β-Catenin-NF-κB-CFTR interactions in cholangiocytes regulate inflammation and fibrosis during ductular reaction
Figures
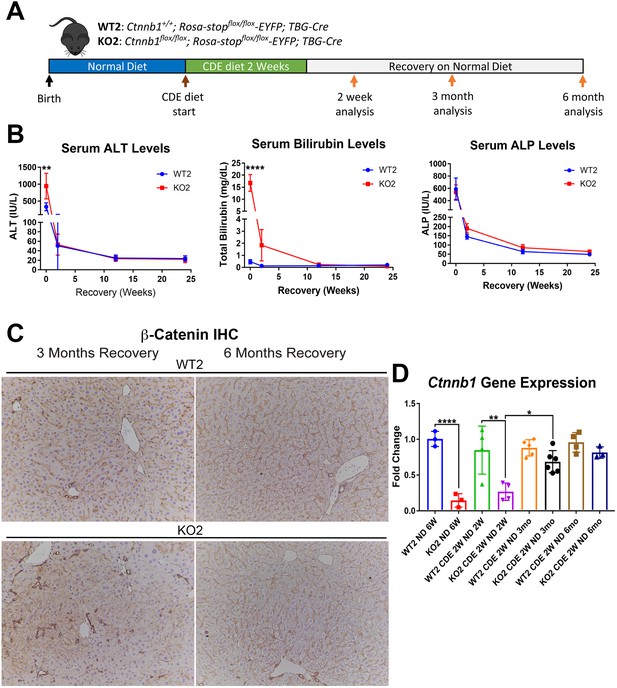
Comparable recovery of WT2 and KO2 on normal diet after initial 2-week choline-deficient, ethionine-supplemented (CDE) diet injury, along with repopulation of KO2 livers with biliary epithelial cell (BEC)-derived β-catenin-positive hepatocytes.
(A) Experimental design showing WT2 and KO2 on 2 weeks of CDE diet and recovery on normal diet for up to 6 months with analysis at intermediate time points as indicated. (B) Serum alanine aminotransferase (ALT), bilirubin, and alkaline phosphatase (ALP) in the two groups over time (one-way ANOVA, **p<0.01, ****p<0.0001, n = 3–6 per group). (C) β-Catenin immunohistochemistry in WT2 and KO2 mice at 3 months and 6 months of recovery showing β-catenin-positive BECs and hepatocytes in KO2 and WT2 (100×). (D) Ctnnb1 gene expression in WT2 and KO2 mice during recovery from CDE diet (one-way ANOVA, *p<0.05, **p<0.01, ****p<0.0001. n = 3–6 per group; individual animal values represented by dots).
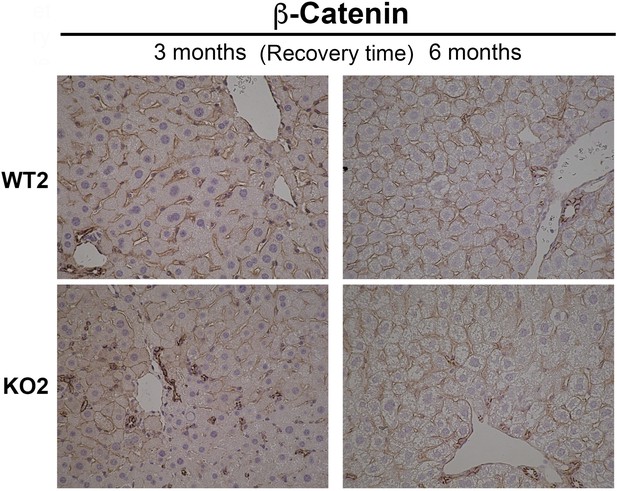
β-Catenin immunohistochemistry in WT2 and KO2 mice at 3 months and 6 months of recovery showing β-catenin-positive biliary epithelial cells (BECs) and hepatocytes in KO2 and WT2 (200×).
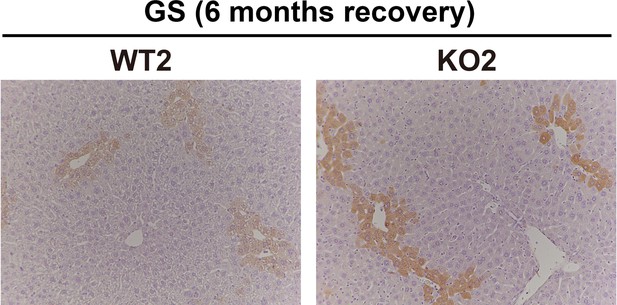
Immunohistochemistry for glutamine synthetase (GS) in WT2 and KO2 mice at 6 months of recovery showing KO2 beginning to express GS in the zone-3 hepatocytes as biliary epithelial cell (BEC)-derived β-catenin-positive hepatocyte repopulate these livers at 6 months after recovery from 2-week choline-deficient, ethionine-supplemented (CDE) injury (100×).
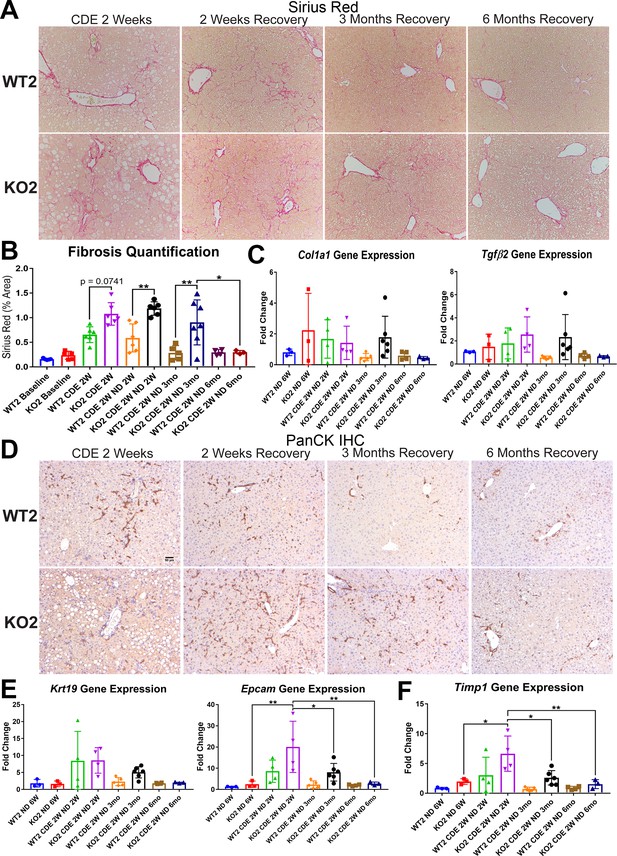
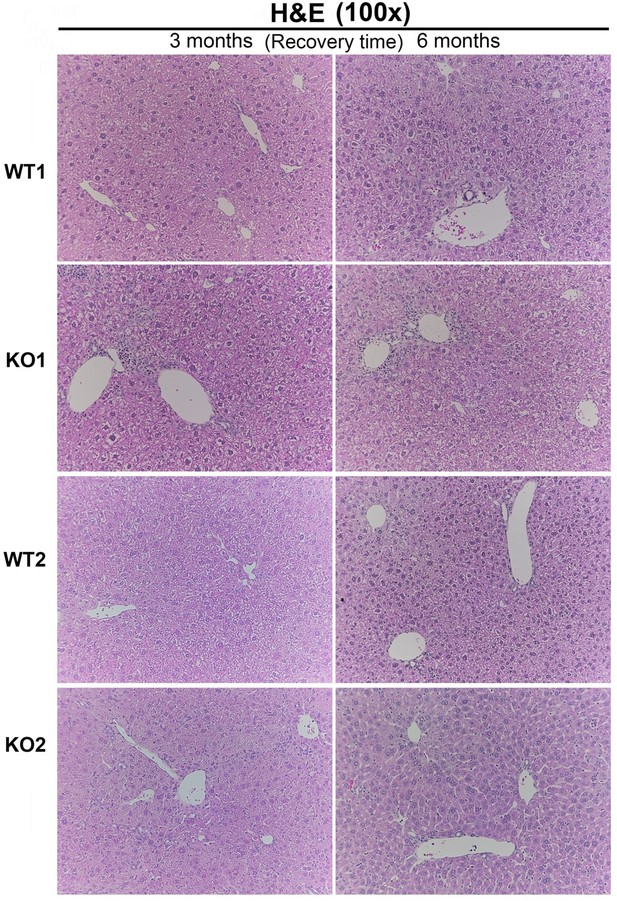
Fibrosis and ductular reaction (DR) is sustained in KO2 mice recovering on normal diet until 3 months, but subsides by 6 months, after initial 2-week choline-deficient, ethionine-supplemented (CDE) diet.
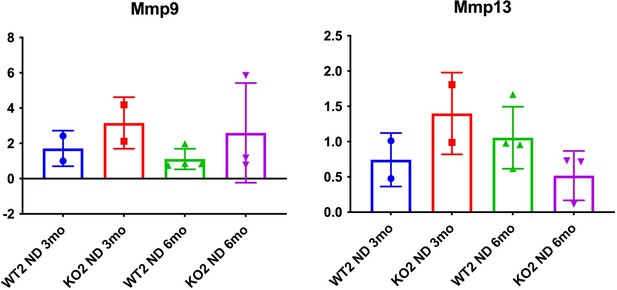
(A) Sirius Red staining in WT2 and KO2 mice over time during recovery from CDE diet (100×). (B) Quantification of Sirius Red staining (one-way ANOVA, *p<0.05, **p<0.01. n = 3–7 per group; individual animal values represented by dots). (C) A trend of increased expression of Col1a1 and Tgfβ2 in KO2 mice at 3 months of recovery but not at 6 months (n = 3–6 per group; individual animal values represented by dots). (D) Pan-cytokeratin (PanCK) staining in WT2 and KO2 mice over time during recovery from CDE diet. Scale bar = 50 µm. (E) A trend of higher Krt19 expression and significantly higher expression of Epcam gene in KO2 up to 3 months on recovery and normalization to WT2 levels at 6 months (one-way ANOVA, *p<0.05, **p<0.01, n = 3–6 per group, individual animal values represented by dots). (F) Significantly higher Timp1 gene expression in KO2 than WT2 up to 3 months on recovery diet and normalization at 6 months (one-way ANOVA, *p<0.05, **p<0.01, n = 3–6 per group, individual animal values represented by dots).
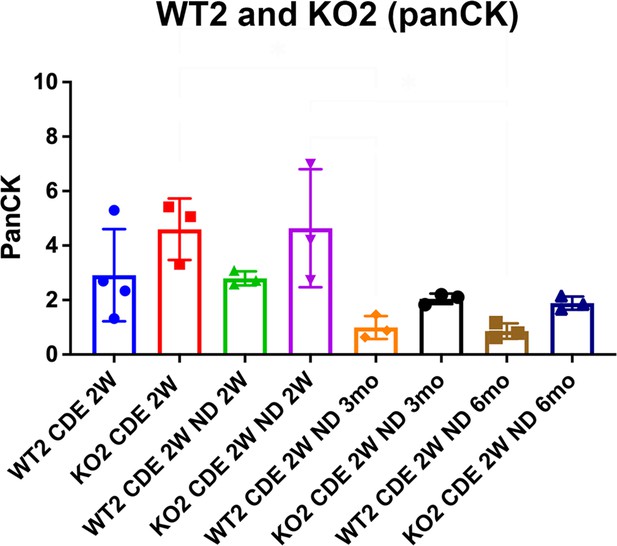
Similar ductular reaction (DR) during recovery from choline-deficient, ethionine-supplemented (CDE) in KO2 compared with WT2.
Bar graph represents quantification of percent area covered by pan-cytokeratin (PanCK)-positive ductular cells in KO2 and at various times following 2 weeks of CDE injury. No significant differences were observed at all recovery time points.
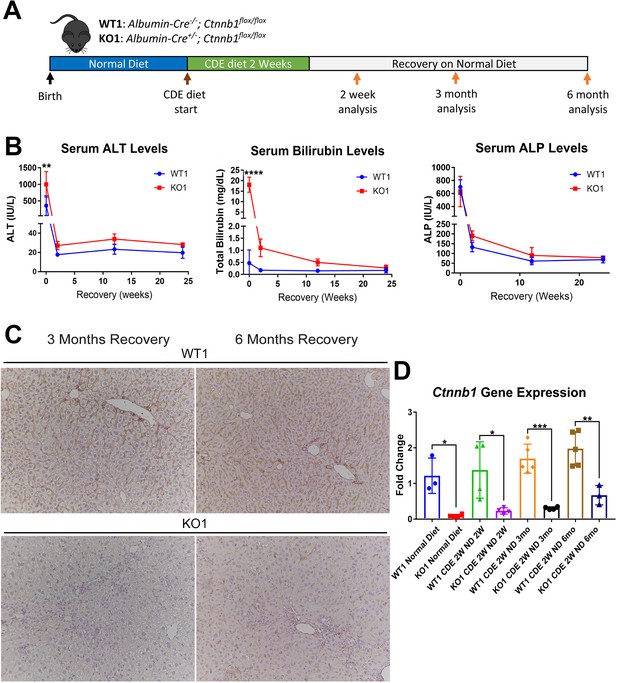
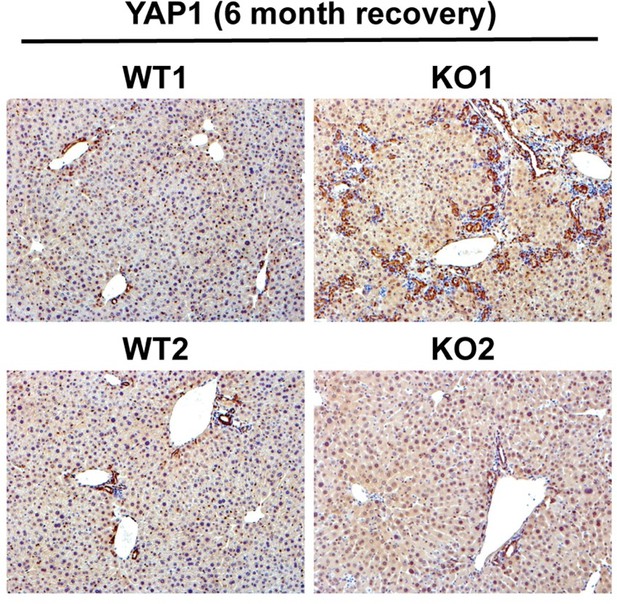
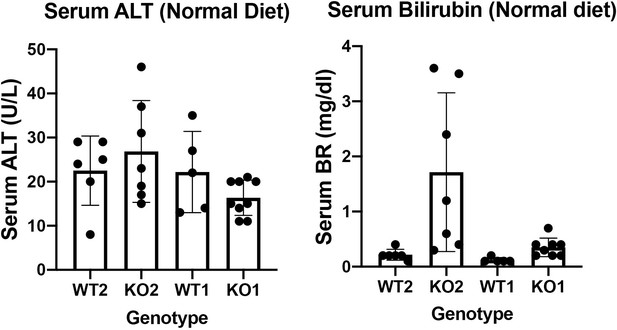
Serum biochemistry suggests comparable recovery on normal diet in WT1 and KO1 after 2-week choline-deficient, ethionine-supplemented (CDE) diet and continued lack of β-catenin in KO1.
(A) Experimental design showing WT1 and KO1 on 2 weeks of CDE diet and recovery on normal diet for up to 6 months with analysis at intermediate time points as indicated. (B) Serum alanine aminotransferase (ALT), bilirubin, and alkaline phosphatase (ALP) in the two groups over time (one-way ANOVA, **p<0.01, ****p<0.0001, n = 3–5 per group). (C) β-Catenin immunohistochemistry in WT1 and KO1 mice at 3 months and 6 months of recovery showing absence of β-catenin in biliary epithelial cells (BECs) and hepatocytes in KO1 (100×). (D) Ctnnb1 gene expression in WT1 and KO1 mice during recovery from CDE diet shows continued β-catenin absence over time in KO1 (one-way ANOVA, *p<0.05, **p<0.01, ****p<0.0001, n = 3–5 per group, individual animal values represented by dots).
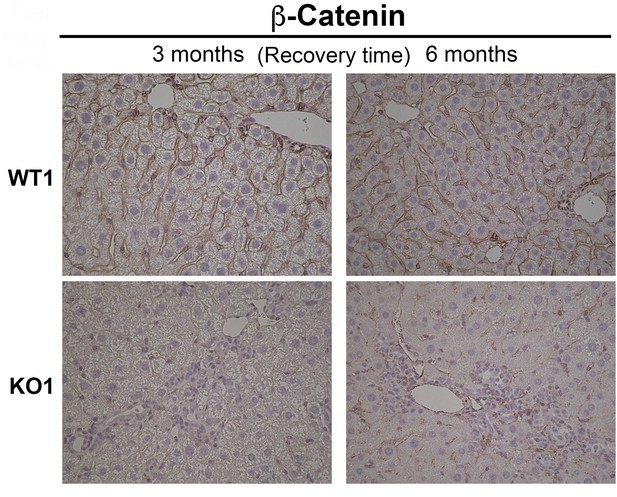
β-Catenin immunohistochemistry in WT1 and KO1 mice at 3 months and 6 months of recovery showing WT1 and KO1 continue to be negative for β-catenin in hepithelial cells (200×).
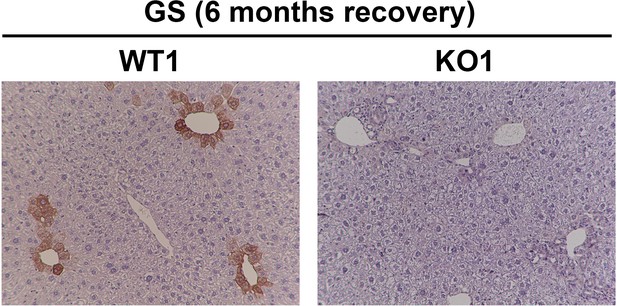
Immunohistochemistry for glutamine synthetase (GS) in WT1 and KO1 mice at 6 months of recovery showing KO1 continue to be negative for GS as there is no cell source for β-catenin+ hepatocytes in this model (100×).
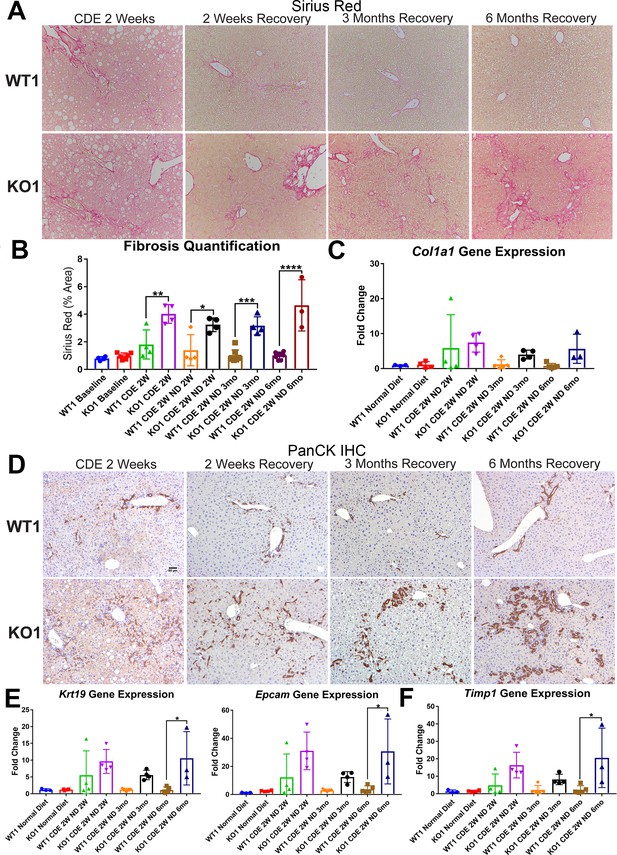
Unresolved fibrosis and ductular reaction in KO1 mice throughout 6 months on recovery, after the initial 2-week choline-deficient, ethionine-supplemented (CDE) diet injury.
(A) Sirius Red staining in WT1 and KO1 mice over time during recovery from CDE diet (100×). (B) Quantification of Sirius Red staining (one-way ANOVA, *p<0.05, **p<0.01, ***p<0.001, ****p<0.0001, n = 3–6 per group, individual animal values represented by dots). (C) A trend of increased expression of Col1a1 in KO1 mice even at 6 months of recovery (n = 3–5 per group, individual animal values represented by dots). (D) Pan-cytokeratin (PanCK) staining in WT1 and KO1 mice over time during recovery from CDE diet. The ductular reaction (DR) changes from flattened, invasive. and without lumen morphology from early time points to numerous small luminal structures lined by a single layer of PanCK-positive columnar cells at 3 months and 6 months. Scale bar = 50 µm. (E) Significantly higher Krt19 and Epcam gene expression in KO1 especially at 6 months of recovery (one-way ANOVA, *p<0.05, n = 3–5 per group, individual animal values represented by dots). (F) Significantly higher Timp1 gene expression IN KO1 especially at 6 months of recovery diet (one-way ANOVA, *p<0.05, n = 3–5 per group, individual animal values represented by dots).
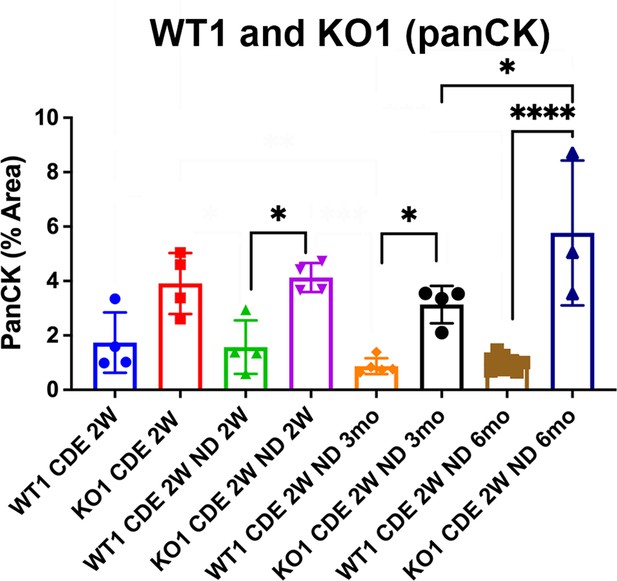
Enhanced ductular reaction (DR) during recovery from choline-deficient, ethionine-supplemented (CDE) in KO1.
Bar graph represents quantification of percent area covered by pan-cytokeratin (PanCK)-positive ductular cells in KO1 and WT1 at various times following 2 weeks of CDE injury. Significant differences were observed in KO1 as compared to WT1 at all recovery time points (one-way ANOVA, *p<0.05, ****p<0.0001).
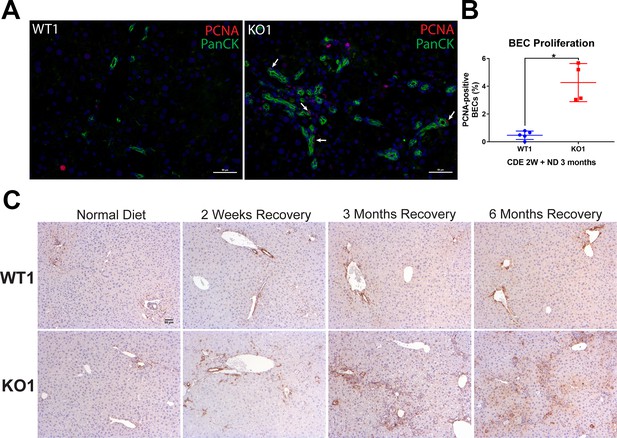
Enhanced proliferating cell nuclear antigen (PCNA) and increased p-Erk staining in biliary epithelial cells (BECs) in KO1 during recovery on normal diet after initial 2-week choline-deficient, ethionine-supplemented (CDE) diet-induced injury.
(A) Pan-cytokeratin (PanCK) (green) and PCNA (red) immunofluorescence in WT1 and KO1 mice at 3 months of recovery on normal diet after 2-week CDE diet. Scale bar = 50 µm. (B) Quantification of PCNA-positive BECs at 3-month recovery on normal diet (ND) from 2-week CDE (one-way ANOVA, *p<0.05). (C) WT1 and KO1 mice staining with an antibody against phospho-Erk1/2 (Thr202/Tyr204) reveals staining in a subset of BECs in the ductular reaction in KO1 mice. Scale bar = 50 µm.
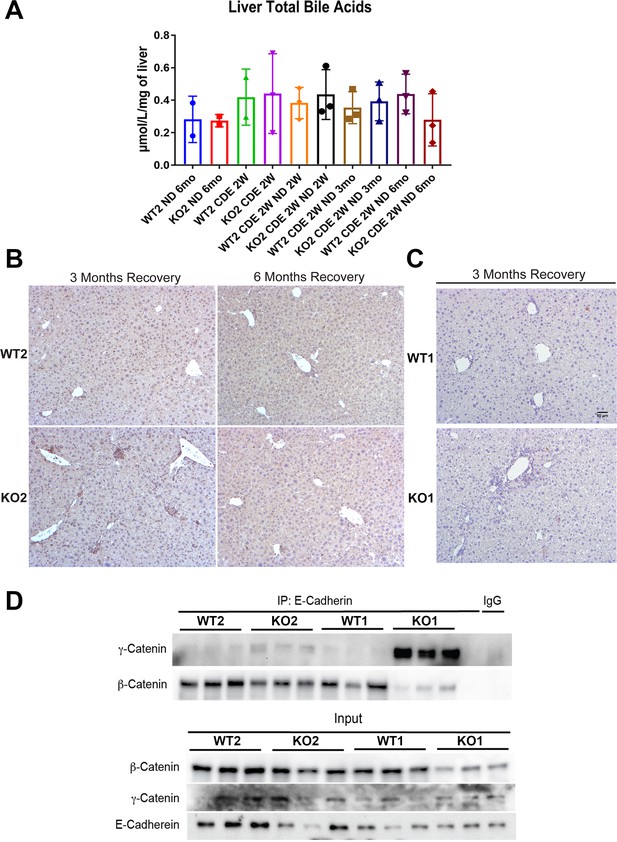
Bile acids and cell death are not the basis of fibrosis and ductular reaction due to choline-deficient, ethionine-supplemented (CDE) diet, and differences in adherens junction integrity do not explain phenotypic differences between KO2 and KO1 at 6 months of recovery.
(A) Quantification of bile acids in whole livers in WT2 and KO2 at 2 weeks after CDE diet and at various time recovery times on normal diet. (B) p21 staining shows no positive cells at 3 months or 6 months of recovery on normal diet in WT2 and KO2 liver sections. (C) Cleaved caspase 3 staining reveals almost no ongoing cell death in the livers of WT1 or KO1 mice after 3 months of recovery from CDE diet-induced liver injury. Scale bar = 50 µm. (D) Immunoprecipitation studies show E-cadherin association with β-catenin in WT1, WT2, and KO2 livers at 6 months of recovery while it associates with γ-catenin in KO1 at the same time due to continued lack of β-catenin in KO1 (top panels). Input verifies low levels of β-catenin in whole liver lysates of KO1 at the same time depicting β-catenin presence in liver non-epithelial cells (bottom panels).
-
Figure 4—figure supplement 3—source data 1
Immunoprecipitation (IP) shows E-cadherin association with β-catenin in WT1, WT2, and KO2 livers at 6 months of recovery while it associates with γ-catenin in KO1 at the same time due to continued lack of β-catenin in KO1 (top panels).
Input verifies low levels of β-catenin in whole liver lysates of KO1 at the same time depicting β-catenin presence in liver non-epithelial cells (bottom panels).
- https://cdn.elifesciences.org/articles/71310/elife-71310-fig4-figsupp3-data1-v3.pdf
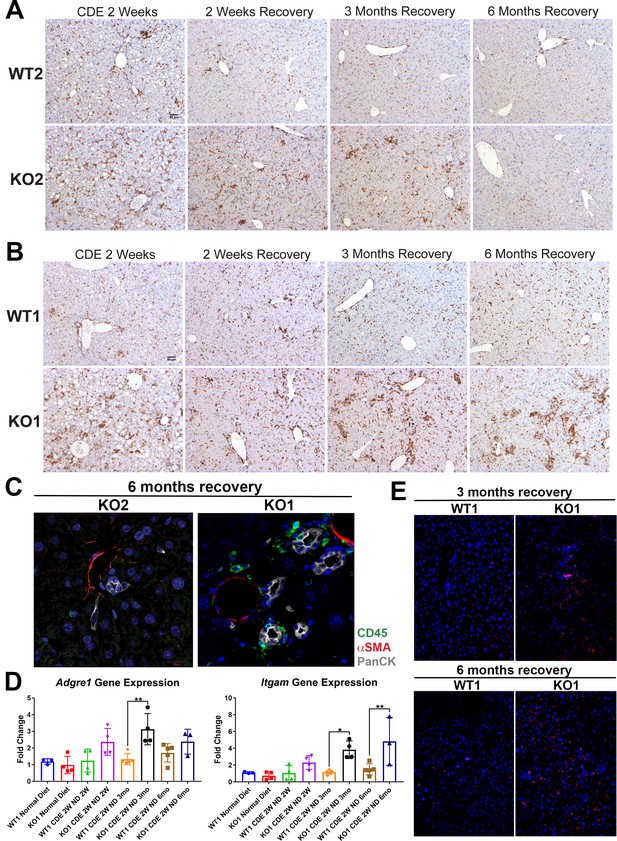
Sustained inflammation during recovery after the initial choline-deficient, ethionine-supplemented (CDE) diet injury in KO1 mice as compared to WT1, WT2, and KO2.
(A) CD45 immunostaining in WT2 and KO2 mice over time during recovery from CDE diet. Scale bar = 50 µm. (B) CD45 staining in WT1 and KO1 mice over time during recovery from CDE diet. Scale bar = 50 µm. (C) Representative confocal image of triple immunofluorescence for pan-cytokeratin (PanCK) (white), α-smooth muscle actin (αSMA) (red), and CD45 (green) in KO1 and KO2 at 6 months of recovery (400×). (D) Changes in Adgre1 and Itgam gene expression in WT1 versus KO1 at various time points (one-way ANOVA, *p<0.05, **p<0.01, n = 3–5 per group, individual animal values represented by dots). (E) Increased Ly6G staining (red) in KO1 mice after 3 months and 6 months of recovery (100×).
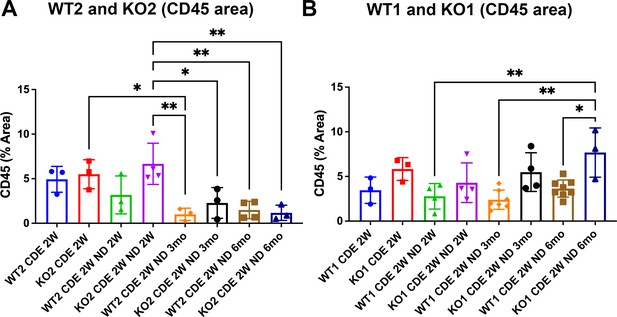
Increased CD45-positive cell infiltration in KO1 especially at 6 months of recovery from 2-week choline-deficient, ethionine-supplemented (CDE) injury.
Bar graph representing percentage of area occupied by CD45-positive cells in all four genotypes of mice at all time points. Statistically significant and higher inflammation was observed in KO1 at 6 months as compared to WT1 at all time points, although KO1 always tended to have higher CD45-positive cells than WT1 at respective times (B). This was in contrast to KO2, which progressively and significantly resolved CD45 infiltrate at 3 months and 6 months of recovery (A) (one-way ANOVA, *p<0.05, **p<0.01).
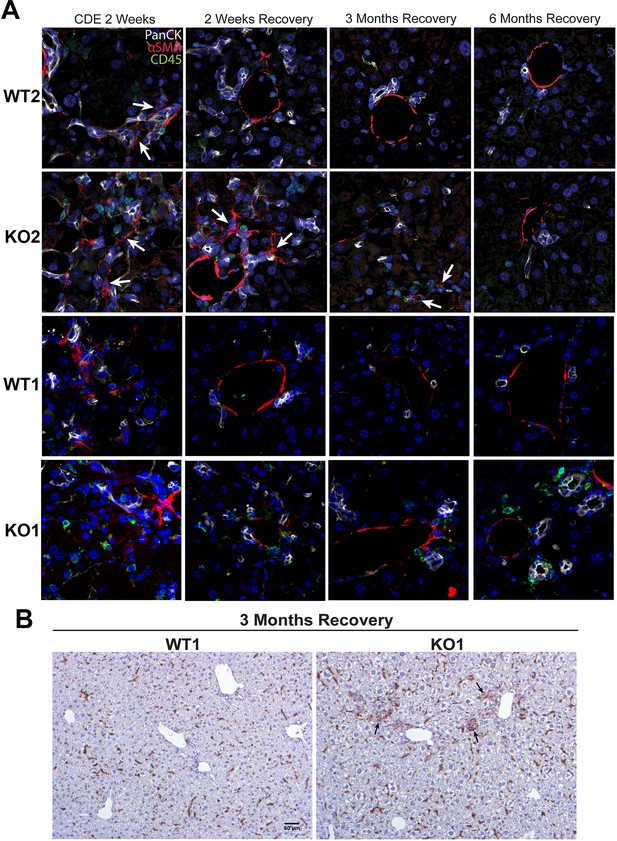
Immune cells continue to prevail in periportal region in KO1 even at 6 months of recovery while they subside in all other genotypes after 3 months of recovery or earlier.
(A) Representative confocal image of triple immunofluorescence for pan-cytokeratin (PanCK) (white), α-smooth muscle actin (αSMA) (red), and CD45 (green) in WT1, KO1, WT2, and KO2 at 2 weeks of choline-deficient, ethionine-supplemented (CDE) diet and recovery on normal diet for 2 weeks, 3 months, or 6 months. Cells expressing αSMA are closely associated with PanCK-positive cells (white arrows). Scale bar = 20 µm. (B) F4/80 staining reveals macrophages closely associated with the ductular reaction (DR) in KO1 mice at 3 months of recovery (black arrows). Scale bar = 50 µm.
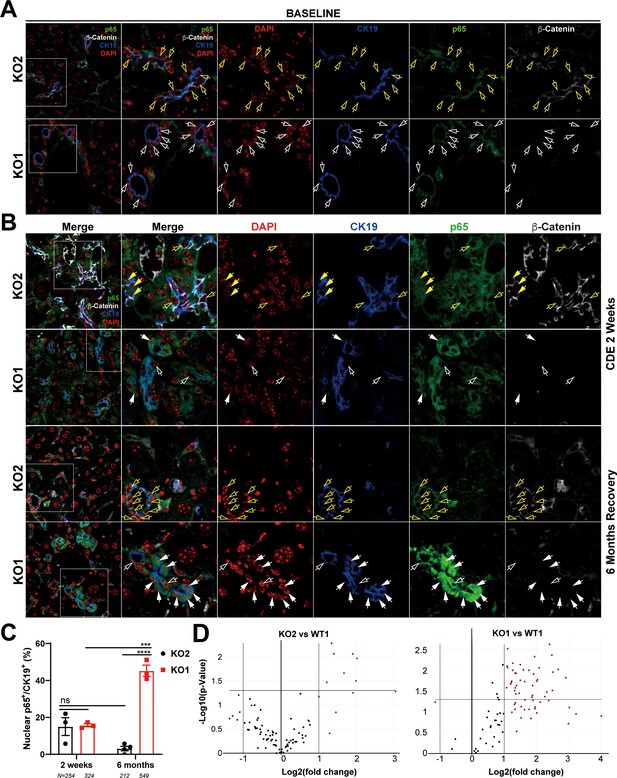
Nuclear translocation of p65 in biliary epithelial cells (BECs) lacking β-catenin during recovery from choline-deficient, ethionine-supplemented (CDE) diet injury shows NF-κB activation in BECs only in KO1.
(A) Representative confocal image of triple immunofluorescence for CK19 (blue), β-catenin (white), and p65 (green) along with DAPI (red) in KO1 and KO2 baseline livers. Merged image at low magnification (100×) is shown in leftmost panel and higher magnification (200×) of selected area (box) along with its individual channels are shown to the right. Yellow open arrows identify CK19-positive BECs with cytosolic β-catenin and p65 in KO2. White open arrows identify CK19-positive BECs with cytosolic p65 and absent β-catenin in KO1. (B) Representative confocal image of triple immunofluorescence for CK19 (blue), β-catenin (white), and p65 (green) along with DAPI (red) in KO1 and KO2 at 2 weeks of CDE diet and after 6 months of recovery on normal diet. The leftmost panel is low magnification (100×) merged image. The higher magnification (200×) of the selected boxed area is presented in the adjacent panel as a merged image followed by individual channels. Yellow open arrows identify CK19-positive BECs with cytosolic β-catenin and p65, and yellow solid arrows with nuclear p65 in KO2. White open arrows indicate CK19-positive BECs with cytosolic p65, and white solid arrows identify CK19-positive BECs with nuclear p65 with absent β-catenin in KO1. (C) Quantification of CK19-positive cells showing nuclear p65 at 2 weeks of CDE diet and 6 months of recovery in KO2 versus KO1 (one-way ANOVA, ***p<0.001, ****p<0.0001, n = 3 per group, individual animal values represented by dots, number of cells counted are indicated). (D) Volcano plots of NF-κB downstream target gene expression in KO2, KO1, and WT1 livers at 6 months of recovery. Genes with fold-change >2 are highlighted in red, with fold-change <2 highlighted in green, and unchanged genes shown as black dots (n = 3 per group).
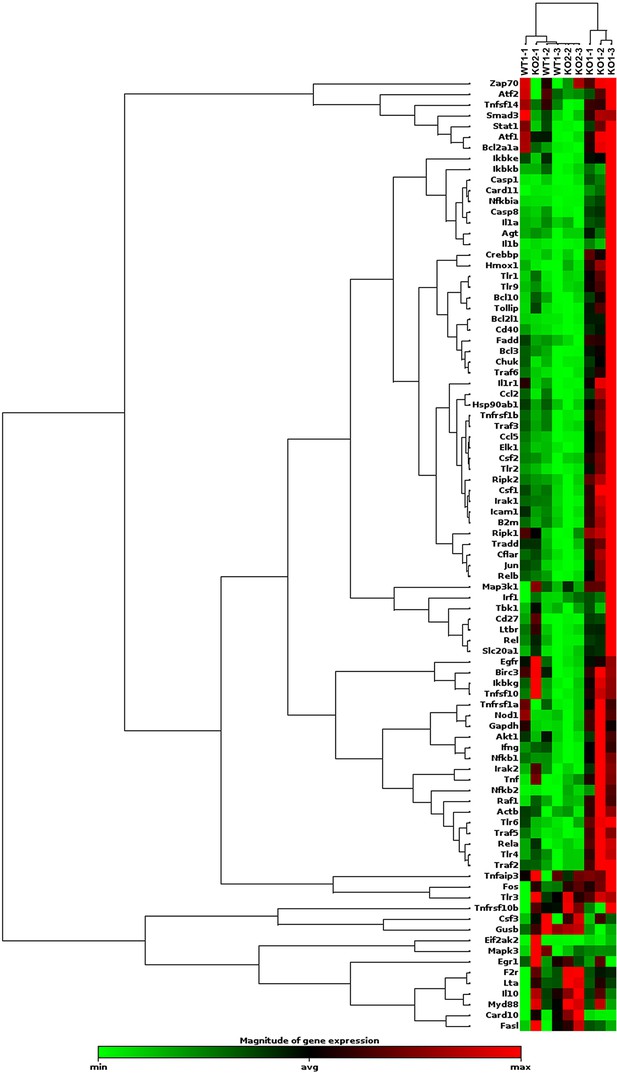
Evidence of NF-κB activation in KO1 livers but not in WT1 or KO2 livers at 6 months of recovery from choline-deficient, ethionine-supplemented (CDE) diet.
RNA from whole livers of WT1, KO1, and KO2 (n = 3 each) at 6-month recovery was assessed for 84 NF-κB downstream target genes by RT-PCR array. Using a fold-change threshold = 2, p-value threshold = 0.05, we identified several genes altered in KO1 only and clustergram showed clear separation of KO1 from KO2 and WT1.
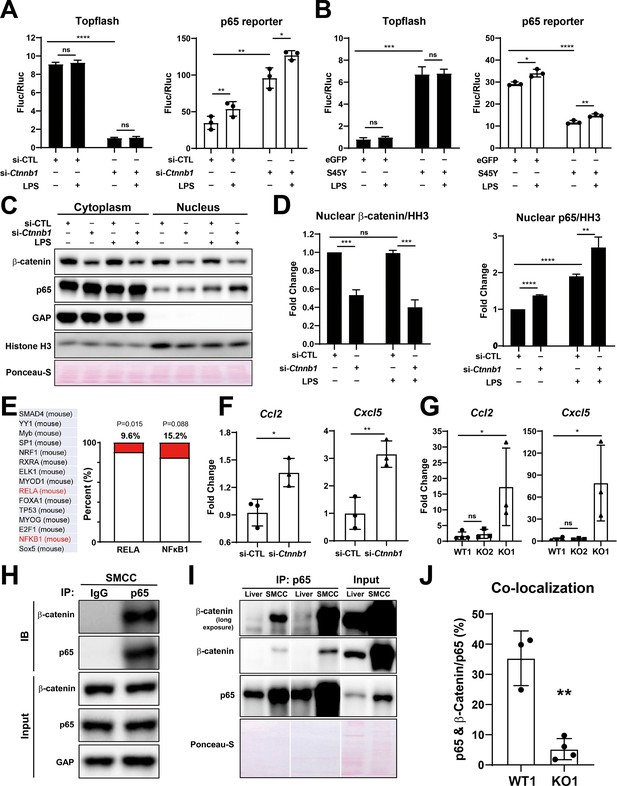
Modulation of β-catenin in biliary epithelial cells (BECs) perturbs its complex with p65 to impact NF-κB activity.
(A) Luciferase reporter assay shows successful knockdown of Ctnnb1 in small cholangiocyte cell (SMCC) line by TOPFlash assay (left), which stimulates p65 transcriptional activity with or without 100 ng/ml lipopolysaccharide (LPS) (right) (unpaired t-test, ns: no significance, *p<0.05, **p<0.01, ****p<0.0001, n = 3 biological replication). (B) Luciferase reporter assay shows expression of constitutively active S45Y-β-catenin enhances TOPFlash (left) and suppresses p65 transcriptional activity with or without 100 ng/ml LPS (right) (unpaired t-test, ns: no significance, *p<0.05, **p<0.01, ***p<0.001, ****p<0.0001, n = 3 biological replication). (C) Representative WB from two independent experiment shows knockdown of Ctnnb1 increases p65 nuclear translocation with or without 500 ng/ml LPS. (D) Quantification of nuclear β-catenin (left) and nuclear p65 (right) to HH3 (blots in (C) were technically quantified three times and p-value was calculated using unpaired t-test, ns: no significance, **p<0.01, ***p<0.001, ****p<0.0001). (E) Identification of RELA and NFKB1 among the top 15 transcription factors identified by applying the 335 differentially expressed genes (DEGs) to JASPAR. (F) qPCR shows knockdown of Ctnnb1 in SMCCs induces Ccl2 (left) and Cxcl5 (right) expression (unpaired t-test, ns: no significance, *p<0.05, **p<0.01, n = 3 biological replication). (G) qPCR shows Ccl2 (left) and Cxcl5 (right) are induced in KO1 after 6-month recovery of choline-deficient, ethionine-supplemented (CDE) diet (unpaired t-test, *p< 0.05, n = 3–4 biological replication). (H) Representative immunoprecipitation (IP) image from two independent experiment shows p65 is strongly associated with β-catenin in SMCC. (I) IP shows that p65 is associated with β-catenin in whole liver lysate (L: liver; S: SMCC; P: equal amount of liver and SMCC lysate). (J) Quantification of colocalization of p65 and β-catenin is significantly diminished in KO1 compared to WT1 (unpaired t-test, **p<0.01, n = 3–4 biological replication).
-
Figure 7—source data 1
WB shows knockdown of Ctnnb1 increases p65 nuclear translocation with or without 500 ng/ml lipopolysaccharide (LPS).
- https://cdn.elifesciences.org/articles/71310/elife-71310-fig7-data1-v3.pdf
-
Figure 7—source data 2
Immunoprecipitation (IP) image shows p65 is strongly associated with β-catenin in small cholangiocyte cell (SMCC).
- https://cdn.elifesciences.org/articles/71310/elife-71310-fig7-data2-v3.pdf
-
Figure 7—source data 3
Immunoprecipitation (IP) shows that p65 is associated with β-catenin in whole liver lysate.
- https://cdn.elifesciences.org/articles/71310/elife-71310-fig7-data3-v3.pdf
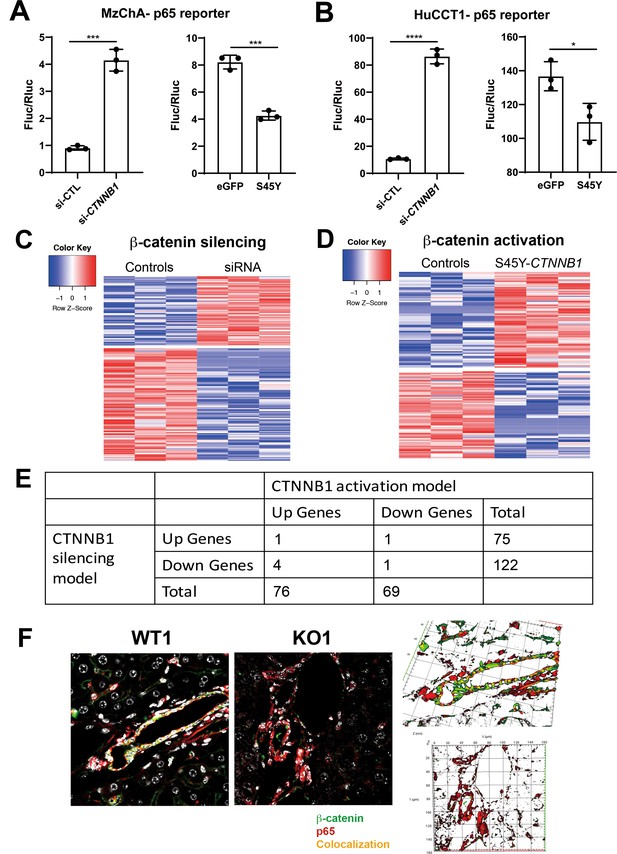
Modulation of β-catenin in cholangiocytes impacts NF-κB activity due to p65-β-catenin complex.
(A) Reporter assay shows knockdown of Ctnnb1 stimulates p65 transcriptional activity (left) while transfection of stable S45Y-β-catenin represses p65 reporter activity (right) in MzChA human cholangiocarcinoma cells (unpaired t-test, ***p<0.001). (B) Reporter assay shows knockdown of Ctnnb1 stimulates p65 transcriptional activity (left) while expression of constitutively active S45Y-β-catenin suppresses p65 transcriptional activity (right) in HuCCT1 human cholangiocarcinoma cells (unpaired t-test, *p<0.05, **p<0.01, ****p<0.0001). (C) Heatmap of the differentially expressed genes in Ctnnb1 loss of function in small cholangiocyte cell (SMCC). (D) Heatmap of the differentially expressed genes in CTNNB1 gain of function in SMCC. (E) Very few common differentially expressed genes between Ctnnb1 loss- and gain-of-function models were identified, although altogether 335 genes were altered. These genes were assessed for transcription factor (TF) binding profiles by JASPAR (presented in Figure 7E). (F) Representative confocal images showing the expression of p65 (red), β-catenin (green), and DAPI (white) in WT1 and KO1 livers (left). To visualize colocalization for p65 (red) and β-catenin (green), 3D images were reconstructed using Zen blue 2012 software (right). The reconstruction was performed with nine confocal z-stacks with 0.8 µm increments, and yellow region indicates colocalization of p65 (red) and β-catenin (green) in the projection on the X, Y, and Z planes.
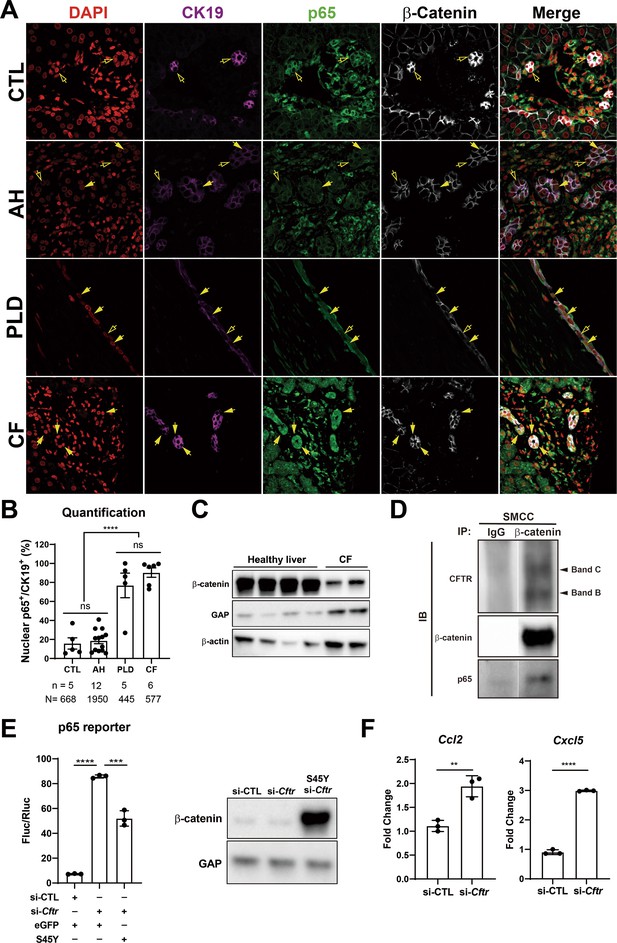
Nuclear p65 is highly present in the ductular cells of cystic type liver disease.
(A) Representative immunofluorescence (IF) images of liver sections from patients with healthy liver (CTL), alcoholic hepatitis (AH), polycystic liver disease (PLD), and cystic fibrosis (CF) (200×). (B) Quantification of the percentage of nuclear p65+ CK19+ cells among CK19+ biliary epithelial cells (BECs) from CTL (668 cells from 5 patients), AH (1950 cells from 12 patients), PF (445 cells from 5 patients), and CF (577 cells from 6 patients) (one-way ANOVA, ****p<0.0001). (C) WB shows β-catenin is decreased in two liver samples from one CF patient as compared to four healthy liver controls. (D) Immunoprecipitation (IP) studies show cystic fibrosis transmembranous conductance regulator (CFTR) (C-band and B-band) and p65 to be pulled down with β-catenin and not with IgG control in small cholangiocyte cell (SMCC). Images are from the same gel with same exposure time. (E) Luciferase reporter assay shows knockdown of CFTR in SMCC line strongly induces p65 transcriptional activity. Overexpression of stable S45Y-β-catenin partially rescues p65 activity (unpaired t-test, ***p<0.001, ****p<0.0001, n = 3 biological replication). Representative WB shows β-catenin levels after si-Cftr or after simultaneous S45Y-β-catenin expression as compared to si-control (si-CTL). (F) qPCR shows knockdown of CFTR induces Ccl2 and Cxcl5 expression in SMCC (unpaired t-test, **p<0.01, ***p<0.001, n = 3 biological replication).
-
Figure 8—source data 1
WB shows β-catenin is decreased in two liver samples from one cystic fibrosis (CF) patient as compared to four healthy liver controls.
- https://cdn.elifesciences.org/articles/71310/elife-71310-fig8-data1-v3.pdf
-
Figure 8—source data 2
Immunoprecipitation (IP) studies show cystic fibrosis transmembranous conductance regulator (CFTR) (C-band and B-band) and p65 to be pulled down with β-catenin and not with IgG control in small cholangiocyte cell (SMCC).
- https://cdn.elifesciences.org/articles/71310/elife-71310-fig8-data2-v3.pdf
-
Figure 8—source data 3
WB shows β-catenin levels after si-Cftr or after simultaneous S45Y-β-catenin expression as compared to si-control (si-CTL).
- https://cdn.elifesciences.org/articles/71310/elife-71310-fig8-data3-v3.pdf
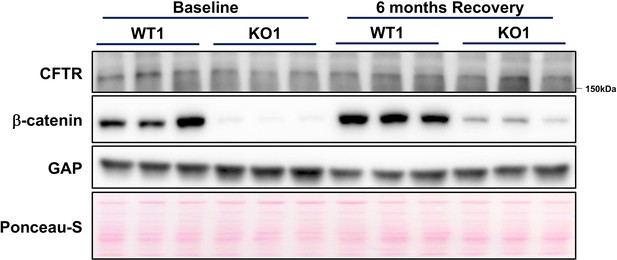
Total cystic fibrosis transmembranous conductance regulator (CFTR) levels are unaffected by loss of β-catenin in KO1 livers at baseline and during recovery from choline-deficient, ethionine-supplemented (CDE) injury.
WB using whole cell lysates from the WT1 and KO1 livers at baseline and at 6-month recovery time point from a 2-week CDE diet-induced injury show continued absence of β-catenin in KO1 (remnant band likely represents presence of immune cells in the whole liver), while CFTR (expressed only in biliary epithelial cells [BECs]) shows no change in total protein levels.
-
Figure 8—figure supplement 1—source data 1
Lack of changes in CFTR protein in whole liver lysates from KO1 livers.
- https://cdn.elifesciences.org/articles/71310/elife-71310-fig8-figsupp1-data1-v3.pdf
Additional files
-
Supplementary file 1
Patient information whose samples were used in the study.
Patient tissue sections including 5 patients with healthy liver, 12 patients with ductular reaction (DR) from alcoholic hepatitis (AH) (n = 10) and/or NASH (n = 2), 5 patients with DR associated with polycystic liver disease (PLD), and 6 patients with DR in cystic fibrosis (CF) cases were provided by the Pittsburgh Liver Research Center’s (PLRC’s) Clinical Biospecimen Repository and Processing Core (CBPRC). Two pieces of frozen livers from one CF patient (TP10-P531) were provided by Pitt Biospecimen Core.
- https://cdn.elifesciences.org/articles/71310/elife-71310-supp1-v3.docx
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/71310/elife-71310-transrepform1-v3.pdf