Impact of protein and small molecule interactions on kinase conformations
Figures
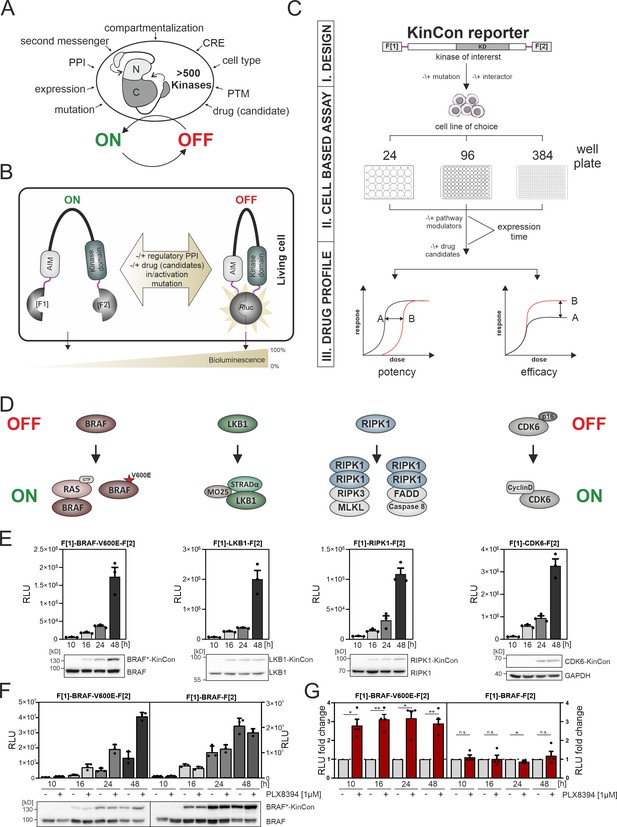
Kinase regulation and kinase conformation (KinCon) reporter technology features.
(A) Impact of indicated factors/features (e.g. protein-protein interactions [PPIs], post-translational modifications [PTM], cis-regulatory elements [CRE]) on the switch-like behavior of kinases. (B) Schematic representation of the KinCon reporter technology using the Renilla luciferase (RLuc) protein-fragment complementation assay (PCA) as it applies to kinases such as BRAF which contain auto-inhibitory modules (AIM); RLuc fragments 1 and 2 are N and C terminally fused to the kinase of interest (with interjacent linker in red) and are labeled with F[1] and F[2]. PPIs, drug (candidate) or small molecule binding, mutations and/or PTMs may convert the KinCon reporter into different conformation states. Protein movements are quantified through measuring alterations of bioluminescence signals upon RLuc substrate addition. (C) Shown is the workflow for the KinCon reporter construct engineering and analyses using KinCon technology. The kinase gene of interest is inserted into the multiple cloning site of a mammalian expression vector which is flanked by respective PCA fragments (F[1]-, -F[2]; KD, kinase domain) and separated with interjacent flexible linkers. Expression of the genetically encoded reporter in indicated multi-well formats allows to vary expression levels and define a coherent drug treatment plan. Moreover, it is possible to alter the kinase sequence (mutations) or to co-express or knock down the respective endogenous kinase, interlinked kinases, or proteinogenic regulators of the respective pathway. After systematic administration of pathway modulating drugs or drug candidates, analyses of KinCon structure dynamics may reveal alterations in potency, efficacy, and potential synergistic effects of the tested bioactive small molecules (schematic dose-response curves are depicted). (D) Simplfied schematic representation of the activation mechanisms of BRAF, LKB1, RIPK1, and CDK6 complexes (with indication of selected regulators or complex components) engaged in altering OFF (top) or ON (bottom) kinase states. (E) Representative KinCon experiments of time-dependent expressions of indicated KinCon reporter constructs in HEK293T cells are shown (mean ± SEM). Indicated KinCon reporters were transiently over-expressed in 24-well format in HEK293T cells for 10 hr, 16 hr, 24 hr, and 48 hr each. Immunoblotting show expression levels of endogenous kinases and over-expressed KinCon reporters. (F) Impact of 1 μM PLX8394 exposure (for 1 hr) on BRAF and BRAF-V600E KinCon reporters (HEK293T cells) is shown. Representative experiment of n=4 independent is presented. (G) RLuc PCA values have been normalized on the untreated conditions. The mean ± SEM of PLX8394 exposure on BRAF conformation opening and closing of n=4 experiments is shown. RLU, relative light units. Statistical significance for G: one-sample t-test (*p<0.05, **p<0.01, ***p<0.001).
-
Figure 1—source data 1
Raw unedited western blots shown in Figure 1.
- https://cdn.elifesciences.org/articles/94755/elife-94755-fig1-data1-v1.zip
-
Figure 1—source data 2
Uncropped and labelled western blots shown in Figure 1.
- https://cdn.elifesciences.org/articles/94755/elife-94755-fig1-data2-v1.pdf
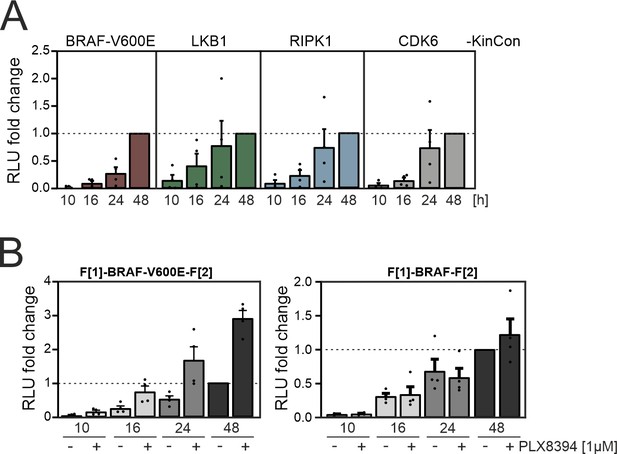
Time-dependent kinase conformation (KinCon) expression.
(A) Summary of n=4 independent experiments of time-dependent expressions of indicated KinCon reporter constructs in HEK293T cells is shown (mean ± SEM). BRAF-V600E, LKB1, RIPK1, and CDK6 KinCon reporters were transiently over-expressed in 24-well format in HEK293T cells for 10 hr, 16 hr, 24 hr, and 48 hr each. (B) Impact of 1 μM PLX8394 exposure for 1 hr on BRAF and BRAF-V600E KinCon reporters transiently over-expressed for indicated time frames in HEK293T cells is shown. Mean of n=4 independent experiments with ± SEM is presented.
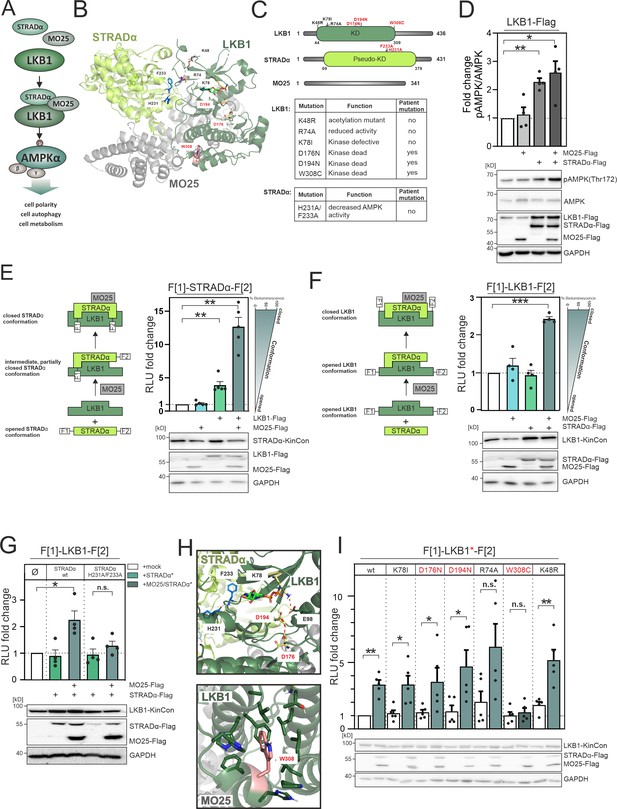
LKB1 emanating complexes and mutation-related kinase activity conformations in intact cells.
(A) Simplified representation of the LKB1-complex composition which promotes AMPKα signaling via phosphorylation at position Thr172. (B) Crystal structure of the LKB1-STRADα-MO25 complex (PDB code 2WTK, Zeqiraj et al., 2009a) representing a snapshot of trimeric complex assembly. The missense mutations we have analyzed are indicated in blue (STRADα) and pale yellow and rose (LKB1). The ATP analogue AMP-PNP is depicted in light green sticks. (C) Domain organization of human LKB1, STRADα, and MO25 (accession numbers: Q15831, Q7RTN6, Q9Y376) with indication of the kinase and pseudokinase domains (KD). Shown in red are tested missense mutations. These are summarized in the table together with their origin and assumed functions (Zubiete-Franco et al., 2019, Qing et al., 2022, Yang et al., 2019, Ui et al., 2014, Al Bakir et al., 2023, Islam et al., 2019, Boudeau et al., 2004). (D) Effect of co-expressions of indicated kinase complex components on AMPK phosphorylation (HeLa cells, 48 hr post transfection) (mean ± SEM, n=4 ind. experiments; 3x-Flag is indicated as flag). (E) Illustration of the kinase conformation (KinCon) reporter setup for STRADα KinCon measurements: Effect of LKB1-STRADα-MO25 complex formation on the STRADα KinCon reporter opening and closing (HEK293T cells, 48 hr post transfection). Expression corrected signals (STRADα-KinCon) are shown (mean ± SEM, n=4 ind. experiments). (F) KinCon reporter setup for LKB1 KinCon measurements: Effect of LKB1-STRADα-MO25 complex formation on the LKB1 KinCon reporter conformation. Expression corrected signals are shown (LKB1-KinCon; HEK293T cells, 48 hr post transfection) (mean ± SEM, n=5 ind. experiments). (G) LKB1-KinCon measurements upon co-expression of indicated proteins displaying the binding deficient STRADα mutations H231A/F233A (HF; see binding interface in B and H). Expression corrected signals are displayed (HEK293T cells, 48 hr post transfection) (mean ± SEM, n=4 ind. experiments). (H) Structure depiction highlights the localization of mutations conferring altered LKB1 functions. LKB1 residues K78, D176, and D194 (pale yellow sticks) are located within the catalytic cleft and in close proximity to AMP-PNP (light green sticks). (I) Impact of LKB1 missense mutations (three patient mutations D176N, D194N, and W308C and three ‘non-patient’ mutations K48R, R74A, K78I) on KinCon conformational changes upon co-expression of interactors. Expression corrected signals are displayed (HEK293T cells, 48 hr post transfection) (mean ± SEM, n=4 ind. experiments). Statistical significance for D, E, F, G, and I: one-sample t-test (*p<0.05, **p<0.01, ***p<0.001).
-
Figure 2—source data 1
Raw unedited western blots shown in Figure 2.
- https://cdn.elifesciences.org/articles/94755/elife-94755-fig2-data1-v1.zip
-
Figure 2—source data 2
Uncropped and labelled western blots shown in Figure 2.
- https://cdn.elifesciences.org/articles/94755/elife-94755-fig2-data2-v1.pdf
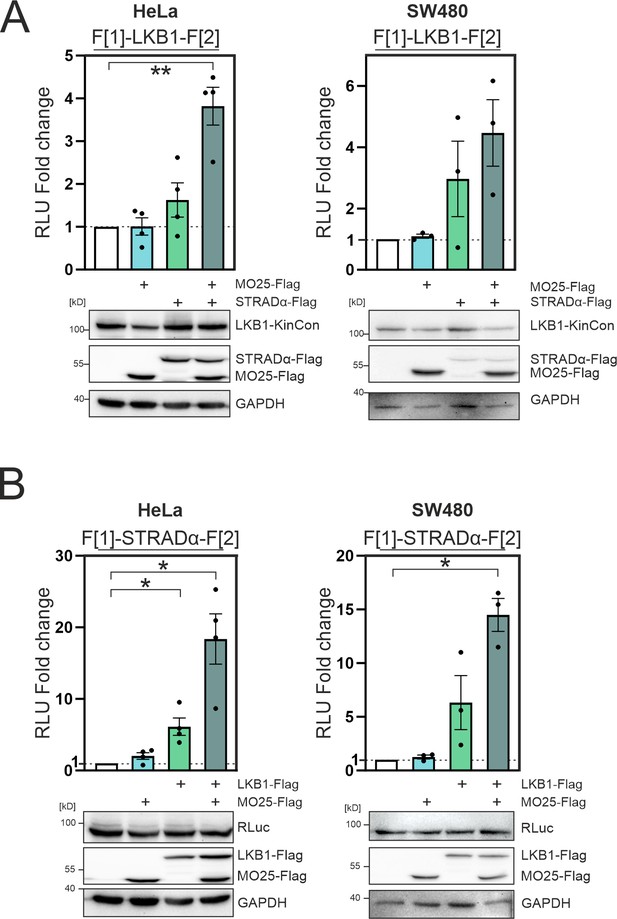
Complex formation of LKB1/STRADα/MO25 in HeLa and SW480 cells.
(A) Effect of LKB1-STRADα-MO25 complex formation on the LKB1 kinase conformation (KinCon) reporter (HeLa and SW480 cells). Bioluminescence signals were compared to the LKB1-KinCon signal (mean ± SEM, n=3–4). Representative western blots are shown below. (B) Effect of LKB1-STRADα-MO25 complex formation on the STRADα KinCon reporter (HeLa and SW480 cells). Bioluminescence signals were compared to the STRADα-KinCon signal (mean ± SEM, n=4 (HeLa) and n=3 (SW480)). Representative western blots are shown below. Statistical significance for A and B: one-sample t-test (*p<0.05, **p<0.01, ***p<0.001).
-
Figure 2—figure supplement 1—source data 1
Raw unedited western blots shown in Figure 2—figure supplement 1.
- https://cdn.elifesciences.org/articles/94755/elife-94755-fig2-figsupp1-data1-v1.zip
-
Figure 2—figure supplement 1—source data 2
Uncropped and labelled western blots shown in Figure 2—figure supplement 1.
- https://cdn.elifesciences.org/articles/94755/elife-94755-fig2-figsupp1-data2-v1.pdf
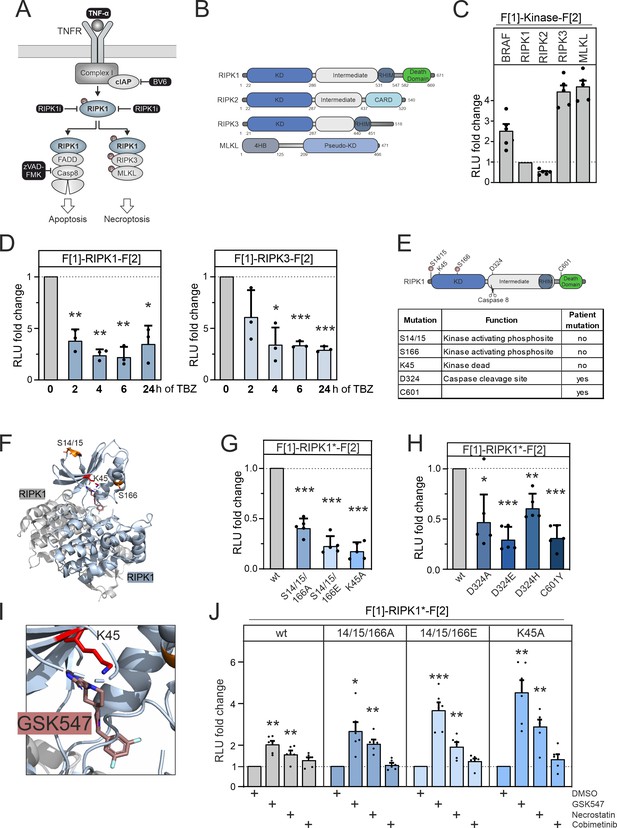
RIPK1 conformation dynamics.
(A) Simplified schematic representations of the activation pathways for apoptosis and necroptosis. Highlighted in black is the combination treatment termed TBZ (10 pg/ml TNFα, 10 nM BV-6, and 20 nM zVAD-FMK) that induces necroptosis. (B) Domain organization of human RIPK1 (accession number: Q13546), RIPK2 (accession number: O43353), RIPK3 (accession number: Q9Y572), and MLKL (accession number: Q8NB16). (C) Basal signals of indicated kinase conformation (KinCon) reporters following transient over-expression in HEK293T cells. Bars represent the RLU fold change relative to RIPK1 (mean ± SD, n=5 ind. experiments). (D) Time-dependent treatments using TBZ of HEK293T cells transiently expressing wild-type (wt) RIPK1 (left) and wt RIPK3 (right) KinCon reporters (expression corrected) (mean ± SD, n=3 ind. experiments). (E) Domain organization of RIPK1 displaying missense mutation sites. (F) 3D structure of RIPK1 dimers with functional mutations highlighted (PDB code: 6HHO, Wang et al., 2018). GSK547 is depicted as brown sticks. (G) KinCon reporter signals with/without mutations (S14/15/166A, S14/15/166E, K45A) were measured in a HEK293T RIPK1 knock-out (KO) cell line (expression corrected) (mean ± SEM, n=5 ind. experiments). (H) KinCon reporter signals of RIPK1 (patient loci: D324A, D324E, D324H, C601Y) were measured in HEK293T RIPK1 KO cells (expression corrected) (mean ± SD, n=5 ind. experiments). (I) 3D structure of RIPK1 with the inhibitor GSK547, which binds to an allosteric site in close proximity to the ATP-binding site (PDB code: 6HHO, Wang et al., 2018). (J) RIPK1 reporter signals with indicated mutations (described in G) upon exposure to GSK547 and Necrostatin 1 μM, and the MEKi Cobimetinib (1 μM, control experiment) or DMSO for 1 hr (mean ± SD, n=6 ind. experiments, HEK293T RIPK1 KO). Statistical significance for C–J: one-sample t-test (*p<0.05, **p<0.01, ***p<0.001).
-
Figure 3—source data 1
Raw unedited western blots underlying Figure 3 and shown in Figure 3—figure supplement 1.
- https://cdn.elifesciences.org/articles/94755/elife-94755-fig3-data1-v1.zip
-
Figure 3—source data 2
Uncropped and labelled western blots Figure 3 and shown in Figure 3—figure supplement 1.
- https://cdn.elifesciences.org/articles/94755/elife-94755-fig3-data2-v1.pdf
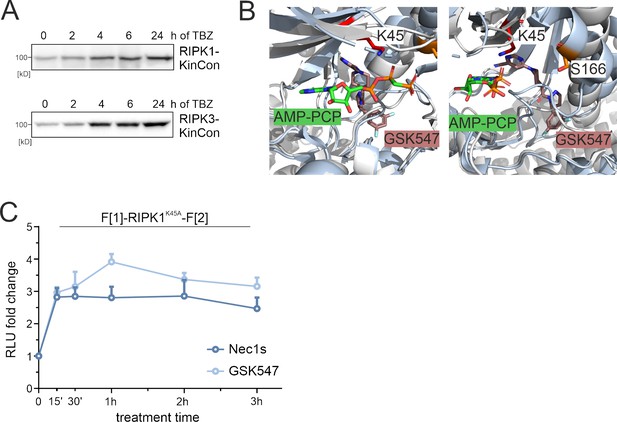
Small molecule effects on RIPK1.
(A) Representative western blots of RIPK1 and RIPK3 kinase conformation (KinCon) expressions after TBZ treatment for indicated time points. HEK293T cells were transfected with KinCon constructs and subjected to bioluminescence measurements after 48 hr. (B) 3D structure of RIPK1 with the inhibitor GSK547 (PDB code: 6HHO, Wang et al., 2018), which binds to an allosteric site in close proximity to ATP. For comparison, the structure of RIPK2 (light gray) in complex with the ATP analogue AMP-PCP (green sticks) is aligned (PDB code: 5NG0, Pellegrini et al., 2017). (C) Time-dependent treatment of RIPK1-K45A KinCon with two RIPK1i (GSK547 and Necrostatin, 1 μM each). The KinCon reporter was over-expressed in HEK293T RIPK1 KO cells for 48 hr. Points represent the mean RLU fold change relative to time point zero for n=3 independent experiments with ± SEM.
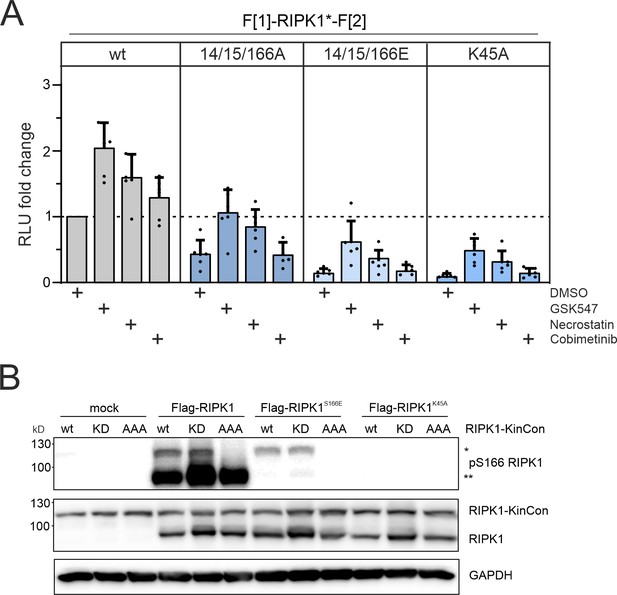
Conformation and phosphorylation of RIPK1.
(A) Conformation dynamics of indicated RIPK1 KinCon reporter upon exposure to the two RIPK1i (GSK547 and Necrostatin 1 μM), and the MEKi Cobimetinib (1 μM, control experiment) or DMSO for 1 hr. Bars represent the RLU fold change relative to the DMSO control of wild-type (wt) RIPK1 (mean ± SEM, n=6 ind. experiments, HEK293T RIPK1 KO). (B) RIPK1 KinCon auto-phosphorylation with co-expressed Flag-tagged RIPK1 constructs following expression in HEK293T RIPK1 KO cells. Indicated RIPK1 wt or mutant constructs were co-expressed for 48 hr and subjected to western blotting. KinCon RIPK1 constructs are annotated with single asterisk and Flag-tagged RIPK1 with double asterisks.
-
Figure 3—figure supplement 2—source data 1
Raw unedited western blots shown in Figure 3—figure supplement 2.
- https://cdn.elifesciences.org/articles/94755/elife-94755-fig3-figsupp2-data1-v1.zip
-
Figure 3—figure supplement 2—source data 2
Uncropped and labelled western blots shown in Figure 3—figure supplement 2.
- https://cdn.elifesciences.org/articles/94755/elife-94755-fig3-figsupp2-data2-v1.pdf
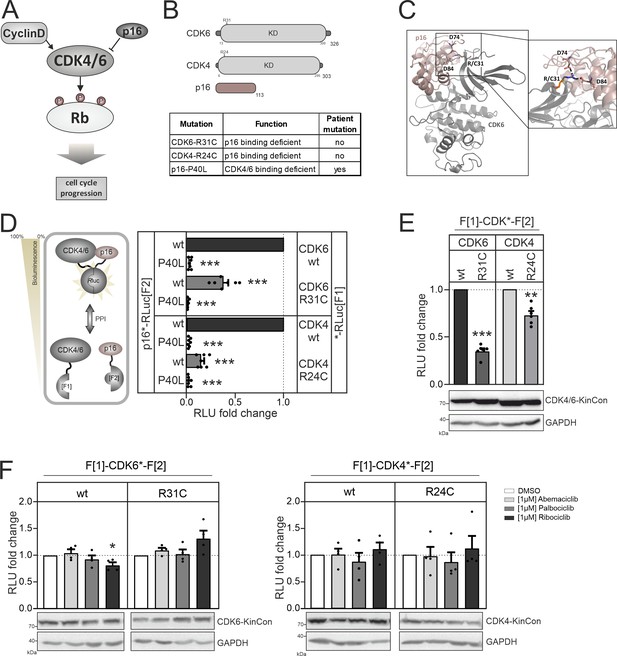
CDK4/6 interactions and conformations.
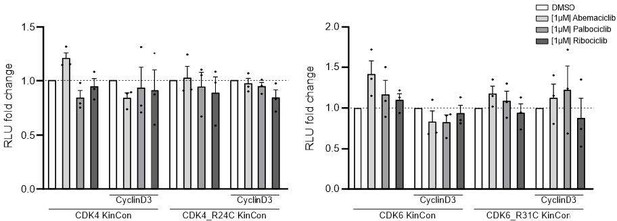
(A) Illustration of regulatory CDK4/6 interactions and Rb activation. (B) Domain organization of CDK4, CDK6, and p16INK4a; tested point mutations are listed. (C) 3D structure of CDK6 in complex with p16INK4a. Crucial amino acids involved in the interaction of the two proteins are highlighted. The R31C mutant is depicted in orange (PDB code: 1BI7, Russo et al., 1998). (D) Protein:protein interaction (PPI) reporter analyses of the kinases CDK4 and CDK6 with p16INK4a. Scheme illustrates CDK4/6 hetero-dimer formation with p16INK4a analyzed using a PCA RLuc PPI reporter system. PPI induces the complementation of RLuc PCA fragments promoting an increase in bioluminescence (HEK293T cells, 48 hr of transient reporter expression). Bars represent the RLU fold change of PPI in relation to wild-type CDK4/6:p16INK4a complex (mean ± SEM, n=7 ind. experiments). (E) Basal signal of CDK4/6 KinCon reporters with indicated mutations are shown (expressed for 48 hr in HEK293T cells) (mean ± SEM, n=6 ind. experiments). (F) Quantification of alterations of CDK4/6 KinCon reporter bioluminescence signals (HEK293T, expression for 48 hr) upon exposure to indicated CDK4/6i (1 μM) or DMSO for 3 hr (mean ± SEM, n=4 ind. experiments). Statistical significance for D–F: one-sample t-test (*p<0.05, **p<0.01, ***p<0.001).
-
Figure 4—source data 1
Raw unedited western blots shown in Figure 4.
- https://cdn.elifesciences.org/articles/94755/elife-94755-fig4-data1-v1.zip
-
Figure 4—source data 2
Uncropped and labelled western blots shown in Figure 4.
- https://cdn.elifesciences.org/articles/94755/elife-94755-fig4-data2-v1.pdf
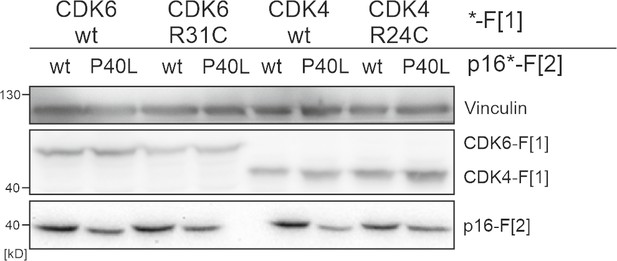
Expression of CDK4/6:p16INK4a PPI reporter constructs.
Exemplary western blot of CDK4/6 :p16INK4a PPI PCA. Immunoblot with respective antibodies show expression levels of RLuc PCA F[1] and RLuc PCA F[2] tagged expression constructs.
-
Figure 4—figure supplement 1—source data 1
Raw unedited western blots shown in Figure 4—figure supplement 1.
- https://cdn.elifesciences.org/articles/94755/elife-94755-fig4-figsupp1-data1-v1.zip
-
Figure 4—figure supplement 1—source data 2
Uncropped and labelled western blots shown in Figure 4—figure supplement 1.
- https://cdn.elifesciences.org/articles/94755/elife-94755-fig4-figsupp1-data2-v1.pdf
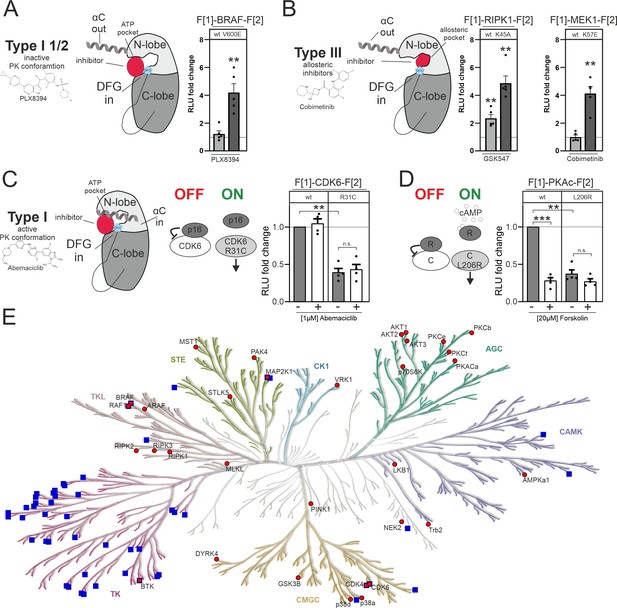
Impact of small molecules and protein interactions on kinase activity conformations.
(A+B) Depiction of molecular interactions of a type I 1/2 and type III kinase inhibitors with a kinase domain (N and C lobe). Impact of PLX8394, Cobimetinib and GSK547 on wild-type (wt) and mutated versions of BRAF, RIPK1, and MEK1 kinase conformation (KinCon) reporters. 48 hr post transfection HEK293T cells expressing respective reporter constructs were treated with indicated inhibitors for 1 hr (1 μM) followed by RLuc PCA analyses (mean ± SEM, n=4/5 ind. experiments). (C) Depiction of molecular interactions of a type I kinase inhibitor with a kinase domain (N and C lobe). Impact of Abemaciclib on indicated CDK6 kinase conformations (wt and p16INK4a binding deficient). 48 hr post transfection HEK293T cells expressing respective reporter constructs were treated with indicated inhibitors for 3 hr (1 μM) followed by RLuc PCA analyses (mean ± SEM, n=4 ind. experiments). (D) Bioluminescence measurement of PKAc wt and L206R KinCon reporters. HEK293T cells expressing the reporter were treated with 20 μM of Forskolin for 15 min followed by RLuc PCA analyses (mean ± SEM, n=4 ind. experiments). (E) Kinome tree displays kinases for which KinCon reporters have been generated (red dots). The blue squares highlight the kinases for which approved drugs are available. Generated with https://kinhub.org/kinmap/. Statistical significance for A–D: one-sample t-test (*p<0.05, **p<0.01, ***p<0.001).
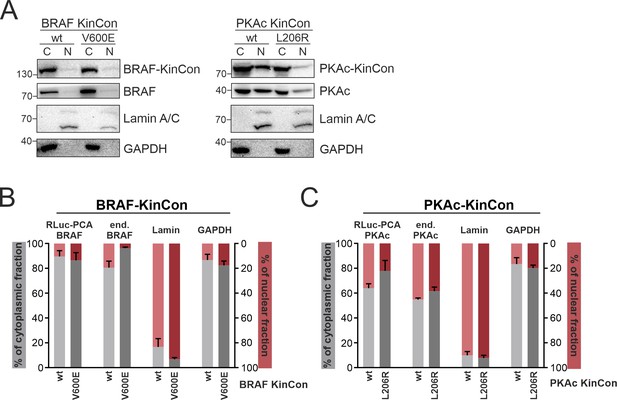
Subcellular localization of kinase conformation (KinCon) reporters.
BRAF and PKAc wild-type (wt) and mutated (BRAF-V600E, PKAc-L206R) reporters were over-expressed in HEK293T cells. (A) Representative western blot from the cytoplasmic (C) and nuclear (N) fractions. (B+C) Quantifications of the signals from n=3 ind. experiments (mean ± SEM).
-
Figure 5—figure supplement 1—source data 1
Raw unedited western blots shown in Figure 5—figure supplement 1.
- https://cdn.elifesciences.org/articles/94755/elife-94755-fig5-figsupp1-data1-v1.zip
-
Figure 5—figure supplement 1—source data 2
Uncropped and labelled western blots shown in Figure 5—figure supplement 1.
- https://cdn.elifesciences.org/articles/94755/elife-94755-fig5-figsupp1-data2-v1.pdf
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Cell line (Homo sapiens) | Kidney; Embryo | ATCC | CRL-3216 | HEK293T |
| Cell line (Homo sapiens) | Uterus; Cervix, Adenocarcinoma | ATCC | CCL-2 | HeLa |
| Cell line (Homo sapiens) | Large intestine; Colon Adenocarcinoma | ATCC | CCL-228 | SW480 |
| Transfected construct human | MA-T-gRNA-RIP1 | Addgene | 48138 | RIPK1-KO |
| Transfected construct human | Cas9-GFP pSp Cas9(BB)–2A-GFP | RIPK1-KO | ||
| Transfected construct human | pcDNA3.1-F[1] -LKB1-F[2] | LKB1-KinCon | ||
| Transfected construct human | pcDNA3.1- F[1]-LKB1-K78I-F[2] | LKB1-KinCon (K78I) | ||
| Transfected construct human | pcDNA3.1-F[1] -LKB1-D176N-F[2] | LKB1-KinCon (D176N) | ||
| Transfected construct human | pcDNA3.1-F[1] -LKB1-D194N-F[2] | LKB1-KinCon (D194N) | ||
| Transfected construct human | pcDNA3.1-F[1]-LKB1-R74A-F[2] | LKB1-KinCon (R74A) | ||
| Transfected construct human | pcDNA3.1-F[1]-LKB1-W308C-F[2] | LKB1-KinCon (W308C) | ||
| Transfected construct human | pcDNA3.1-F[1] -LKB1-K48R-F[2] | LKB1-KinCon (K48R) | ||
| Transfected construct human | pcDNA3.1-F[1] -STRADα-F[2] | STRADα-KinCon | ||
| Transfected construct human | pcDNA3.1-LKB1-3xFlag | LKB1-3xFlag | ||
| Transfected construct human | pcDNA3.1- STRADα–3xFlag | STRADα–3x Flag | ||
| Transfected construct human | pcDNA3.1- STRADα- H231A/F233A-3xFlag | STRADα–3xFlag (H231A /F233A) | ||
| Transfected construct human | pcDNA3.1- F[1]-MLKL-F[2] | MLKL- KinCon | ||
| Transfected construct human | pcDNA3.1- F[1]-RIPK1- S14/S15/S166A-F[2] | RIPK1-KinCon (S14A/S15A/S166A) | ||
| Transfected construct human | pcDNA3.1- F[1]-RIPK1- S14/S15/S166E-F[2] | RIPK1-KinCon (S14E/S15E/S166E) | ||
| Transfected construct human | pcDNA3.1- F[1]-RIPK1-K45A-F[2] | RIPK1-KinCon (K45A) | ||
| Transfected construct human | pcDNA3.1-F[1]-RIPK1-D324H-F[2] | RIPK1-KinCon (D324H) | ||
| Transfected construct human | pcDNA3.1- F[1]-RIPK1-D324A-F[2] | RIPK1-KinCon (D324A) | ||
| Transfected construct human | pcDNA3.1- F[1]-RIPK1-D324E-F[2] | RIPK1-KinCon (D324E) | ||
| Transfected construct human | pcDNA3.1- F[1]-RIPK1-C601Y-F[2] | RIPK1-KinCon (C601Y) | ||
| Transfected construct mouse | pcDNA3.1- F[1]-CDK6-F[2] | CDK6-KinCon | ||
| Transfected construct mouse | pcDNA3.1- F[1]-CDK6-R31C-F[2] | CDK6-KinCon (R31C) | ||
| Transfected construct mouse | pcDNA3.1- F[1]-CDK4-F[2] | CDK4-KinCon | ||
| Transfected construct mouse | pcDNA3.1- CDK6-F[1] | CDK6-PPI reporter | ||
| Transfected construct mouse | pcDNA3.1- CDK6-R31C-F[1] | CDK6-PPI reporter (R31C) | ||
| Transfected construct mouse | pcDNA3.1- CDK4-F[1] | CDK4-PPI reporter | ||
| Transfected construct mouse | pcDNA3.1- CDK4-R24C-F[1] | CDK4-PPI reporter (R24C) | ||
| Transfected construct mouse | pcDNA3.1- p16INK4a-F[2] | p16INK4a-PPI reporter | ||
| Transfected construct mouse | pcDNA3.1- p16INK4a-P40L-F[2] | p16INK4a-PPI reporter (P40L) | ||
| Transfected construct human | pcDNA3.1- F[1]-MEK1-F[2] | MEK1-KinCon | ||
| Transfected construct human | pcDNA3.1- F[1]-MEK1-K57E-F[2] | MEK-KinCon (K57E) | ||
| Transfected construct human | pcDNA3.1- F[1]-PKAc-F[2] | PKAc-KinCon | ||
| Transfected construct human | pcDNA3.1- F[1]-PKAc-L206R-F[2] | PKAc-KinCon (L206R) | ||
| Transfected construct human | pcDNA3.1- MO25-3xFlag | MO25-3xFlag | ||
| Transfected construct human | pcDNA3.1- F[1]-BRAF-F[2] | BRAF-KinCon | ||
| Transfected construct human | pcDNA3.1- F[1]-BRAF-V600E-F[2] | BRAF-KinCon (V600E) | ||
| Transfected construct human | pcDNA3.1- F[1]-RIPK1-F[2] | RIPK1-KinCon | ||
| Transfected construct human | pcDNA3.1- F[1]-RIPK2-F[2] | RIPK2-KinCon | ||
| Transfected construct human | pcDNA3.1- F[1]-RIPK3-F[2] | RIPK3-KinCon | ||
| Antibody | GAPDH Rabbit monoclonal | Cell Signaling | 2118 | 1:10000 |
| Antibody | AMPKα Rabbit monoclonal | Cell Signaling | 2532 | 1:1000 |
| Antibody | Phospho- AMPKα(Thr172) Rabbit monoclonal | Cell Signaling | 2535 | 1:1000 |
| Antibody | LKB1 Rabbit monoclonal | Cell Signaling | 3047 | 1:1000 |
| Antibody | Vinculin Rabbit monoclonal | Cell Signaling | 4650 | 1:1000 |
| Antibody | RIP XP Rabbit monoclonal | Cell Signaling | 3493 | 1:1000 |
| Antibody | RIP3 Rabbit monoclonal | Cell Signaling | 13526 | 1:1000 |
| Antibody | CDK6 Mouse monoclonal | Cell Signaling | 3136 | 1:1000 |
| Antibody | FLAG-M2 Mouse monoclonal | Sigma-Aldrich | F3165 | 1:1000 |
| Antibody | Renilla Luciferase Rabbit monoclonal | Abcam | Ab185926 [EPR17792] | 1:10000 |
| Antibody | Renilla Luciferase clone 1D5.2 Mouse monoclonal | Millipore | MAB4410 | 1:1000 |
| Drug, chemical compound | Inhibitor | MCE Med Chem Express | HY-50767 | Palbociclib PD 0332991 |
| Drug, chemical compound | Inhibitor | MCE Med Chem Express | HY-16297A | Abemaciclib LY2835219 |
| Drug, chemical compound | Inhibitor | MCE Med Chem Express | HY-15777 | Ribociclib LEE011 |
| Drug, chemical compound | Inhibitor | MCE Med Chem Express | HY-18972 | PLX8394 |
| Drug, chemical compound | Inhibitor | MCE Med Chem Express | HY-114492 | GSK-547 |
| Drug, chemical compound | Inhibitor | MCE Med Chem Express | HY-14622A | Necrostatin 1 S |
| Drug, chemical compound | Inhibitor | MCE Med Chem Express | HY-13064 | Cobimetinib |
| Drug, chemical compound | Inhibitor | MCE Med Chem Express | HY-P7090A | TNFα |
| Drug, chemical compound | Inhibitor | MCE | HY-16658 | z-VAD(OMe)-FMK |