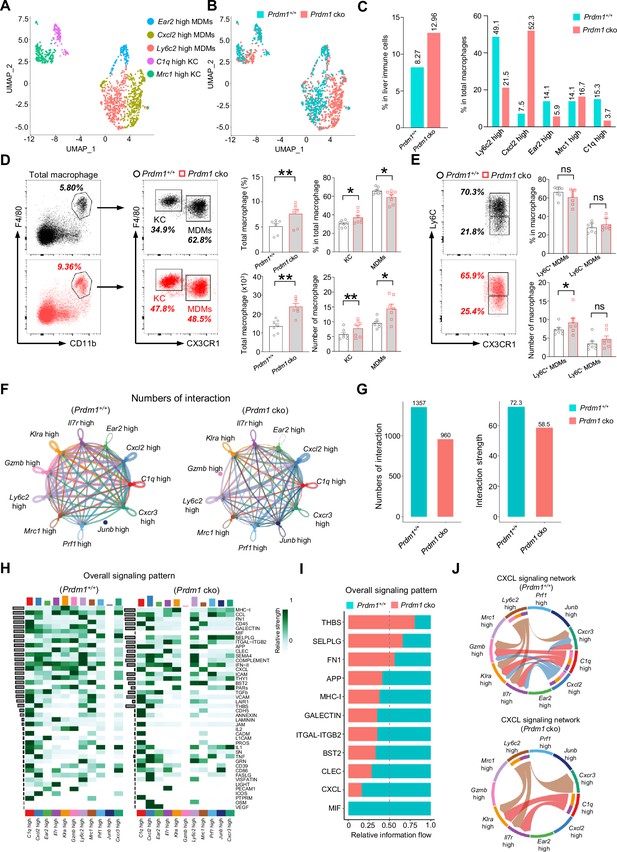
Prdm1 facilitates the intercellular communication between liver group 1 ILCs and macrophages.
(A and B) UMAP visualization of monocyte-derived macrophages (MDMs) and Kupffer cells (KCs) cluster (A) between Prdm1+/+ and Prdm1 cko (B). (C) Proportions of total macrophages in liver immune cells (left), and proportions of MDMs and KCs among total macrophages in different genotypes (510 cells in Prdm1+/+, and 624 cells in Prdm1 cko). (D) Representative flow cytometric plots (left) and cumulative data (right) of the percentage and absolute numbers of liver total macrophages (CD45+Ly6G-CD11b+F4/80+), MDMs (CX3CR1-), and KCs (CX3CR1+) between Prdm1+/+ and Prdm1 cko (n=7). (E) Representative flow cytometric plots (left) and cumulative data (right) of the percentage and absolute numbers of Ly6C- and Ly6C+ cells in MDMs. (F and G) Circle plots (F) and summary data (G) illustrating the interaction numbers and strength of significant enriched ligand–receptor pairs among cluster of liver cNK cells, ILC1s, and macrophages from Prdm1+/+ (left) and Prdm1 cko (right) cells. The thickness of the line indicates the number of enrich pairs, and the arrow reflects the direction of the interaction. (H) Heatmap of overall signaling pattern recognized from ligand-receptor pairs, which contained the sum of signaling from the sender and target cells. (I) Bar graphs showing the information flow in selected active signaling patterns between Prdm1+/+ and Prdm1 cko cells. Relative information flow was calculated as the sum of the communication probability in given signaling patterns. (J) Chord plot of the CXCL signaling interaction network among cluster of liver cNK cells, ILC1s, and macrophages between Prdm1+/+ and Prdm1 cko cells. Data are presented as the mean ± SEM and were analyzed by two-tailed, paired t-test. Differences were evaluated between littermates. Each circle and square on graphs represents an individual mouse; P, p-value; *, p<0.05; **, p<0.01, ns, not significant.
According to the transcriptional profile, liver macrophages were further clustered and labeled as ‘Ly6c2 high’; ‘Cxcl2 high’; ‘Ear2 high’ MDMs, and ‘Mrc1 high’; ‘C1q high’ KCs (Figure 6A, Figure 6—figure supplement 1, A-E). Increased proportion of MDMs and KCs was observed in Prdm1 cko cells, which was consistent with flow cytometry data (Figure 6—figure supplement 1B, and Figure 6D). Within MDMs clusters, Ly6c2 high MDMs mainly compose of Prdm1+/+ cells, while Prdm1 cko cells concentrated in Cxcl2 high cluster (Figure 6C). The scRNA-seq data revealed that following Prdm1 knockout in NKp46+ cells, there was a decrease in the proportion of KCs within the macrophage population, while the proportion of MDMs increased (Figure 6D). CX3CR1, a chemokine receptor, is extensively utilized to distinguish KCs and MDMs within macrophages. Cells expressing CX3CR1 are classified as MDMs, whereas those without CX3CR1 expression are categorized as KCs (Heymann et al., 2015). Using flow cytometry and assessing CX3CR1 expression, we determined the ratios of KCs and MDMs. In contrast to the scRNA-seq findings, flow cytometry indicates that following Prdm1 knockout in group 1 ILCs, there is a minor increase in the proportion of KCs within the total liver macrophages, and a decrease in the proportion of MDMs (Figure 6D; Figure 6—figure supplement 1B). This discrepancy might stem from the different bases of classification: scRNA-seq defines KCs based on gene expression profiles, whereas flow cytometry differentiates between KCs and MDMs using the single surface marker, CX3CR1. Analysis of the macrophage subsets identified by scRNA-seq reveals that, while MDM clusters generally show high CX3CR1 expression, there exists a subset within MDMs, labeled Mrc1 high, that also exhibits high levels of CX3CR1 (Figure 6—figure supplement 1C). Consequently, if flow cytometry solely employs CX3CR1 for differentiating between KCs and MDMs, it could result in disparities when compared to scRNA-seq data. Both KCs and MDMs has significantly increased in Prdm1 cko mice, which was consist with the scRNA-seq data (Figure 6—figure supplement 1, B and F). Despite the decrease in the proportion of Ly6c2 high MDMs in Prdm1 cko mice, the expression levels of Ly6c2 exhibited minimal variation between Prdm1+/+ and Prdm1 cko mice (Figure 6—figure supplement 1D). Intriguingly, within certain cellular subsets, notably the Ear2 high cluster, the Ly6c2 expression levels in Prdm1 cko mice were found to be higher than those in Prdm1+/+ mice. Additionally, we employed flow cytometry to examine Ly6C expression within the macrophages. Similar with the scRNA-seq findings, there were no notable differences in Ly6C expression levels between Prdm1+/+ and Prdm1 cko mice (Figure 6E; Figure 6—figure supplement 1G).
High-resolution interactions among liver cNK cells, ILC1s, and macrophages were established and compared between Prdm1+/+ and Prdm1 cko mice using the CellChat program (Jin et al., 2021). Interactions between ILC1s and total macrophages were higher than that between cNK cells and macrophages (Figure 6—figure supplement 2, A, C, E, and G). Cross-talk between liver group 1 ILCs with macrophages enriched in macrophage migration inhibitory factor (MIF), MHC-I, CXC chemokine ligand (CXCL), Thy-1 cell surface antigen (THY1), and C-type lectin (CLEC) pathways (Figure 6—figure supplement 2, B, D, F, and H). Although the quantity of macrophages significantly increases in Prdm1 cko mice, there is a significant decrease in the interaction number and interaction strength between liver group 1 ILCs and macrophages (~1.5 fold) in Prdm1 cko mice (Figure 6, F and G). The reduction of interaction mostly occurred in the cross-talk of ILC1-MDM and ILC1-KC, whereas no difference was observed in cNK-MDM and cNK-KC interaction (Figure 6—figure supplement 2, A-H). A reduction in the interaction of ligand-receptor, such as Mif-CD74, Cxcl16-Cxcr6, and Cxcl10-Cxcr3 was observed in Prdm1 cko mice compared to Prdm1+/+ mice (Figure 6—figure supplement 3). Compared to Prdm1+/+ mice, the information flow of CXCL and MIF pathways significantly decreased in Prdm1 cko mice (Figure 6, H and I; Figure 6—figure supplement 2, B, D, F, and H). These pathways play a crucial role in facilitating macrophage migration. The CXCL signaling was sent from Ly6c2 high Cxcl2 high MDMs and C1q high KC, targeting all ILC1 clusters and Cxcr3 high cNK cell clusters (Figure 6J). Of note, although the population of Cxcl2 high macrophage primarily comprised cells from Prdm1 cko mice, the interaction within the CXCL pathway between macrophages and group 1 ILCs was obviously less than Prdm1+/+ sample (Figure 6J). These changes could be linked to a decreased population of ILC1s and Cxcr3 high cNK cell cluster in Prdm1 cko mice, implying that the homeostasis of Cxcl2 high macrophages required sufficient signals from cNK cells and ILC1s. The impaired CXCL-CXCR interactions might subsequently lead to reduced recruitment and activation of group 1 ILCs and macrophages within the tumor microenvironment.
Prdm1 safeguards group 1 ILCs from exhaustion-like phenotypes in the tumor microenvironment
The suppression of mitochondrial related pathways in Junb high cNK cell cluster, along with a significant increase of this cNK cell cluster in Prdm1 cko mice, encouraged us to explore mitochondrial function through flow cytometry. MitoTracker, MitoSOX, and Tetramethylrhodamine methyl ester (TMRM) were used to assess the mitochondrial mass, superoxide production, and mitochondrial membrane potential. A substantial decrease in MFIs of MitoTracker was observed in Prdm1 cko splenic cNK cells, liver cNK cells, and liver ILC1s when compared to their Prdm1+/+ counterparts (Figure 7A). This observation aligns with the enrichment of downregulated genes from Prdm1-deficient sample in mitochondrial related pathway, as revealed by RNA sequencing data (Figure 4—figure supplement 2, D-F). There was no significant difference in MitoSOX and TMRM between Prdm1 cko and Prdm1+/+ mice (Figure 7, B and C), which suggested that the ATP synthesize capacity was minimally affected by Prdm1.
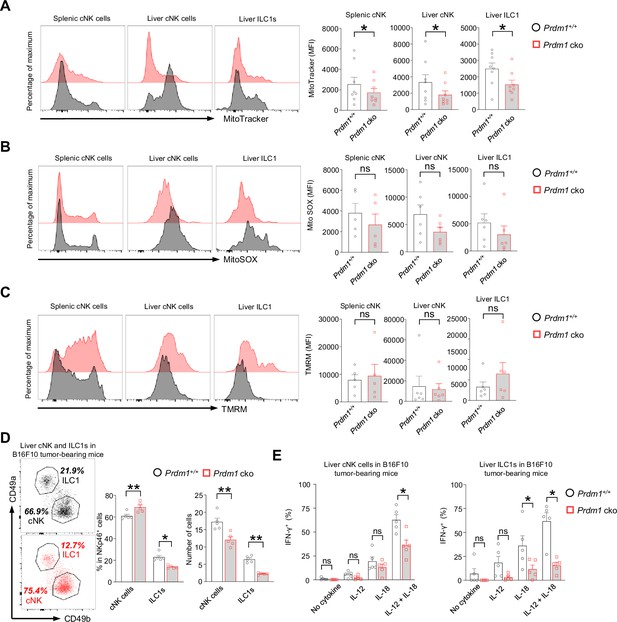
Prdm1 safeguards group 1 ILCs from exhaustion-like phenotypes in the tumor microenvironment.
(A–C) The Mitochondrial mass (MitoTracker Green staining; n=8) (A), Mitochondrial ROS (MitoSOX staining; n=5) (B), and Mitochondrial membrane potential (TMRM staining; n=5) (C) of splenic cNK cells, liver cNK cells and ILC1s were analyzed by flow cytometry. Representative flow cytometric plots (left) and cumulative data (right) showing the relative mean fluorescence intensities (MFIs) of each group. (D) Representative flow cytometric plots (left) and cumulative data (right) showing the percentage and absolute number of liver cNK cells and ILC1s in Prdm1+/+ and Prdm1 cko tumor-bearing mice at day 14 after inoculation with B16F10 melanoma cells via intrasplenic injection(n=5). (E) Percentages of IFN-γ+ liver cNK cells and ILC1s from Prdm1+/+ and Prdm1 cko tumor-bearing mice (n=5). Data are presented as the mean ± SEM and were analyzed by two-tailed, paired t-test. Differences were evaluated between littermates. Each circle and square on graphs represents an individual mouse; P, p-value; *, p<0.05; **, p<0.01, ns, not significant.
IFN-γ is a critical cytokine for NK cells mediated cancer surveillance (Takeda et al., 2011; Lin et al., 2021) and impaired production of IFN-γ was considered as a key hallmark of exhausted NK cells (Roe, 2022; Zhang et al., 2018). To evaluate the IFN-γ secreting capacity of liver cNK cells and ILC1s in tumor microenvironment, B16F10 tumor cells were inoculated to the liver via splenic injection and the IFN-γ levels in response to stimulation of IL-12 and/or IL-18 were assessed by flow cytometry (Figure 7—figure supplement 1A). The proportion changes of cNK cells and ILC1s in Prdm1 cko mice was similar with the no tumor-burden condition, while the number of both cNK cells and ILC1s have significant decreased in tumor-bearing liver (Figure 7D). Compared with Prdm1+/+ mice, significant deceased of IFN-γ+ cells were observed in Prdm1 cko mice liver cNK cells and ILC1s under the combinate stimulation of IL-12/IL-18 (Figure 7E; Figure 7—figure supplement 1B), which was more remarkable in liver ILC1s. Similar trends were observed when IL-12 or IL-18 was used alone, although only liver ILC1s showed a significant decrease in response to IL-18 stimulation (Figure 7E). These findings were consistent with the heavy tumor burden observed in Prdm1 cko mice.
Discussion
Prdm1 is a pivotal transcription factor that has attracted substantial research interest due to its role in lymphocytes. In a study involving systemic knockout combined with competitive transplantation, it was found that Prdm1 promotes NK cell maturation and the expression of Gzmb. On the contrary, the same study also found that NK cells with Prdm1 deficiency exhibit heightened proliferation, increased survival, enhanced migratory abilities toward tumors, and greater cytotoxicity against subcutaneously implanted RMAS tumors (Kallies et al., 2011). Using Ncr1-driven conditional knockout transgenic mice, which specifically delete Prdm1 in group 1 ILCs, we not only validated Prdm1’s positive regulation of NK cell maturation, but also demonstrated its indispensable role in NK cell anti-tumor activity. The compromised mitochondrial function and reduced IFN-γ, granzyme B, and perforin production appear to be potential contributing factors to Prdm1-mediated NK cell cancer surveillance. Reduction of CX3CR1+ NK cells in multiple tissues, and decreased expression of Cx3cr1 and Cxcr6 was observed in Prdm1 cko splenic cNK cells (Figure 3F), both of which are essential for NK cells egressing from bone marrow (Sciumè et al., 2011; Chea et al., 2015). Our results not only confirmed a decrease in cytotoxic molecules in Prdm1-deficient NK cells (Kallies et al., 2011) but also showed that the reduction in Gzmb and perforin is not solely attributable to the diminished maturation of these cells. Mature NK cells in bone marrow obtained the expression of CX3CR1 and acquired ability to enter the circulation (Sciumè et al., 2011; Chea et al., 2015). The quantity of cNK cells increased exclusively in the bone marrow, with reductions observed in all other tissues (Figure 1G), indicating Prdm1 might regulate chemokine receptors to facilitate the egression of cNK cells from the bone marrow to peripheral tissues. In addition, higher expression of Cxcr6 compared to cNK cells is also a key factor for liver tissue residence of ILC1s (Hudspeth et al., 2016). Decreased expression of Cxcr6 in Prdm1 deficient group 1 ILCs may also contribute to the balance shift towards cNK cells. Furthermore, although both liver NK cells and liver ILC1s require Prdm1 to maintain their quantity, liver ILC1s demonstrate a more pronounced dependency on Prdm1. However, it is currently widely believed that liver NK cells and liver ILC1s originate from different progenitors. It is worth noting that while we observed changes in the NK and ILC1 proportions after Prdm1 knockout, our data does not support the hypothesis that Prdm1 affects progenitor differentiation decisions, thereby influencing the fate selection of NK and ILC1. Further research may be needed to elucidate how Prdm1 regulates the balance between NK cells and ILC1s.
The scRNA-seq analysis reveals that both liver cNK cells and ILC1s can be further divided into three subgroups based on their gene expression patterns. JunB is a crucial transcriptional factor for the cytotoxic function of CD8+ T cells and NK cells. However, excessive Junb expression has been found to promote T cell exhaustion (Lynn et al., 2019). Our previous study showed that as NK cells mature, the expression level of Prdm1 increased while the expression level of Junb gradually decreased (Wang et al., 2018). The current study demonstrated that in NK cells, the expression level of Junb significantly increases upon the deletion of Prdm1, indicating that Junb expression is suppressed by Prdm1. As Junb expression decreases with NK cell maturation, and it is inhibited by the gradually increasing Prdm1 during maturation. This implies that constraining Junb expression is likely a fundamental prerequisite for NK cell maturation. However, the precise mechanism by which Prdm1 downregulates Junb in NK cells still needs further research. Furthermore, Junb high NK cells exhibit lower expression levels of cytotoxic genes and reduced mitochondrial-related signaling pathways. Mitochondria are pivotal organelles crucial for cellular metabolism. Disruptions in mitochondrial function have been linked to T Cell exhaustion, attributed to glycolytic reprogramming (Wu et al., 2023). Similarly, mitochondrial fragmentation has been closely associated with NK cell exhaustion (Zheng et al., 2019). However, the concept of NK cell exhaustion isn't as firmly established as it is for T cells. Exhausted NK cells should primarily exhibit diminished functions. This is characterized by a diminished ability to destroy tumor cells, a reduced capability to activate other components of the immune system, and compromised proliferation and survival rates. Additionally, this reduced functionality is associated with a decline in the expression of molecules responsible for cytotoxic activity, lower production of IFN-γ, and metabolic disturbances that may arise from mitochondrial dysfunction. While our current data is not sufficient to definitively classify these cells as exhausted NK cells, it supports that a subpopulation, referred to Junb high cluster, demonstrates an exhaustion-like phenotype. The significant increase in this cell population following Prdm1 knockout in NK cells may potentially be one of the reasons why Prdm1 cko mice lose their tumor-killing capacity. Whether the excessive expression of JunB in NK cells is also a contributing factor to their exhaustion, similar to T cells (Lynn et al., 2019), requires further investigation.
The scRNA-seq data revealed that Prdm1 plays distinct roles in regulating cNK cells and ILC1s despite being required for the quantity of both lineages. Specifically, Prdm1 appears to be more involved in promoting the resistance against exhaustion in cNK cells, whereas in ILC1s, it may play a role in the plasticity between ILC1s and ILC3s. In both our previous study and a study by Colonna et al. (Wang et al., 2018; Cheng et al., 2021), it was demonstrated that Smad4, a transcriptional factor involved in TGF-β signal pathway, upregulated Prdm1 in NK cells and depletion of Smad4 resulted in a decreased ratio of NK cells to ILC1s in the liver. However, knocking out Prdm1 in Ncr1+ cells increased the ratio of NK cells to ILC1s in liver group 1 ILCs (Figure 2A). These findings suggest the possibility of a Smad4-independent pathway through which Prdm1 promotes the maintenance of ILC1s, or that Prdm1 plays a more significant role in maintaining ILC1s compared to its role in NK cells.
Previous studies have identified Hobit and Prdm1 as central regulators instructing tissue-dependent programs and retention of diverse tissue-resident lymphocytes (Mackay et al., 2016; Friedrich et al., 2021; Yomogida et al., 2021). Liver ILC1s required Hobit, but not necessary for cNK cells (Ducimetière et al., 2021). Expression of Prdm1 was remarkably higher in cNK cells versus ILC1s (Mackay et al., 2016). While in our study, cNK cells and liver ILC1s reduced simultaneously in Prdm1 cko mice, and even more significant in ILC1s. This indicates that while Prdm1 is expressed at lower levels in ILC1s, its role in preserving the quantity of ILC1s may be more crucial. Thus, Prdm1 and Hobit may have parallel program in instructing ILC1s functional development and maturation. Prdm1 and Hobit directly bound and repressed Tcf7 (Mackay et al., 2016), which encoded TCF-1, a TF binding and limiting the activity of Gzmb regulatory element (Jeevan-Raj et al., 2017). Gzmb has been demonstrated directly bound and activated by Junb in NK cells, which suggested Gzmb expression regulated by multiple Prdm1/Hobit downstream signals (Wang et al., 2018). In human T cells, binding motif of JUNB was enriched in the binding sites of PRDM1 (Guo et al., 2022), indicating the essential role of PRDM1-JUNB axis during NK cell and T cell development. In Prdm1 deficient NK cells, we noted a decrease in Gzmb levels alongside with an elevation in Junb expression. This indicates that Prdm1 not only facilitates the expression of Gzmb in NK cells but also suppresses Junb expression. Given that Junb is recognized as a positive regulator of Gzmb (Babichuk and Bleackley, 1997), this presents a complex interplay that seems contradictory. Therefore, it is imperative to develop a theoretical framework to comprehensively understand and interpret this paradoxical relationship. Here, we hypothesize that during the early stages of NK cell development, JunB may enhance the expression of certain molecules associated with cytotoxicity, thereby aiding NK cells in acquiring the capacity to eliminate target cells. At these initial stages, Prdm1 level are comparatively low, thus exerting a weaker inhibitory effect on JunB. As NK cells mature, there is a progressive increase in Prdm1 level, which then exerts a stronger inhibitory influence on JunB, contributing to the reduced JunB level in fully matured NK cells. Consequently, this reduces the potential for NK cell exhaustion caused by elevated levels of JunB.
Chronic inflammation is a crucial factor in promoting tumorigenesis, and macrophages play a significant role in this process. Macrophages interact with both cNK cells and ILC1s. However, the TFs that regulate these interactions are poorly understood. Fortunately, recent advances in scRNA-seq technology and the CellChat software tool Jin et al., 2021 have allowed us to gain a better understanding of the Prdm1 signaling pathway in group 1 ILCs and its impact on macrophages at the transcriptional level. Increased metastasis in Prdm1 cko mice may be due to a decrease in the killing ability of NK cells, making them more prone to exhaustion, or it could be due to abnormalities in group 1 ILCs, leading to a decrease in the anti-tumor abilities of macrophages and an enhancement of their pro-tumor capabilities. It is worth noting that normal ILC1-macrophage interactions are more prevalent than the interaction between NK cells and macrophages (Figure 6—figure supplement 2). We also found that CXCL signaling-based interaction remarkably diminished in Prdm1 cko mice, which suggested CXCL-CXCR may contribute to keep the sufficient interaction between group 1 ILC and macrophage. Specifically, Prdm1 in group 1 ILCs may be critical in preventing the overactivation of macrophages that can lead to cancer development. Increased population of both MDMs and KCs in Prdm1 cko mice, as well as different distribution of macrophage clusters, indicating the homeostasis of macrophages require environment with functional cNK cells and ILC1s. Although not all macrophage phenotypes have been verified in this study, the present research serves primarily to offer initial insights and preliminary data for investigating the interactions between group 1 ILCs and macrophages. It aims to inspire further research into the role of transcription factors within the liver and the cancer microenvironment.
While our findings underscore the importance of Prdm1 in liver cNK cells and ILC1s tumor immune surveillance, it does not be validated in human NK cells, whereas previous studies have found that PRDM1 might inhibit the proliferation and function of human NK cells, or human NK cell derived cancer cells (Smith et al., 2010; Küçük et al., 2011). Furthermore, we did not provide an in-depth evaluation in multiple tumor models. Further research may provide deeper insight into the role of PRDM1 in the anti-tumor function of human NK cells, enabling a more direct investigation of its application in cancer therapies. Given its important role in preserving liver cNK cells and ILC1s functional heterogeneity, enhancing Prdm1 function in human NK cells could potentially be a strategy to promote NK-cell-based immunotherapy for cancer.
Methods
Mice
Prdm1fl/fl mice were purchased from The Jackson Laboratory. Ncr1-iCre and B2m-/- mice were purchased from Shanghai Model Organisms Center, Inc. Cre is targeted to the Ncr1 locus. Six- to twelve-week-old littermates were used for the experiment. All animal experiments were approved by Tianjin University Institutional Animal Care and Use Committee.
Cell lines
B16F10 melanoma cells were obtained from ATCC (CRL-6475) and negative for mycoplasma contamination.
Experimental metastasis model
For lung metastasis model, 0.3×106 B16F10 cells were intravenous injected into mice. Three weeks later, mice were euthanized for analysis. For liver metastasis model, mice were inoculation with 0.5×106 B16F10 via intrasplenic injection. Three weeks later, mice were euthanized for analysis. Lung and liver from tumor-bearing mice were fixed in 10% formalin and embedded in paraffin. Sections were stained with H&E.
In vivo cytotoxicity assay
Donor splenocytes harvested from B2m deficient (B2m-/-) mice were labeled with 5 µM CFDA-SE. Donor splenocytes harvested from B2m-adequate (B2m+/+) mice were labeled with 5 µM eF670. Labeled splenocytes from two mouse strains were mixed in a 1:1 ratio, and 1×107 cells in total were injected i.v. into Prdm1+/+ and Prdm1 cko mice. One day after administration, spleen and liver cells were isolated from recipient mice, and the population of labeled cells was analyzed by flow cytometry. Rejection % was quantified according to the following formula:
1-(%B2m+/+/%B2m-/-)Prdm1+/+ or Prdm1 cko ×100%.
Cell isolation
Mice were perfused with PBS by portal vein puncture before harvesting tissues. Liver and lung was digested with 0.05% collagenase II for 30 min and filtered through 70 µm cell strainers, and mononuclear cells were isolated after subjected to density gradient using 30% and 70% percoll. Spleen were also removed and pressed through 70 µm filterers to obtain splenocytes. Peripheral blood mononuclear cells were obtained from peripheral blood after lysis of red blood cells (Biolegend, 420301). Flushing femurs and mechanical disruption of inguinal lymph nodes were performed to obtain cells from bone marrow and lymph nodes.
Real-time RT-PCR
RNA was extracted from FACS-sorted NK cells or splenocytes using RNASimple Total RNA Kit (TIANGEN Biotech, 4992858) and subsequently reverse transcribed to cDNA with SuperScript VILO Master Mix (Thermo Fisher Scientific, 11755050) according to manufacturer’s instructions. qPCR was performed with SYBR Green Mix (Thermo Fisher Scientific, A25742) and CFX Opus 96 Real-Time PCR System (Bio-Rad). The relative mRNA expression level was calculated using 2-ddCt method. Primer sequences: Prdm1: 5’-CAGAAACACTACTTGGTACA-3’; 5’-GATTGCTTGTGCTGCTAA-3’.
Flow cytometry
Flow cytometry and cell sorting were performed with a Cytoflex S/SRT (Beckman Coulter). The following antibodies were used (all purchased from BioLegend unless otherwise indicated): CD45-PE-Cy7 (catalog 103114, clone 30-F11); CD3ε-PerCP-Cy5.5 (catalog 100327, clone 145–2 C11); NK1.1-BV421 (catalog 108741, clone PK136); CD335-AF647 (catalog 560755, clone 29A1.4, BD Bioscience); CD49a-PE (catalog 562115, clone Ha31/8, BD Bioscience); CD49b-FITC (catalog 108906, clone DX5); CD27-BV510 (catalog 124229, clone LG.3A10); CD11b-AF700 (catalog 101222, clone M1/70); KLRG1-APC (catalog 561620, clone 2F1, BD Bioscience); CD49a-BV421 (catalog 740046, clone Ha31/8, BD Bioscience); IFN-γ-PE (catalog 505807, clone XMG1.2); Granzyme B (catalog 372207, clone QA16A02); Perforin (catalog 154305, clone S16009A); CD49b-APC-Cy7 (catalog 108919, clone DX5); IgG-PE (catalog 402203, clone 27–35); CD335-BV510 (catalog 137623, clone 29A1.4);; CD3ε-APC-Cy7 (catalog 100330, clone 145–2 C11); CD11b-BV421 (catalog 101235, clone M1/70); CD3ε-BV421 (catalog 100335, clone 145–2 C11); CD3ε-FITC (catalog 553061, clone 145–2 C11, BD Bioscience); CX3CR1-PE (catalog 149005, clone SA011F11); Ly6G-PerCP-Cy5.5 (catalog 127615, clone 1A8); Ly6C-BV510 (catalog 108437, clone RB6-8C5); F4/80-APC-Cy7 (catalog 123118, clone BM8). For mitochondrial metabolic assay, fresh cells were incubated in 37℃ media for 30 min with 100 nM MitoTracker Green (catalog M7514, Invitrogen), 100 nM TMRM (catalog T668; Invitrogen), and 10 uM MitoSOX Red (catalog M36008; Invitrogen), respectively. Surface-stained after washing with PBS and then detected by flow cytometry. For intracellular IFN-γ staining, Cells freshly obtained from liver and spleen were stimulated 12 hr with or without cytokine. GolgiStop (BD Biosciences) was added 4 hr before intracellular staining.
Bulk RNA sequencing
Total RNA from FACS sorted splenic NK cells of Prdm1+/+ and Prdm1 cko mice was extracted by TRIzol reagent (Invitrogen), then reverse transcribed into cDNA. Library construction was prepared using Illumina mRNA Library kit, and sequencing was performed by the BGISEQ-500. Standard methods were used to analyze the RNA-seq data, including aligning the reads to the genome by HISAT2 (v2.1.0) (Kim et al., 2015), and gene expression values (Counts) were calculated using RSEM (v1.3.1) (Li and Dewey, 2011). DEGs were identified using DEseq2 (v1.4.5) (Love et al., 2014) with a cutoff of log2(fold change)>0.5 and p<0.05. The ‘clusterProfiler’ package (v4.4.4) (Wu et al., 2021) and gene sets from molecular signatures database (MSigDB) were used for GSEA and GO analysis. The heatmap was plotted using the ‘Pheatmap’ package (v1.0.12).
Single-cell RNA sequencing
FACS-sorted liver CD45+ cells with more than 80% cell viability were used for library preparation. Each sample contained cells from three Prdm1+/+ or Prdm1 cko mice. Gel Bead-in-Emulsions (GEMs) were generated using the 10 X Genomics Chromium system, which combinates Master Mix, Single Cell 3’ v3.1 Gel Beads, and Partitioning Oil with single cells. GEMs were mixed with cell lysate and reverse transcription reagent to produce full-length cDNA. After incubation, the GEMs were broken, and recovered cDNA were amplified via PCR. Fragmentation, End repair, A-tailing, and Adaptor Ligation were performed to obtain final libraries, which contain P5 and P7 sequences. The 3’ library was sequenced on Novaseq 6000 with approximately 50 k read pairs/cell sequencing depth. The ‘Seurat’ R package (v4.2.0) (Hao et al., 2021) was used for data analysis. Initial quality control was performed to filter out the low-quality cells and cell doublets. Cells with 200–5500 expressed genes and no more than 10% mitochondrial genes were considered for high-quality. Doublets were filtered with the ‘scDblFinder’ Package (v1.10.0) (Germain et al., 2021). After quality control, we totally recovered 6161 cells and 4817 cells from Prdm1+/+ and Prdm1 cko mice, respectively. Principal component analysis (PCA) was used for cluster analysis. The first 15 PCs were picked for clustering and further visualized by UMAP. Clusters-specific marker was defined using the ‘FindAllMarkers’ function, and clusters were manually annotated based on the top 30 or 15 markers. The ‘clusterProfiler’ package and gene set from MSigDB were used for GSEA and GO analysis. ‘CellChat’ package (v1.4.0) (Jin et al., 2021) was utilized to predict the cell-to-cell communication from scRNA-seq data.
TCGA datasets assay
The normalized gene expression and survival datasets of cancer patients collected in The Cancer Genome Atlas (TCGA) were downloaded from UCSC Xena (http://xena.ucsc.edu/) (Caicedo et al., 2020). NK cell-associated genes including CD160, CD244, CTSW, FASLG, GZMA, GZMB, GZMH, IL18RAP, IL2RB, KIR2DL4, KLRB1, KLRC3, KLRD1, KLRF1, KLRK1, NCR1, NKG7, PRF1, XCL1, XCL2, according to the previous study (Cursons et al., 2019). NK-cell-associated genes, together with PRDM1, constitute the NK-PRDM1 signature in this study. The mean expression of per genes was ordered from high-to-low and plotted by heatmap using the ‘Pheatmap’ package. The overall survival of patients in the high and low expression of NK-PRDM1 signature was selected for analysis. Kaplan-Meier curves were plotted by GraphPad Prism.
Statistics
For experiment results, two-tailed t tests were used to measure the continuous and normally distributed between the two independent groups. Paired t-tests were used to determine the statistical significance between two paired groups. Log-rank tests were used to compare the overall survival distribution between the two groups of patients. A p value less than 0.05 was considered significant and data were presented as mean ± SEM.
Study approval
All animal experiments were approved by The Tianjin University Animal Care and Use Committee. No human subjects were performed in this study (protocol: 00000000202010100023).
Data availability
Sequencing data have been deposited in Gene Expression Omnibus (GEO) dataset under accession codes GSE271233 (scRNA-seq) and GSE271380 (RNA-seq). All other data are available within the article and its supplementary information.
-
NCBI Gene Expression OmnibusID GSE271233. Prdm1 Positively Regulates Liver Group 1 ILCs Cancer Immune Surveillance and Preserves Functional Heterogeneity.
-
NCBI Gene Expression OmnibusID GSE271380. Prdm1 Positively Regulates Liver Group 1 ILCs Cancer Immune Surveillance and Preserves Functional Heterogeneity.
-
NCBI Gene Expression OmnibusID GSE185346. Hobit confers tissue dependent programs to type 1 innate lymphoid cells.
-
NCBI Gene Expression OmnibusID GSE163452. Effector differentiation downstream of lineage commitment in ILC1 is driven by Hobit across tissues.
-
NCBI Gene Expression OmnibusID GSE180973. Fate mapping of single NK cells identifies a type 1 innate lymphoid-like lineage that bridges innate and adaptive recognition of viral infection [bulk_RNAseq_steady_state].
References
-
PRDM1 decreases sensitivity of human NK cells to IL2-induced cell expansion by directly repressing CD25 (IL2RA)Journal of Leukocyte Biology 109:901–914.https://doi.org/10.1002/JLB.2A0520-321RR
-
Mutational analysis of the murine granzyme B gene promoter in primary T cells and a T cell cloneThe Journal of Biological Chemistry 272:18564–18571.https://doi.org/10.1074/jbc.272.30.18564
-
Inducible down-regulation of MHC class I results in natural killer cell toleranceThe Journal of Experimental Medicine 216:99–116.https://doi.org/10.1084/jem.20181076
-
Overcoming barriers to early disease interventionNature Biotechnology 38:669–673.https://doi.org/10.1038/s41587-020-0550-z
-
CXCR6 Expression is important for retention and circulation of ILC precursorsMediators of Inflammation 2015:368427.https://doi.org/10.1155/2015/368427
-
Ly49E separates liver ILC1s into embryo-derived and postnatal subsets with different functionsThe Journal of Experimental Medicine 219:e20211805.https://doi.org/10.1084/jem.20211805
-
MicroRNA-362 negatively and positively regulates SMAD4 expression in TGF-β/SMAD signaling to suppress cell migration and invasionInternational Journal of Medical Sciences 18:1798–1809.https://doi.org/10.7150/ijms.50871
-
Generation of drug-resistant variants in metastatic B16 mouse melanoma cell linesCancer Research 47:2604–2608.
-
A gene signature predicting natural killer cell infiltration and improved survival in melanoma patientsCancer Immunology Research 7:1162–1174.https://doi.org/10.1158/2326-6066.CIR-18-0500
-
T-bet and Eomes instruct the development of two distinct natural killer cell lineages in the liver and in the bone marrowThe Journal of Experimental Medicine 211:563–577.https://doi.org/10.1084/jem.20131560
-
Granzyme A and CD160 expression delineates ILC1 with graded functions in the mouse liverEuropean Journal of Immunology 51:2568–2575.https://doi.org/10.1002/eji.202149209
-
Approaches to treat immune hot, altered and cold tumours with combination immunotherapiesNature Reviews. Drug Discovery 18:197–218.https://doi.org/10.1038/s41573-018-0007-y
-
CD27 dissects mature NK cells into two subsets with distinct responsiveness and migratory capacityThe Journal of Immunology 176:1517–1524.https://doi.org/10.4049/jimmunol.176.3.1517
-
NK cell maturation and peripheral homeostasis is associated with KLRG1 up-regulationJournal of Immunology 178:4764–4770.https://doi.org/10.4049/jimmunol.178.8.4764
-
Inference and analysis of cell-cell communication using CellChatNature Communications 12:1088.https://doi.org/10.1038/s41467-021-21246-9
-
HISAT: a fast spliced aligner with low memory requirementsNature Methods 12:357–360.https://doi.org/10.1038/nmeth.3317
-
NK cells with tissue-resident traits shape response to immunotherapy by inducing adaptive antitumor immunityScience Translational Medicine 14:eabm9043.https://doi.org/10.1126/scitranslmed.abm9043
-
The liver-immunity nexus and cancer immunotherapyClinical Cancer Research 28:5–12.https://doi.org/10.1158/1078-0432.CCR-21-1193
-
Transcriptional repressor Blimp-1 regulates T cell homeostasis and functionNature Immunology 7:457–465.https://doi.org/10.1038/ni1320
-
TREM2 macrophages drive NK cell paucity and dysfunction in lung cancerNature Immunology 24:792–801.https://doi.org/10.1038/s41590-023-01475-4
-
Liver-resident NK cells confer adaptive immunity in skin-contact inflammationJournal of Clinical Investigation 123:1444–1456.https://doi.org/10.1172/JCI66381
-
Highlighting novel targets in immunotherapy for liver cancerExpert Review of Gastroenterology & Hepatology 16:1029–1041.https://doi.org/10.1080/17474124.2022.2150841
-
PRDM1/Blimp-1 Controls Effector Cytokine Production in Human NK CellsThe Journal of Immunology 185:6058–6067.https://doi.org/10.4049/jimmunol.1001682
-
Innate lymphoid cells--a proposal for uniform nomenclatureNature Reviews. Immunology 13:145–149.https://doi.org/10.1038/nri3365
-
IFN-γ production by lung NK cells is critical for the natural resistance to pulmonary metastasis of B16 melanoma in miceJournal of Leukocyte Biology 90:777–785.https://doi.org/10.1189/jlb.0411208
-
TLR-dependent cross talk between human Kupffer cells and NK cellsThe Journal of Experimental Medicine 205:233–244.https://doi.org/10.1084/jem.20072195
-
SMAD4 promotes TGF-β–independent NK cell homeostasis and maturation and antitumor immunityJournal of Clinical Investigation 128:5123–5136.https://doi.org/10.1172/JCI121227
-
Mitochondrial fragmentation limits NK cell-based tumor immunosurveillanceNature Immunology 20:1656–1667.https://doi.org/10.1038/s41590-019-0511-1
Article and author information
Author details
Funding
National Key Research and Development Plan of China (2022YFF1202901)
- Youwei Wang
National Natural Science Foundation of China (82372801)
- Youwei Wang
The Zhejiang Provincial Natural Science Foundation of China (LY21H150002)
- Zhouxin Yang
Natural Science Foundation of Tianjin (23JCYBJC01560)
- Xue Li
- Huaiyong Chen
- Youwei Wang
Science and Technology Planning Project of Tianjin Municipal Education Commission (2022YGYB14)
- Xue Li
Natural Science Foundation of Tianjin (23JCYBJC01370)
- Xue Li
- Huaiyong Chen
- Youwei Wang
Natural Science Foundation of Tianjin (21JCZDJC00430)
- Xue Li
- Huaiyong Chen
- Youwei Wang
The funders had no role in study design, data collection and interpretation, or the decision to submit the work for publication.
Acknowledgements
This work was supported by National Key Research and Development Plan of China (2022YFF1202901); National Natural Science Foundation of China (82372801); The Zhejiang Provincial Natural Science Foundation of China (LY21H150002); Natural Science Foundation of Tianjin (23JCYBJC01560, 23JCYBJC01370, 21JCZDJC00430); and Science and Technology Planning Project of Tianjin Municipal Education Commission (2022YGYB14).
Ethics
All animal experiments were approved by The Tianjin University Animal Care and Use Committee (protocol: 00000000202010100023).
Version history
- Sent for peer review:
- Preprint posted:
- Reviewed Preprint version 1:
- Reviewed Preprint version 2:
- Version of Record published:
Cite all versions
You can cite all versions using the DOI https://doi.org/10.7554/eLife.92948. This DOI represents all versions, and will always resolve to the latest one.
Copyright
© 2024, He, Gao, Wang et al.
This article is distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use and redistribution provided that the original author and source are credited.
Metrics
-
- 704
- views
-
- 79
- downloads
-
- 0
- citations
Views, downloads and citations are aggregated across all versions of this paper published by eLife.
Download links
Downloads (link to download the article as PDF)
Open citations (links to open the citations from this article in various online reference manager services)
Cite this article (links to download the citations from this article in formats compatible with various reference manager tools)
Further reading
-
- Immunology and Inflammation
Adipose tissue inflammation is now considered to be a key process underlying metabolic diseases in obese individuals. However, it remains unclear how adipose inflammation is initiated and maintained or the mechanism by which inflammation develops. We found that microRNA-802 (Mir802) expression in adipose tissue is progressively increased with the development of dietary obesity in obese mice and humans. The increasing trend of Mir802 preceded the accumulation of macrophages. Adipose tissue-specific knockout of Mir802 lowered macrophage infiltration and ameliorated systemic insulin resistance. Conversely, the specific overexpression of Mir802 in adipose tissue aggravated adipose inflammation in mice fed a high-fat diet. Mechanistically, Mir802 activates noncanonical and canonical NF-κB pathways by targeting its negative regulator, TRAF3. Next, NF-κB orchestrated the expression of chemokines and SREBP1, leading to strong recruitment and M1-like polarization of macrophages. Our findings indicate that Mir802 endows adipose tissue with the ability to recruit and polarize macrophages, which underscores Mir802 as an innovative and attractive candidate for miRNA-based immune therapy for adipose inflammation.
-
- Immunology and Inflammation
Chronic antigenic stimulation can trigger the formation of interleukin 10 (IL-10)-producing T-regulatory type 1 (TR1) cells in vivo. We have recently shown that murine T-follicular helper (TFH) cells are precursors of TR1 cells and that the TFH-to-TR1 cell transdifferentiation process is characterized by the progressive loss and acquisition of opposing transcription factor gene expression programs that evolve through at least one transitional cell stage. Here, we use a broad range of bulk and single-cell transcriptional and epigenetic tools to investigate the epigenetic underpinnings of this process. At the single-cell level, the TFH-to-TR1 cell transition is accompanied by both, downregulation of TFH cell-specific gene expression due to loss of chromatin accessibility, and upregulation of TR1 cell-specific genes linked to chromatin regions that remain accessible throughout the transdifferentiation process, with minimal generation of new open chromatin regions. By interrogating the epigenetic status of accessible TR1 genes on purified TFH and conventional T-cells, we find that most of these genes, including Il10, are already poised for expression at the TFH cell stage. Whereas these genes are closed and hypermethylated in Tconv cells, they are accessible, hypomethylated, and enriched for H3K27ac-marked and hypomethylated active enhancers in TFH cells. These enhancers are enriched for binding sites for the TFH and TR1-associated transcription factors TOX-2, IRF4, and c-MAF. Together, these data suggest that the TR1 gene expression program is genetically imprinted at the TFH cell stage.






