Precise in vivo functional analysis of DNA variants with base editing using ACEofBASEs target prediction
Figures
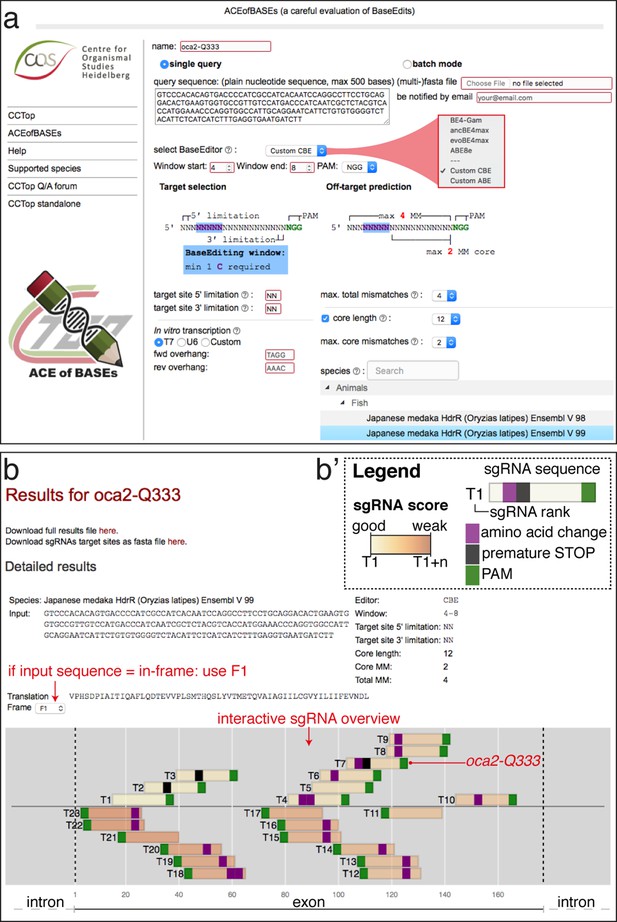
ACEofBASEs (a careful evaluation of base edits) enables simple and tailored use of base editors.
(a) User interface of the ACEofBASEs base editing design tool with base editor choice dropdown menu and a compendium of model species to select from. (b) Results page of the ACEofBASEs design tool for cytosine base editing of the Oryzias latipes oca2 locus. Using an in-frame sequence of the target site will directly provide the translation frame F1. Alternatively, frames can be selected from the dropdown menu. All sgRNA target sites found in the query are shown with potential amino acid change (magenta box) or nonsense mutation (PTCs, black box); here: standard editing window: nucleotides 4–8 on the protospacer; PAM: positions 21–23. For off-target prediction, a comprehensive list of potential off-target sites that contain an A or C in the respective base editing window is provided per sgRNA target. Potential off-target sites are sorted according to a position-weighted likelihood to introduce an off-target, that is the closer a mismatch at the potential off-target site to the PAM, the more unlikely this site is falsely edited (Stemmer et al., 2015).
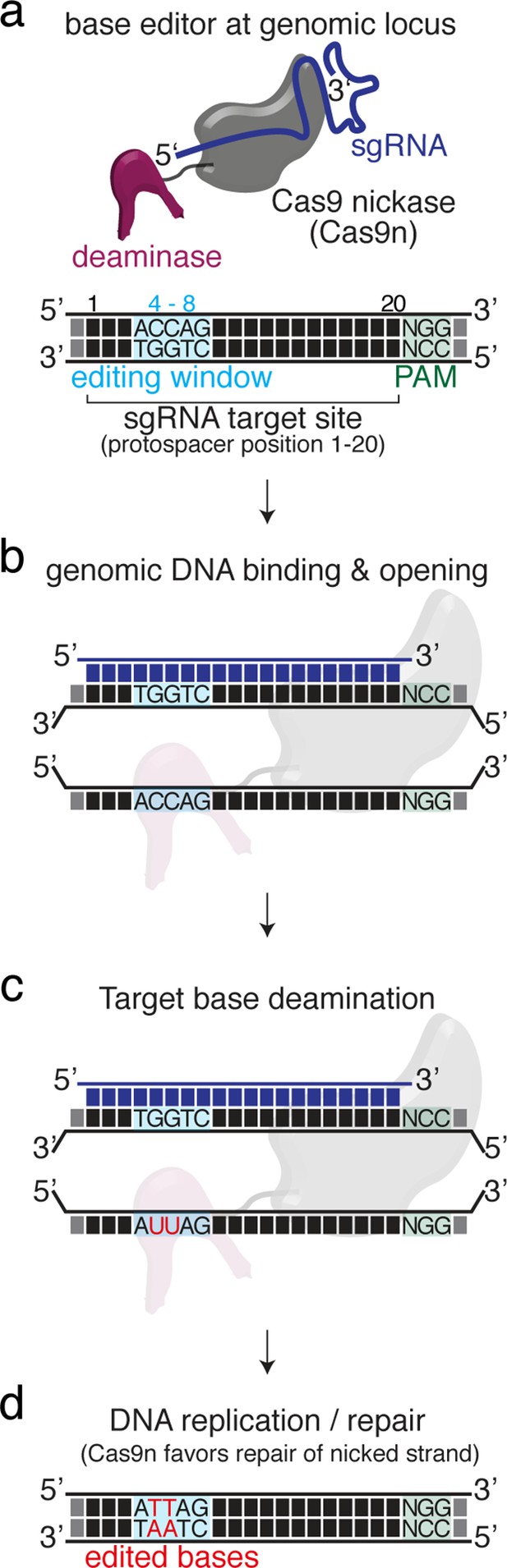
Basic mechanism of cytosine base editing exemplifying the base editing principle.
(a) The cytosine base editor (CBE) complex will be guided to the genomic locus of interest by the selected sgRNA. While the protospacer adjacent motif (PAM) is essential for target recognition the editing window on the protospacer is, however, more volatile depending on the editor used and resides between positions 4 and 8 for standard CBEs and ABEs. (b–d) Mechanism of CBE-DNA binding, DNA opening and hydrolytic deamination. Following cellular DNA repair or replication processes target cytosines are converted to thymines. Note that within the hypothetical editing window any cytosine may be deaminated.
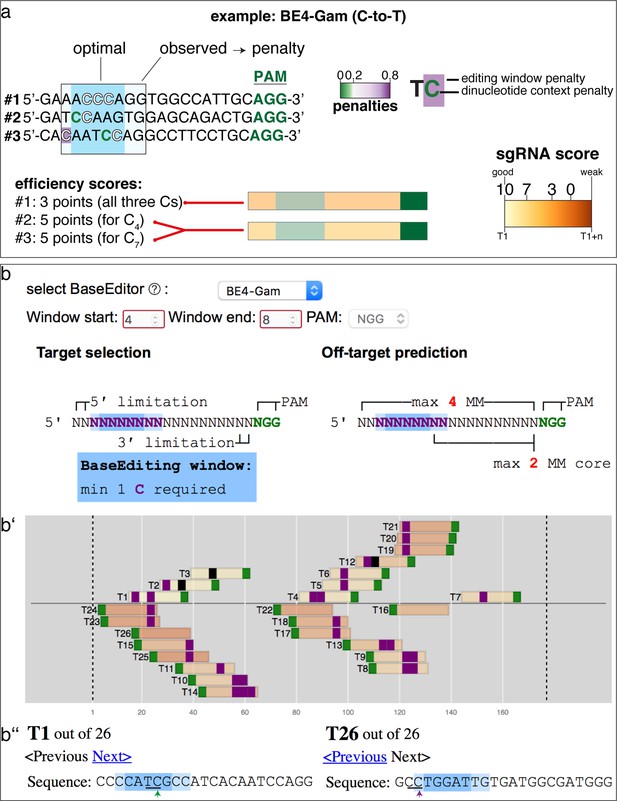
sgRNA score explanation and example.
(a) Exemplified by three potential sgRNAs, the scoring system penalizes cytosines or adenines to be edited (1) outside the editing window and (2) where the dinucleotide context is unfavorable. See Materials and methods section for details. Note that only the best scoring cytosine or adenine is considered for the ranking. Here, both #2 and #3 score equally well. #2 or #3: favorable TC (C4 or C7) context within optimal editing window; #1: unfavorable AC and CC within optimal editing window. (b) Shows sgRNA ranking exemplified by using the cytosine base editor BE4-Gam and the input sequence as in Figure 1a. (b’) Note the different order of sgRNAs compared to Figure 1b. (b’’) The TC context in the optimal editing window (green arrow) strongly favors T1, whereas the CC context in the observed editing window (magenta arrow) predicts reduced efficiencies for T26.
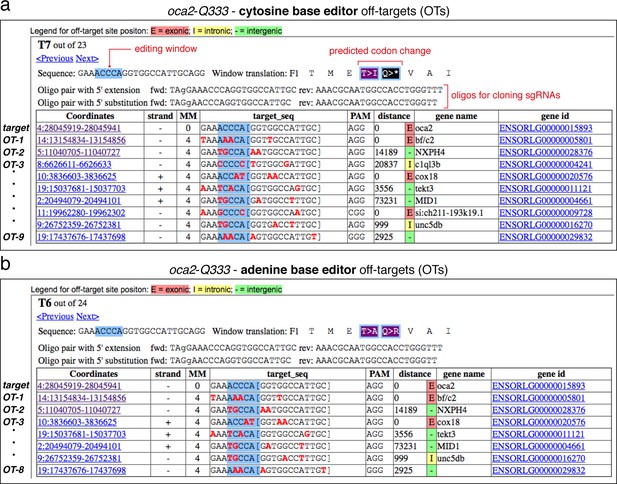
Details on selected sgRNA in ACEofBASEs.
Overview of sgRNA oca2-Q333 with target sequence, potential codon changes, and oligonucleotides required for cloning into the DR274 in vitro transcription vector for use with cytosine (a) and adenine base editors (b). All sgRNAs designed and tested were cloned with 5' substitution. A list of genomic targets is shown below, including a list of potential DNA off-target sequences (OTs) provided with sequence coordinates, mismatches (shown in red), gene name, and gene ID.
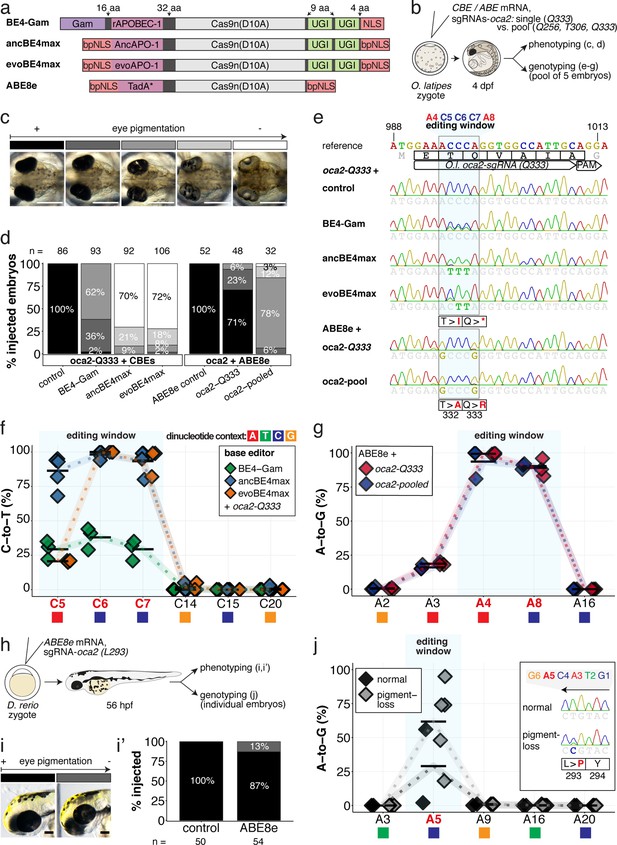
Somatic cytosine and adenine base editing at the oculocutaneous albinism II (oca2) locus in medaka and zebrafish allows direct functional assessment.
(a) Schematic diagram of the cytosine base editors BE4-Gam, ancBE4max and evoBE4max (evoAPOBEC1-BE4max) and the adenine base editor ABE8e. Cas9n-D10A nickase (light grey) with N-terminally linked cytidine or deoxyadenosine deaminase (pink) and C-terminal SV40 or bipartite (bp) nuclear localization sequence (NLS, red). All except BE4-Gam also contain the bpNLS N-terminally. CBEs contain variations of the rat APOBEC-1 cytidine deaminase, whereas ABE8e contains the TadA* domain (tRNA adenine deaminase), CBEs further contain C-terminally linked Uracil glycosylase inhibitors (UGI, green). Gam protein from bacteriophage Mu (purple) and linkers of varying lengths (dark grey). (b) Scheme of the experimental workflow. Cytosine or adenine base editor (CBE/ABE) mRNA and oca2-Q333 or a pool of three oca2-sgRNAs (–Q256, –T306, –Q333) were injected into the cell of a medaka zygote. Control injections only contained oca2-Q333 or ABE8e mRNA. (c) Phenotypic inspection of eye pigmentation was performed at 4 dpf (dorsal view). (d) Grouped and quantified pigmentation phenotypes shown for BE4-Gam, ancBE4max, evoBE4max, and ABE8e experiments. Control only contains oca2-Q333 sgRNA. n shown excludes embryos that are otherwise abnormal or dead, with abnormality rate given in supplement 1c. (e) Exemplary Sanger sequencing reads for each experimental condition, obtained from a pool of five randomly selected embryos at the oca2-Q333 locus. (f–g) Quantification of Sanger sequencing reads (by EditR, Kluesner et al., 2018) for BE4-Gam (n = 3), ancBE4max (n = 5) and evoBE4max (n = 3) (f), and ABE8e for single (n = 3) and pooled oca2-sgRNA experiments (n = 3) (g). Pools of five embryos per data point summarizes editing efficiencies. Mean data points are summarized in Supplementary files 1 and 2. To highlight the dinucleotide context, the nucleotide preceding the target C or A is shown by red (A), green (T), blue (C) and yellow (G) squares below the respective C or A. (h) Microinjections into the yolk of one-cell stage zebrafish were performed with ABE8e mRNA and oca2-L293 sgRNA. Zebrafish larvae were phenotypically analyzed at 56 hpf and individual larvae were subsequently genotyped. (i-i’) Larvae were scored as without (‘normal’, black) or with loss of eye pigment (grey). (j) Sanger sequencing on individually scored larvae was analyzed by EditR and plotted according to phenotype. Scale bars = 400 µm (c) or 100 µm (i). dpf / hpf = days/hours post fertilization.
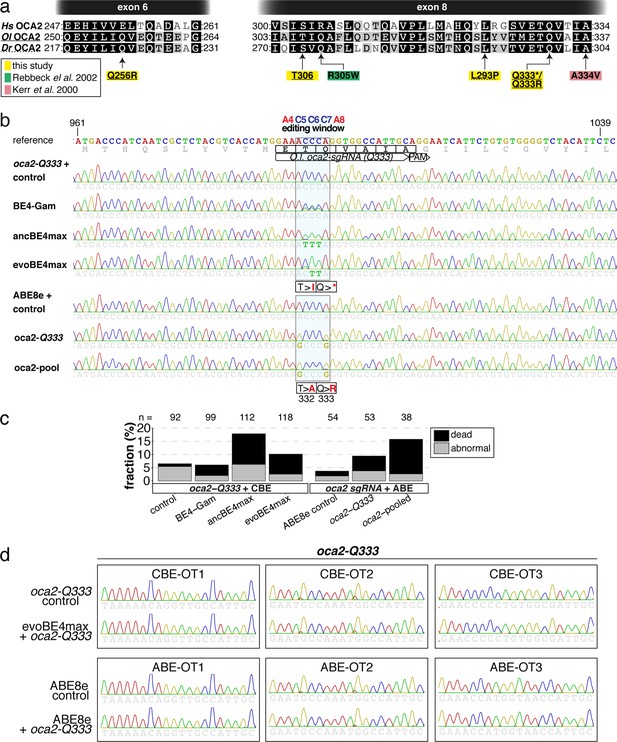
Somatic cytosine and adenine base editing at the medaka oculocutaneous albinism II (oca2) gene occurs in the absence of detectable indels and DNA off-target editing.
(a) Translation of sections of exon 6 and 8 of human (Hs), medaka (Ol) and zebrafish (Dr) OCA2 shows high degree of conservation. Amino acids edited or targeted in this study are highlighted in yellow. Missense mutations R305W (green) and A334V (pink) highlight albinism-associated human OCA2 mutations (Kerr et al., 2000; Rebbeck, 2002). (b) Broader window of the Sanger sequencing result displayed in Figure 2e, indicates lack of indels or unwanted editing (Kluesner et al., 2018). (c) Microinjection of oca2 sgRNAs with respective editors at the 1 cell stage causes low levels of aberrant phenotypes in medaka. Abnormal embryos either showed developmental delay or sublethal phenotype, at rates of 6.5%, 6.1%, 17.9%, 10.1%, 3.7%, 9.4%, and 15.8% (left to right), respectively n indicates total number of injected embryos, respectively. (d) The top three DNA off-targets (OTs) as predicted by ACEofBASEs for evoBE4max and ABE8e were evaluated for off-target base editing.
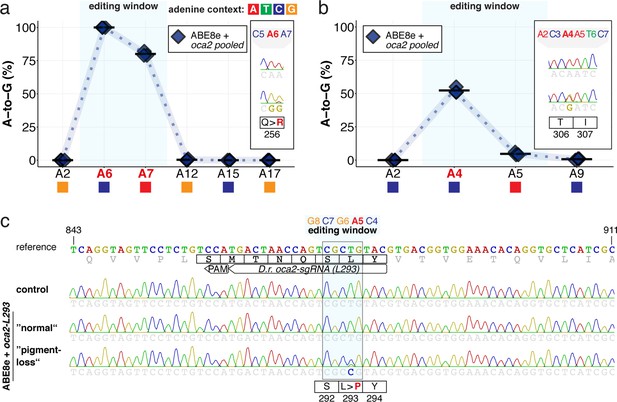
ABE8e efficiently introduces A-to-G mediated missense mutations in medaka and zebrafish.
(a–b) EditR quantification of Sanger sequencing reads for pooled oca2 ABE8e experiments (Q256, T306, Q333) from pools of five medaka embryos per data point, summarizes editing efficiencies for Q256 (a) and T306 (b) loci. For mean values see Supplementary file 2. To highlight the dinucleotide context, the nucleotide preceding the target A is shown by red (A), green (T), blue (C) and yellow (G) squares below the respective A. (c) Wider window of exemplary Sanger sequencing reads for individual, base edited embryos, from each experimental condition following ABE8e experiments at the zebrafish oca2-L293 locus.
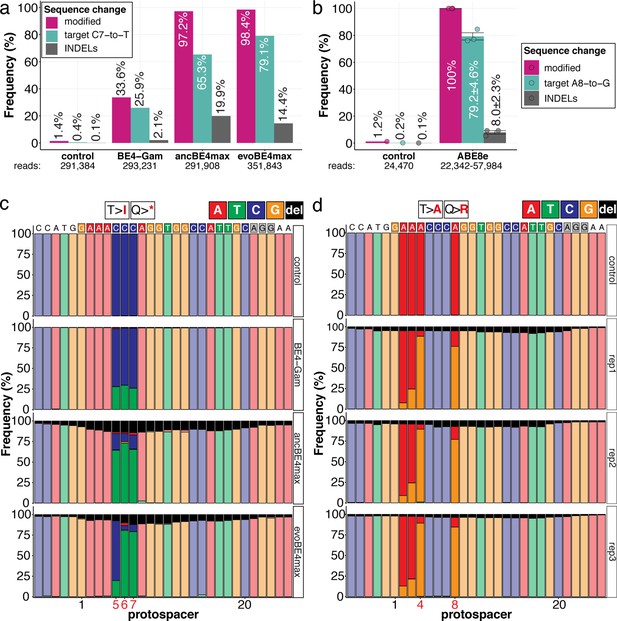
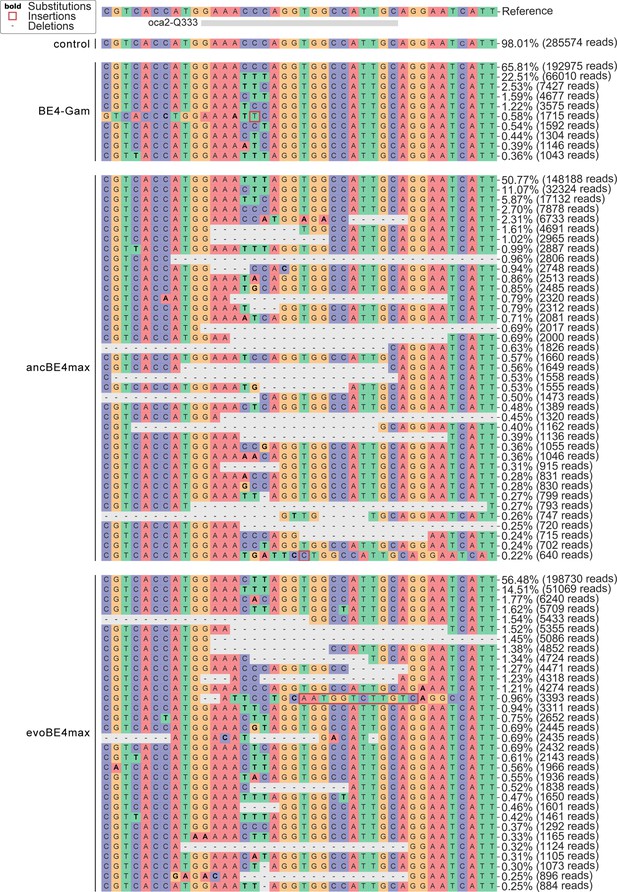
Amplicon-seq of cytosine base editors (a, c) and ABE8e (b, d) reveals prominent on-target editing efficiencies with low- to moderate levels of indels.
Genomic DNA samples from oca2-Q333 base editing experiments (Figure 2b–g) were used to query the outcome of intended base editing and indel formation by quantitative means by Illumina sequencing of the target region. Note: for oca2-Q333 control, BE4-Gam, ancBE4max, and evoBE4max, two pools of five embryos were used as sample input (a, c), whereas all three biological replicates of ABE8e samples were sequenced separately (b, d). The proportion of all reads aligned per sample to a reference is plotted, distinguishing (1) all modified reads, (2) target cytosine (C7, a) or adenine (A8, b) nucleotide changes and (3) INDELs. The number of reads shown (a, b) refers to all aligned Illumina reads per sample. The frequencies of base calls at the oca2-Q333 sgRNA target site ± 5 bp is shown for the three different cytosine editors (c) and ABE8e (d).
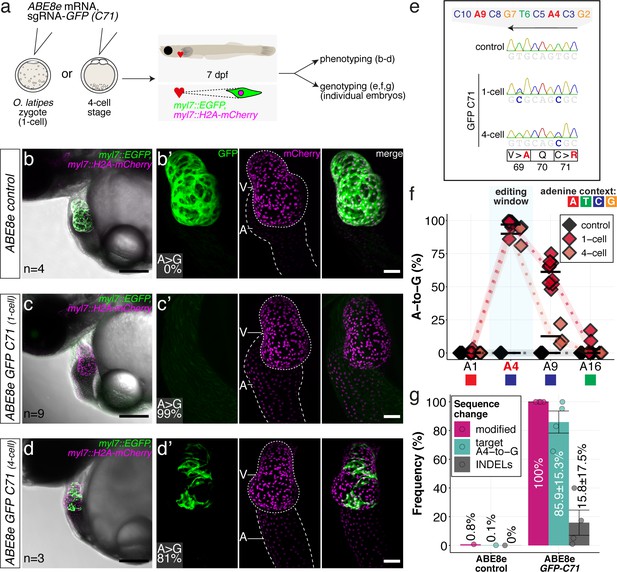
ABE8e efficiently introduces missense mutation and completely abolishes GFP fluorescence in F0.
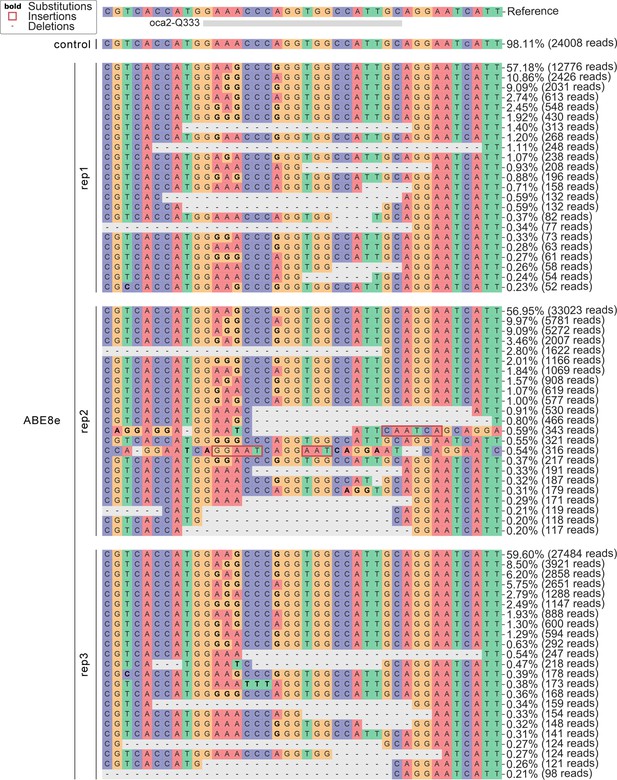
(a) Co-injection of ABE8e mRNA with GFP-C71 sgRNA into a single cell of one-cell or four-cell stage medaka embryos (myl7::EGFP, myl7::H2A-mCherry). Control injections with ABE8e mRNA. Scoring of fluorescence was performed at 7 dpf followed by genotyping of each individual embryo. Confocal microscopy of chemically arrested hearts (representative images) at 7 dpf (lateral view with V = ventricle, A = atrium). Overview images, overlaid with transmitted light, show maximum z-projections of optical slices acquired with a z-step size of 5 µm. Scale bar = 200 µm (b–d). Close-up images show maximum z-projections of optical slices acquired with a z-step size of 1 µm. Note the display of A-to-G conversion rates for A4 causing the p.C71R missense mutation (see g) and Supplementary file 2. Scale bar = 50 µm (b-d’). (e–f) Quantification of Sanger sequencing reads show close to homozygosity rates of A-to-G transversions installing the C71R missense mutation (Supplementary file 2). Note: sgRNA GFP-C71 targets the complementary strand (arrow in f). To highlight the dinucleotide context, the nucleotide preceding the target A is shown by red (A), green (T), blue (C) and yellow (G) squares below the respective A. dpf = days post fertilization. (g) Amplicon-seq of the target region a subset (n = 4) of 1 cell stage ABE8e experiment gDNA samples (single embryos) was used to quantify the outcome of intended base editing and indel formation. Aligned Illumina-reads analyzed, 14,653 (control); 23,201 (ABE8e rep1); 10,696 (ABE8e rep2); 66,311 (ABE8e rep3); 48,126 (ABE8e rep4).
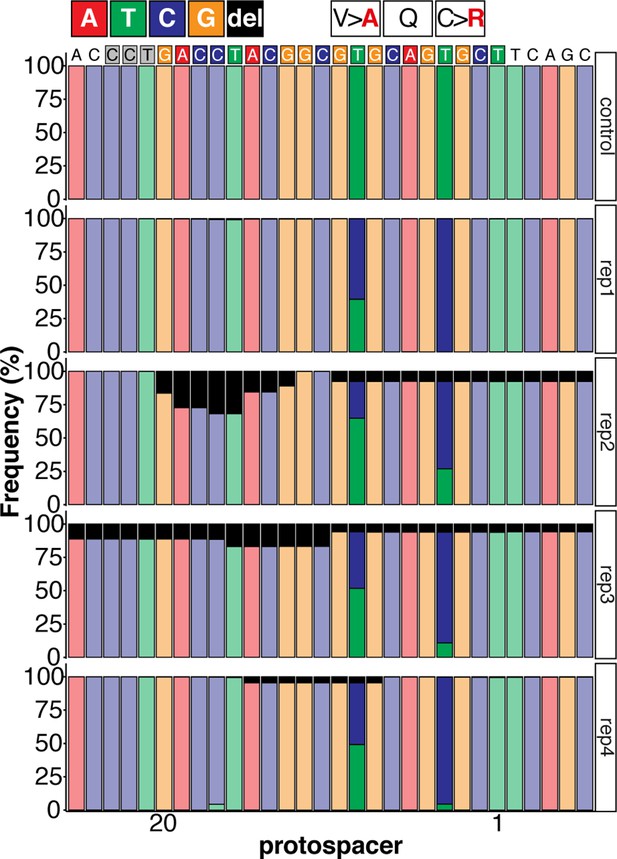
Sequence composition following Amplicon-seq of ABE8e GFP-C71 editants surrounding the GFP-C71 sgRNA target site ±5 bp.
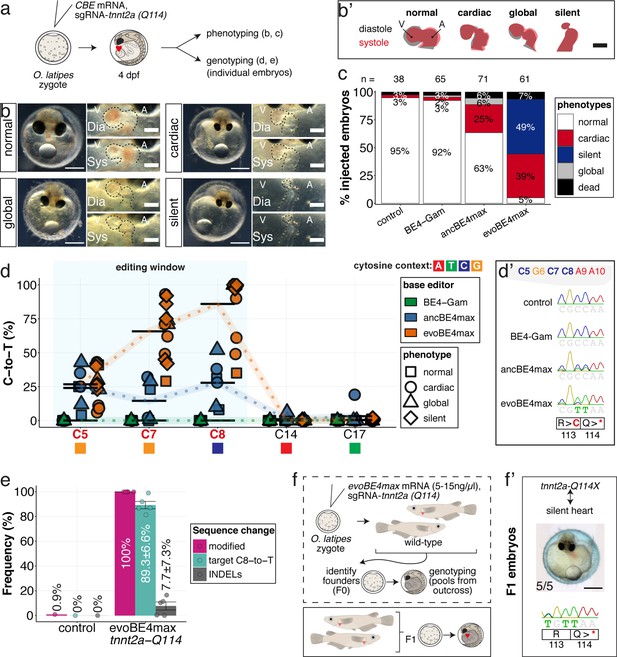
Stop-gain mutation in medaka troponin T gene by cytosine base editing accurately recreates mutant phenotypes in F0.
(a) Schematic diagram of protocol after co-injection of cytosine editor mRNA (comparing BE4-Gam, ancBE4max, evoBE4max) with sgRNA tnnt2a-Q114 (PTC) into one-cell stage medaka embryos; control injections only contained the sgRNA. (b) Editing of tnnt2a resulted in a range of phenotypes classified into five categories, including general morphogenic (global), dysmorphic but still functional heart chambers (cardiac), and non-contractile hearts (silent), where cardiac and silent phenotype groups displayed homogeneously additional developmental retardation. Scale bar = 400 µm (overview) and 100 µm (zoom-in). Ventricle (V), atrium (A), diastole (Dia) and systole (Sys) are indicated. (b') Representative scheme of fractional shortening of the heart chambers in specified phenotype groups highlighting significant morphological consequences (small ventricle) in the silent heart group. (c) Fraction of phenotype scores as a consequence of cytosine base editor injections. (d) Summary of editor type-specific C-to-T conversion efficiencies relative to the target C protospacer position grouped by phenotype class for BE4-Gam (n = 6), ancBE4max (n = 6) and evoBE4max (n = 12). To highlight the dinucleotide context, the nucleotide preceding the target C is shown by red (A), green (T), blue (C), and yellow (G) squares below the respective C. (d') Example Sanger sequencing reads of single edited embryos with resulting missense and stop-gain mutations through editing at C5, C7, and C8, respectively (e) Amplicon-seq of the target region of a subset (n = 5) of evoBE4max edited gDNA samples (single embryos) quantified target C8-to-T editing as well as indel frequencies. Aligned Illumina-reads analyzed, 7094 (control); 11,557 (evoBE4max rep1); 2561 (evoBE4max rep2); 2481 (evoBE4max rep3); 37,751 (evoBE4max rep4); 48,791 (evoBE4max rep5). (f) Phenotypic analysis of F1 tnnt2a-Q114X mutants revealed complete penetrance (n = 5) of the silent heart phenotype with same phenotypic profile as for F0 edits. dpf = days post fertilization.
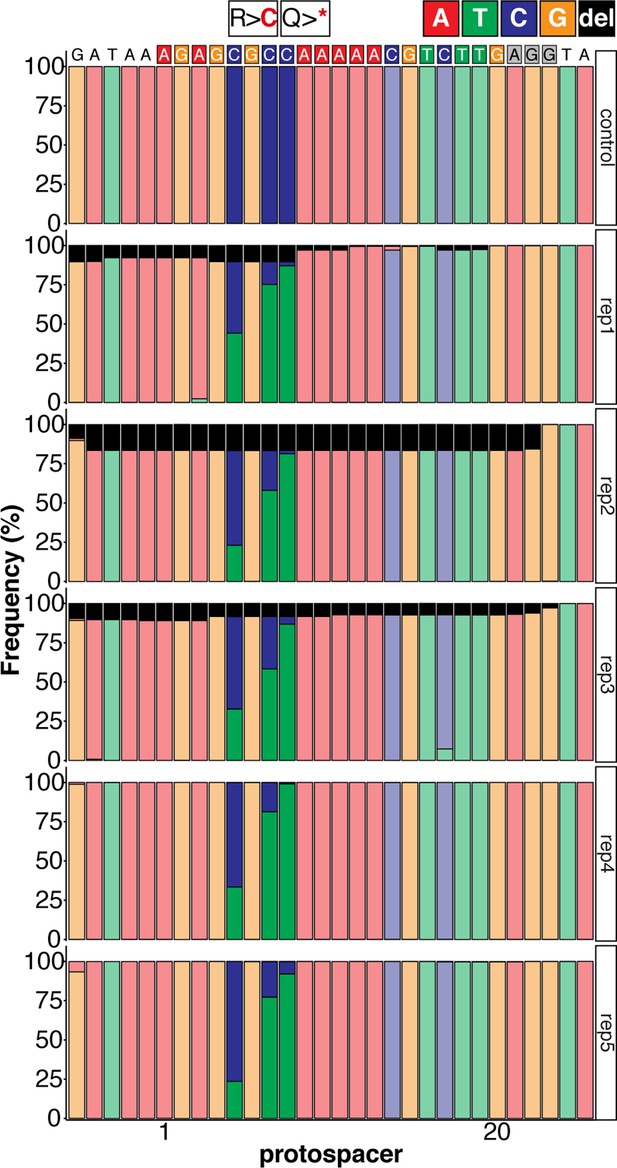
Sequence composition determined by Amplicon-seq of evoBE4max tnnt2a-Q114 editants surrounding the tnnt2a-Q114 sgRNA target site ± 5 bp.
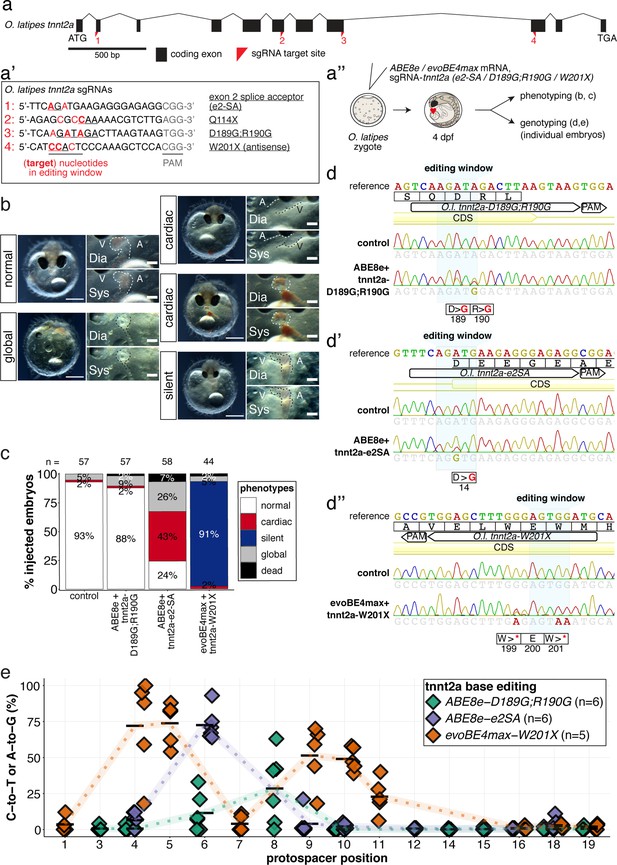
The installation of an additional PTC at tnnt2-W201X with evoBE4max leads to a recapitulation of the silent heart phenotype.
(a) O. latipes tnnt2a coding region with annotation of all sgRNAs (a’) targeting tnnt2a used in this study. (b) Overview of phenotypic categories and the quantification of phenotypic outcomes following base editing F0 tnnt2a base editing experiments (c). (d) Representative Sanger sequencing reads. (e) Summary of C-to-T or A-to-G conversion efficiencies.
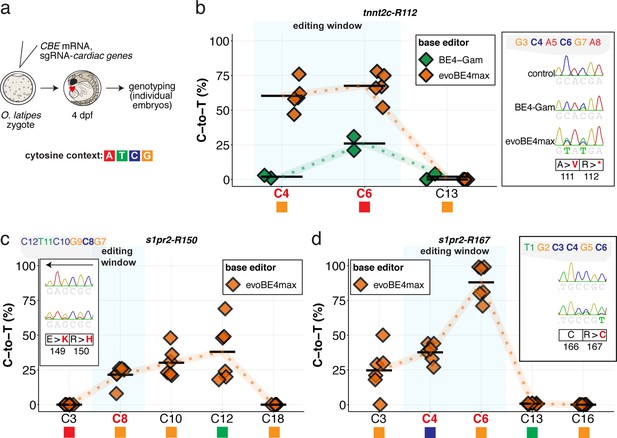
Cytosine base editing enables efficient installation of PTCs or missense mutations in two additional cardiac genes.
Summary of C-to-T conversion efficiencies. Sample number used for evoBE4max: n = 5, 6, 6, respectively for (b–d); and BE4-Gam: n = 2 (b). To highlight the dinucleotide context, the nucleotide preceding the target C is shown by red (A), green (T), blue (C) and yellow (G) squares below the respective C.
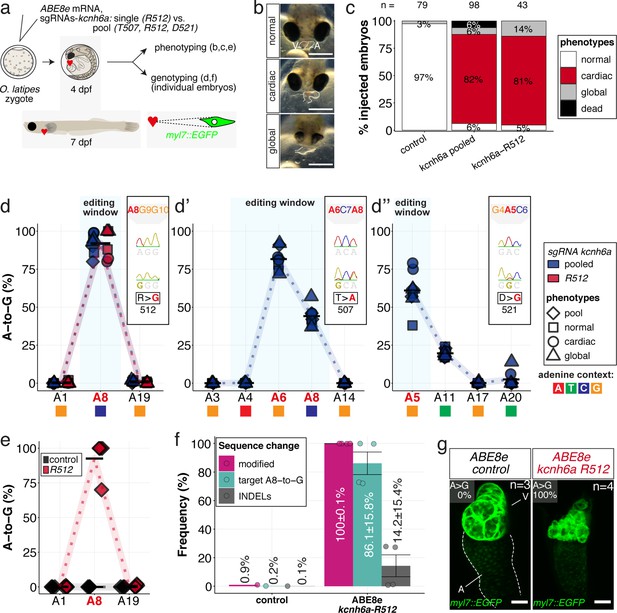
In vivo modeling of human LQTS-associated mutations using adenine base editing of the medaka ERG channel gene kcnh6a.
(a) Regime of ABE8e mRNA injections with a single (kcnh6a-R512) or pooled sgRNAs (kcnh6a-T507, -R512, -D521) targeting different amino acid codons in the voltage sensor S4 domain/S4-S5 linker of the medaka potassium channel ERG in myl7::EGFP (Cab strain) transgenic embryos; control injection included ABE8e mRNA only. (b) Phenotypes in F0 comprised primary cardiac malformation (dysmorphic ventricle with impaired contractility) and more severe global phenotypes with general retarded development and prominently dysmorphic hearts the proportions of which are given in (c). Scale bar = 400 µm. (d-d'') Genotyping summaries of the three sgRNA loci with phenotype class annotations for each genotyped specimen with a comparison of single sgRNA-R512 injection to a pool with two additional sgRNAs (T507 and D521) targeting the medaka ERG S4 voltage sensor; inlets display Sanger reads with the editing of A8 (d), A6 and A8 (d') and A5 (d'') contained in the core editing windows; sgRNA pool (n = 8) and sgRNA-R512 (n = 6). To highlight the dinucleotide context, the nucleotide preceding the target A is shown by red (A), green (T), blue (C) and yellow (G) squares below the respective A. (e–g) Confocal microscopy of the heart in a myl7::EGFP reporter line injected with ABE8e mRNA and sgRNA-R512 at 7 dpf reveals significant chamber wall defects of non-contractile/spastic ventricles with A-to-G editing of 100% in 3/4 of the specimen as determined by Sanger sequencing (e). (f) Amplicon-seq of the same gDNA samples (single embryos, n = 4) quantified target A8-to-G editing and indel frequencies. Aligned Illumina-reads analyzed, 11,387 (control); 24,936 (ABE8e rep1); 4038 (ABE8e rep2); 75,148 (ABE8e rep3); 86,327 (ABE8e rep4). Images show maximum z-projections of optical slices acquired with a z-step size of 1 µm (g). Note the display of A-to-G conversion rates. Scale bar = 50 µm. V = ventricle, A = atrium, dpf = days post fertilization.
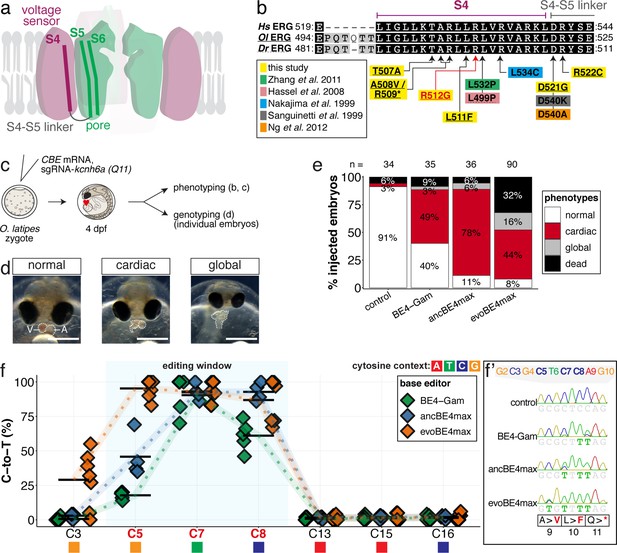
Robustness of interrogating gene function by introducing stop-gain mutations in medaka demonstrated at the N-terminal kcnh6a-Q11 locus in F0 through CBEs.
(a) Cartoon of ERG structure according to Wang and MacKinnon, 2017, highlighting the S4-6 domains. (b) Summary of mutations in the S4 domain in model systems studying ERG function in human (Hs ERG), medaka (Ol ERG) and zebrafish (Dr ERG) (Hassel et al., 2008; Nakajima et al., 1999; Ng et al., 2012; Sanguinetti and Xu, 1999; Zhang et al., 2011). (c) Workflow. Control injections only contained the sgRNA. (d) Phenotypes observed following editing of kcnh6a were grouped by being either isolated ‘cardiac’ phenotypes or with a ‘saturated’ outcome of ventricular asystole concomitant with additional unspecific developmental defects (‘global’). (e) Fraction of phenotype scores as a function of base editor generation. (f) Summary of editor type-specific C-to-T conversion efficiencies relative to the target C protospacer position for BE4-Gam (n = 5), ancBE4max (n = 4) and evoBE4max (n = 7). To highlight the dinucleotide context, the nucleotide preceding the target C is shown by red (A), green (T), blue (C) and yellow (G) squares below the respective C. (f') Example Sanger sequencing reads of single edited embryos with resulting missense and stop-gain mutations through editing at C3, C5, C7, and C8, respectively.
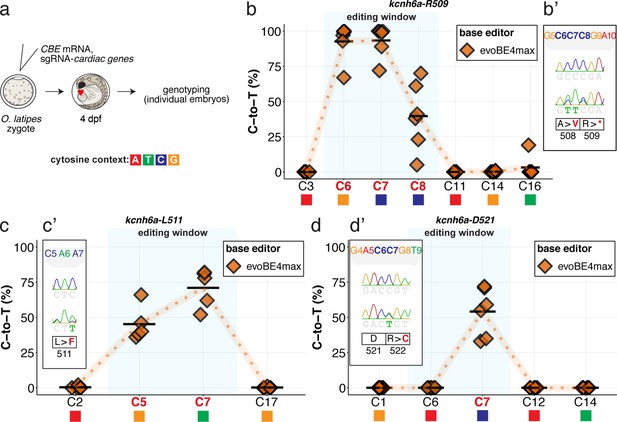
evoBE4max cytosine base editing enables efficient installation of PTCs or missense mutations at three additional kcnh6a loci.
Summary of C-to-T conversion efficiencies relative to the target C protospacer position for Sample number: n = 6, 4, 6 respectively (b–d). To highlight the dinucleotide context, the nucleotide preceding the target C is shown by red (A), green (T), blue (C) and yellow (G) squares below the respective C.
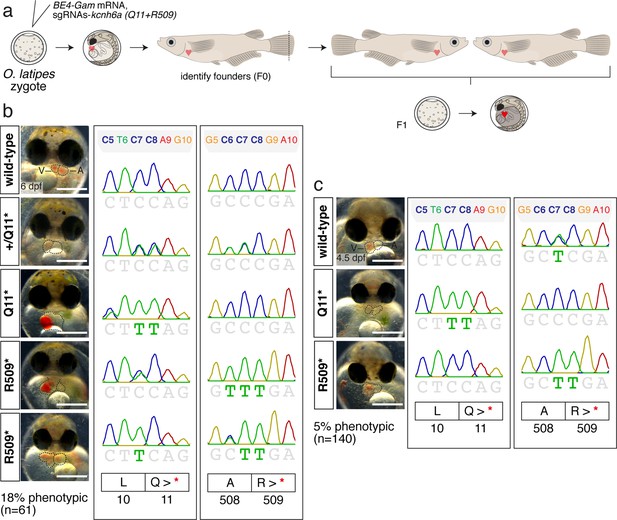
Analysis of BE4-Gam mediated PTC installation in kcnh6a in F1.
(a) Injected embryos (BE4-Gam mRNA, pooled sgRNAs kcnh6a-Q11 and -R509) were raised to adulthood. Identified founders were crossed and F1 embryos further examined. (b–c) Analysis of F1 embryos at 6 dpf (b) and 4.5 dpf (c) from two independent founder couples.
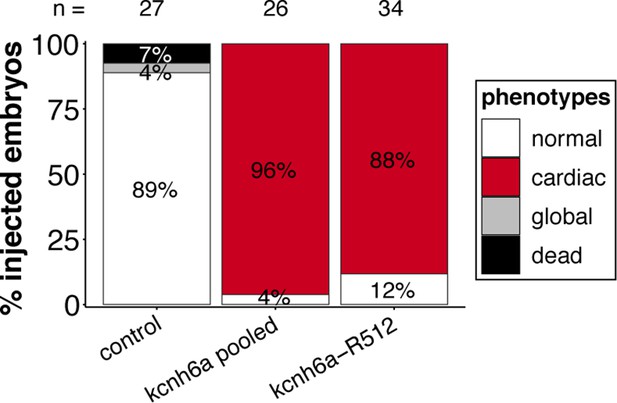
Targeting the three kcnh6a loci simultaneously or kcnh6a-R512 alone in a different genetic background (myl7::EGFP, myl7::H2A-mCherry; HdrR strain) recapitulates phenotypic proportions.
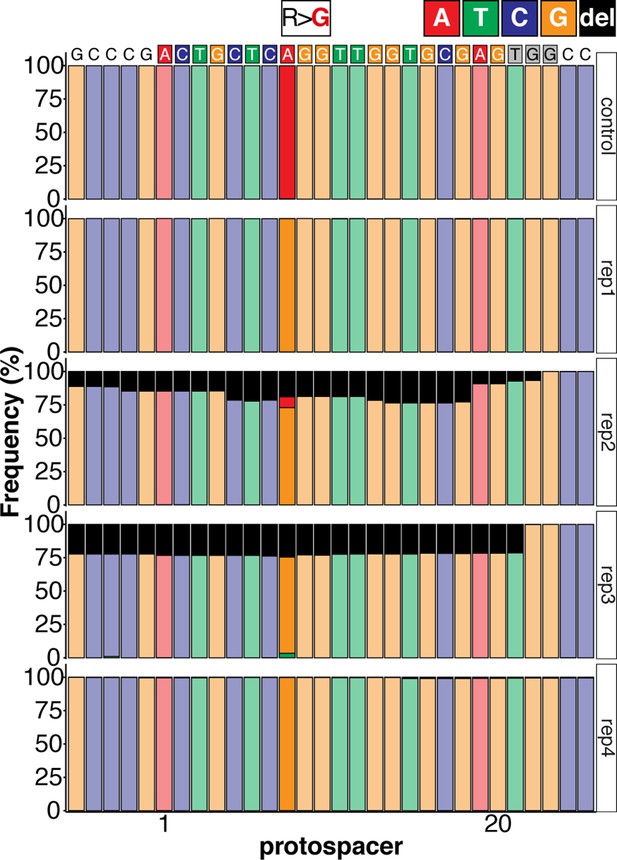
Sequence composition determined by Amplicon-seq of ABE8e kcnh6a-R512 editants surrounding the kcnh6a-R512 sgRNA target site ± 5 bp.
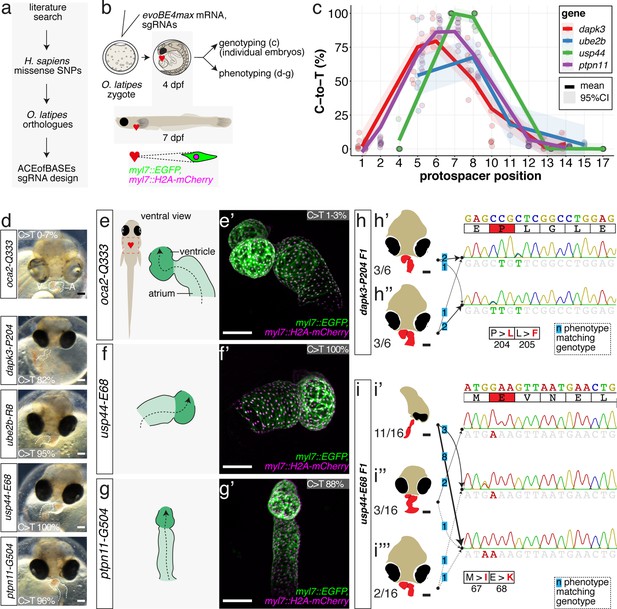
Cytosine base editing enables human CVD-associated SNV validation.
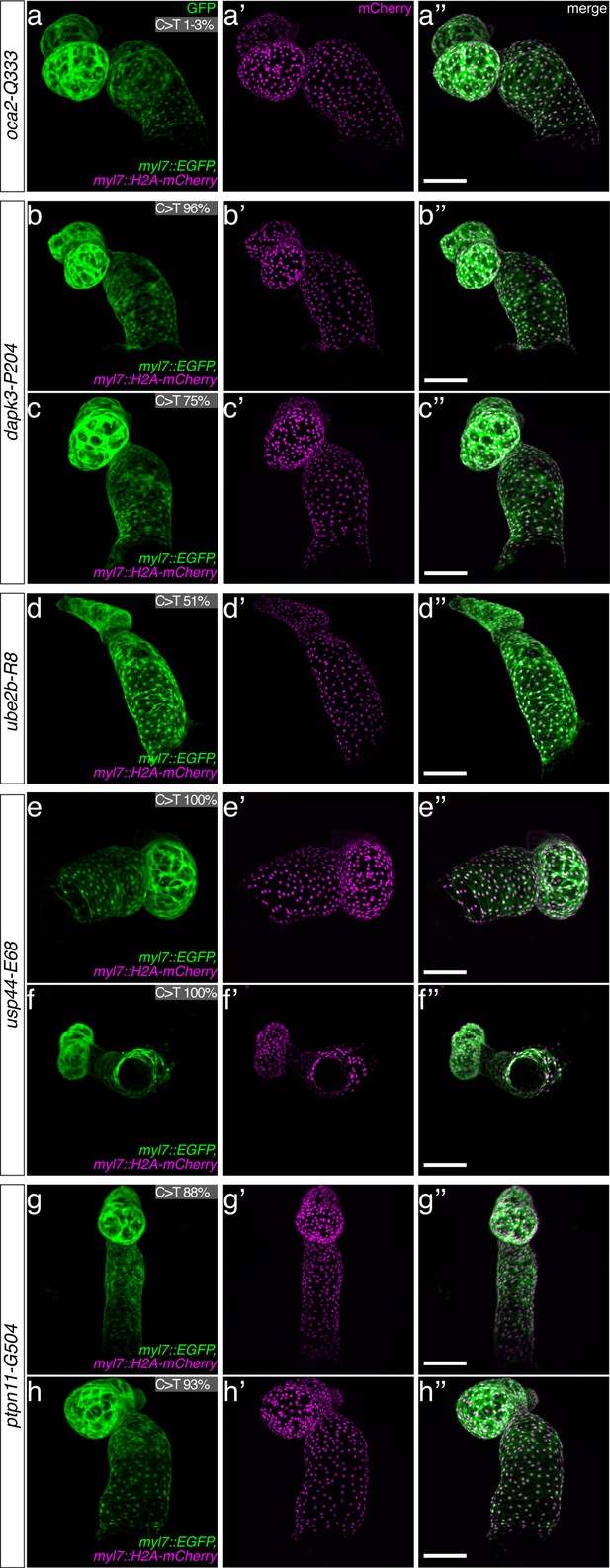
(a) Candidate human CVD gene SNV validation workflow. (b) To target the SNVs evoBE4max mRNA was co-injected into the 1 cell stage of the medaka wild-type or myl7::EGFP, myl7::H2A-mCherry reporter strain together with the corresponding target or oca2-Q333 (control) sgRNAs. Individual, imaged, embryos were then further analyzed to determine the rate of C-to-T transversions. (c) Cytosine editing efficiencies are substantial for all candidate genes tested. Data shown in Figure 7—figure supplement 2 was replotted, including all data points from a-d across all target cytosine along the protospacer. Sample numbers: dapk3-P204 (n = 7), ube2b-R8 (n = 5), usp44-E68 (n = 11), and ptpn11-G504 (n = 11). (d) Representative phenotypes of 4 dpf base edited embryos are shown for all four tested candidate CVD genes including oca2-Q333 controls. Top, ventral view, with V = ventricle, A = atrium. (e–g) Confocal microscopy of selected candidate validations in the reporter background. Hearts were imaged in 7 dpf hatched double fluorescent embryos. Images show maximum projections of the entire detectable cardiac volume with a step size of 1 µm. Cartoons (left) highlight the looping defects observed in usp44 and ptpn11 base edited embryos with ventricle-atrium inversion (f) or tubular heart (g). (e’-g’) Imaged embryos were subsequently genotyped and quantified C-to-T transversions for the target codon are shown. Note: due to the inverted nature of the confocal microscope used, raw images display a mirroring of observed structures, which we corrected here for simpler appreciation. Phenotypic analysis of F1 dapk3-P204L (h) and usp44-E68K (i) embryos revealed that homozygous changes at P204L or E68K lead to cardiac malformations with varying degree: looping (h’) and mild looping defects (h’’, i’’’); altered heart morphology (i’’). Bystander edits (hetero- or homozygous, usp44-E68K) lead to additional developmental defects, including brain and eye abnormalities (i’). Scale bar = 100 µm (d, e–i). dpf = days post fertilization.
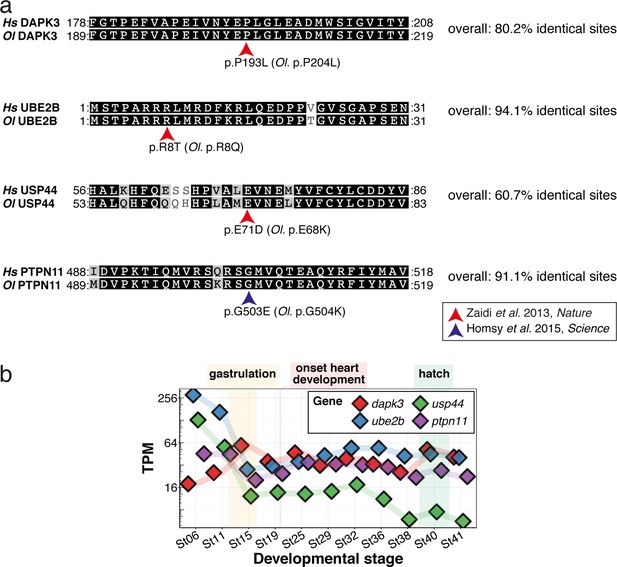
CVD-associated SNVs can be mapped to the orthologous medaka peptide sequence with high conservation and are expressed during heart development.
(a) Human (Hs) coding sequences for DAPK3, UBE2B, USP44, and PTPN11 around the missense mutations reported (Homsy et al., 2015; Zaidi et al., 2013), were obtained from the Ensemble genome browser and the orthologous medaka (Ol) sequences were mapped to these (release 103). (b) Gene expression data for medaka dapk3, ube2b, usp44, and ptpn11 at embryonic stages was extracted from Li et al., 2020 and plotted to show presence of transcript during heart development and the contribution of maternal transcripts, which dominate before zygotic genome activation at around stages 7–8 (32–64 cell stages; Kraeussling et al., 2011).
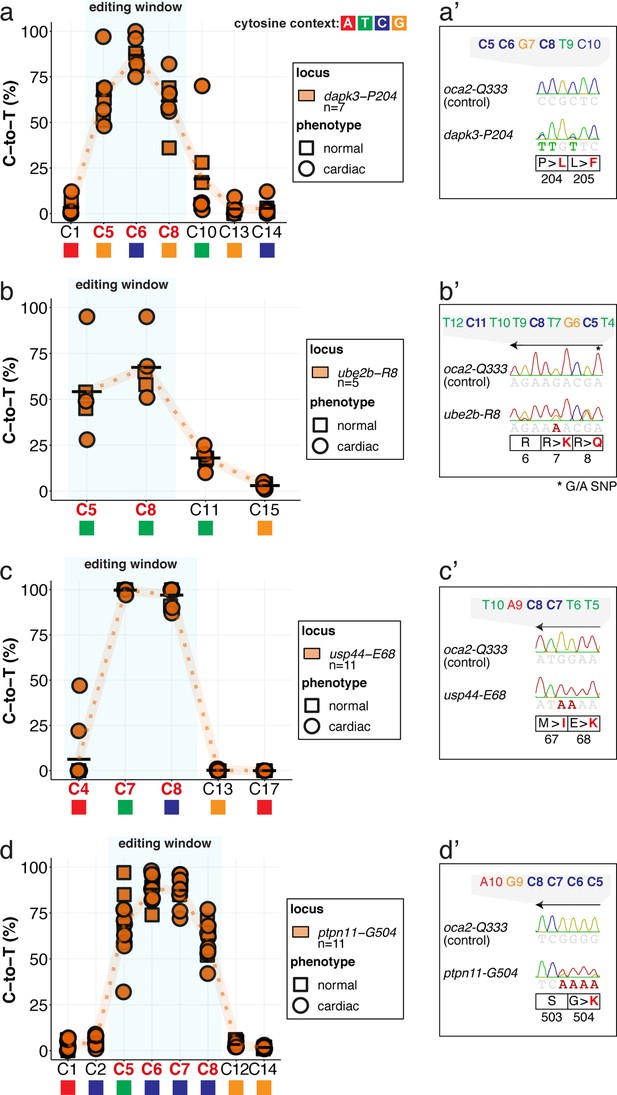
Cytosine base editing allows the introduction of human CVD-associated missense mutations in medaka in F0.
(a–d) Following microinjection at the 1 cell stage of evoBE4max together with the respective sgRNAs, Sanger sequencing results were quantified by EditR (Kluesner et al., 2018). Results are plotted for the genomic loci of dapk3-P204 (n = 7), ube2b-R8 (n = 5), usp44-E68 (n = 11), and ptpn11-G504 (n = 11) and are depicted for the underlying phenotype (square: normal; circle: cardiac) according to the phenotypes grouped in supplement 3. The standard base editing window is depicted in blue. To highlight the dinucleotide context, the nucleotide preceding the target C is shown by red (A), green (T), blue (C) and yellow (G) squares below the respective C. (a’-d’) Sanger sequencing reads within the region of edited amino acids for the respective sgRNA experiment and locus. (b’-d’) sgRNAs mediating the C-to-T transition map to the complementary strand. Note: nucleotide position 27 shows a G/A SNP in the Cab genetic background used in this study for ube2b.
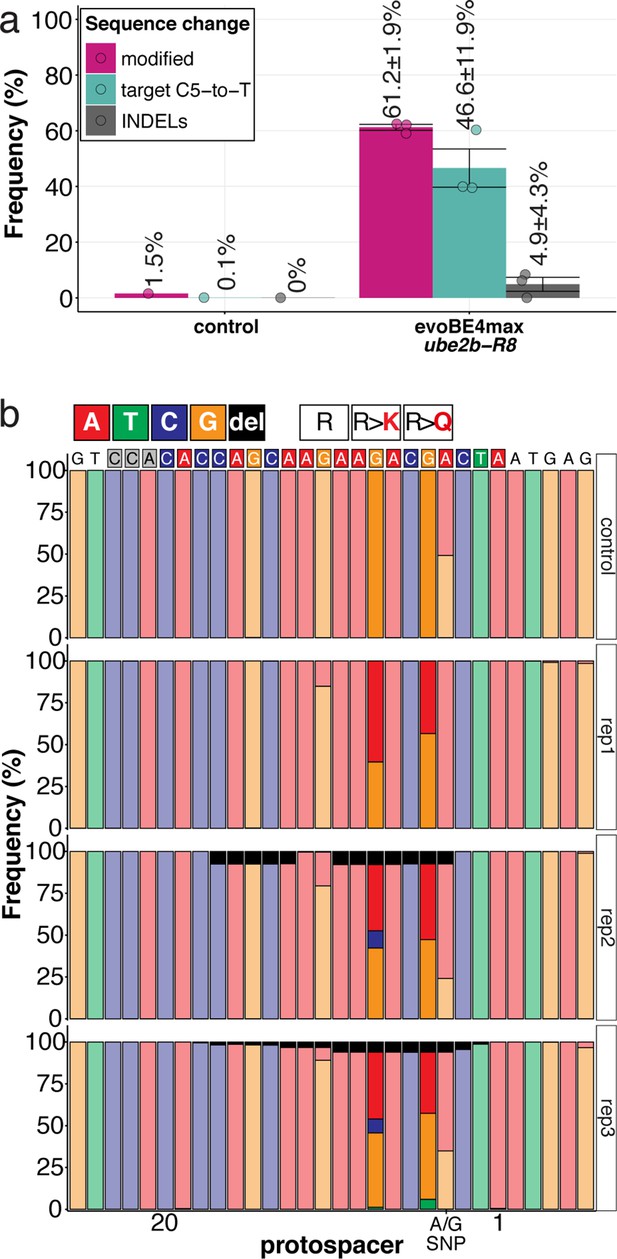
Amplicon-seq of ube2b-R8 evoBE4max editants.
(a) Illumina sequencing of the target region in a subset (n = 3) of gDNA samples (single embryos) reveals quantitative target C5-to-T editing efficiencies as well as indel frequencies by. Aligned Illumina-reads analyzed, 21,768 (control); 24,613 (evoBE4max rep1); 57,332 (evoBE4max rep2); 51,273 (evoBE4max rep3). (b) Sequence composition following Amplicon-seq analysis surrounding the ube2b-R8 sgRNA target site ± 5 bp.
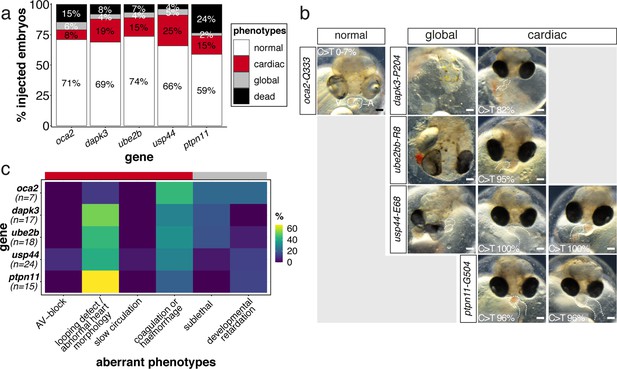
Phenotypic categorization of cytosine base edited embryos in medaka in F0.
(a) Microinjection of evoBE4max with the respective sgRNA for dapk3 (n = 74), ube2b (n = 91), usp44 (n = 79), and ptpn11 (n = 85) results in an increase in cardiovascular-associated phenotypes (‘cardiac’) compared to oca2 control editing (n = 62). (b) Exemplary phenotypes of 4 dpf embryos for the categories given in (a) (ventral view) with highlighting of the ventricle (V) and atrium (A). Here, embryos with morphological abnormalities or looping defects are shown in the ‘cardiac’ category. Quantification of base editing efficiencies (supplement 2) for the individual embryos is shown for the oca2 control (here: sequencing results for the four candidate loci given as range) and the ‘cardiac’ examples of the respective experiment. Scale bar = 100 µm. (c) Sub-categorization of the ‘cardiac’ and ‘global’ phenotypic groups shown as percentage of the sum of the two categories. dpf = days post fertilization.
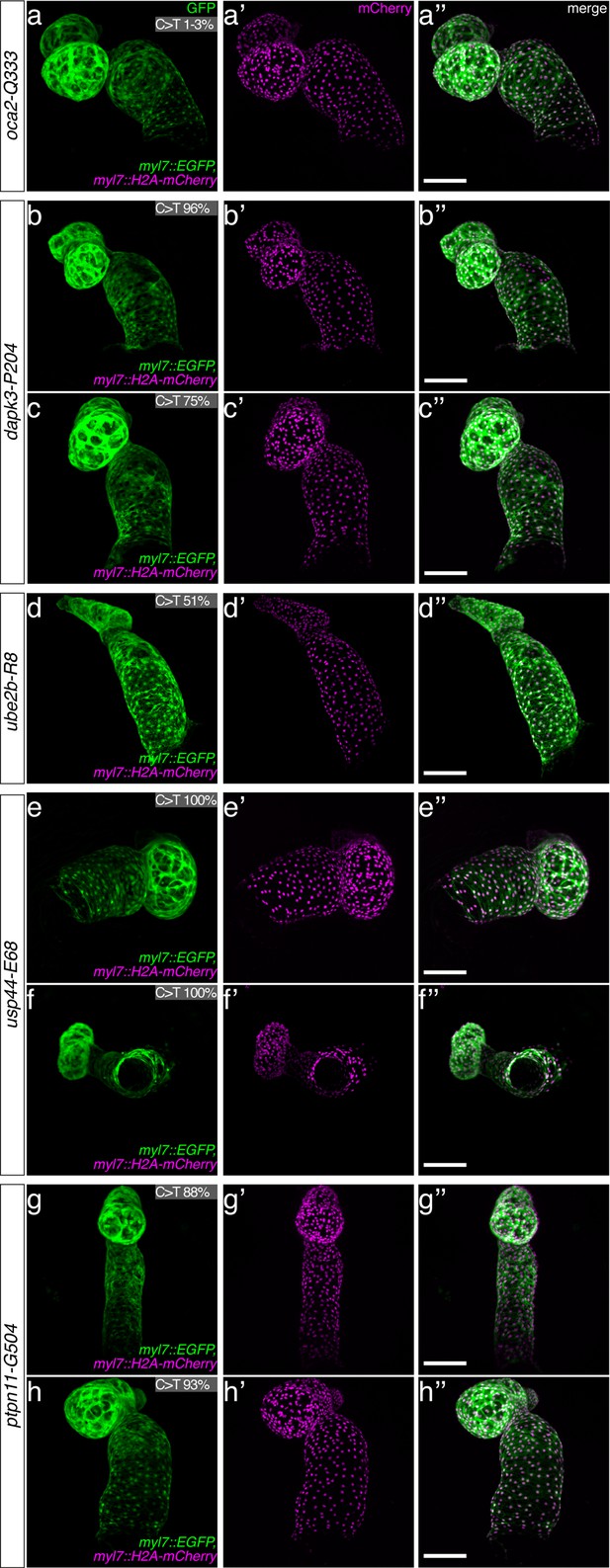
Confocal microscopy of evoBE4max validated CVD genes.
The full complement of analyzed CVD associated phenotypes in cytosine base editing of myl7::GFP, myl7::H2A-mCherry reporter embryos at 7 dpf. Note the mirroring effect of the confocal microscope used, placing the ventricle to the right, and the atrium to the left in the control. Quantified C-to-T transversions are shown for each imaged embryo for the target codon. Scale bar = 100 µm. dpf = days post fertilization.
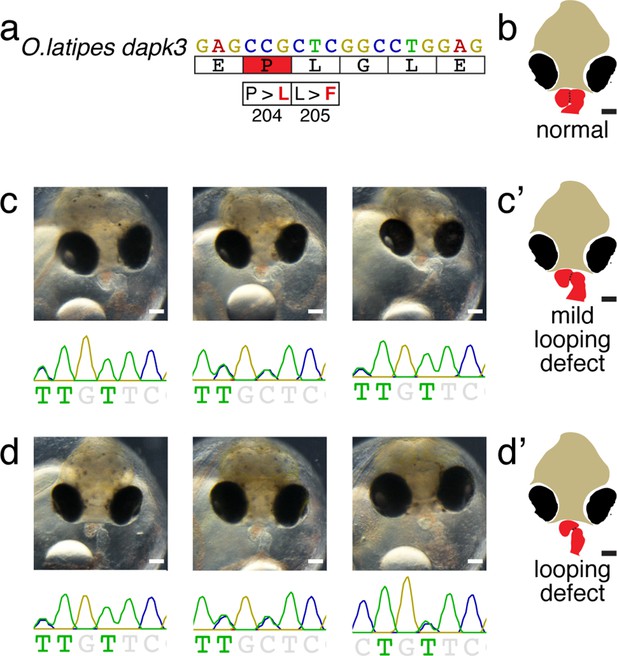
Phenotype-genotype correlation of dapk3-P204L/L205F embryos with mild (c) and moderate looping defects (d).
Cartoons (c’ and d’) are shown in the main figure as summary of the phenotypic manifestation.
Phenotype-genotype correlation of usp44-M67I/E68K embryos with mild looping defects (c), altered heart morphology with slight developmental delay (d) and severe alterations of heart morphology with concomitant strong global developmental defects (e).
Cartoons (c’, d’ and e’) are shown in the main figure as summary of the phenotypic manifestation.
Recapitulation of in vitro base editing characteristics combined with a plethora of conserved variants make fish excellent models to validate human pathogenic SNVs.
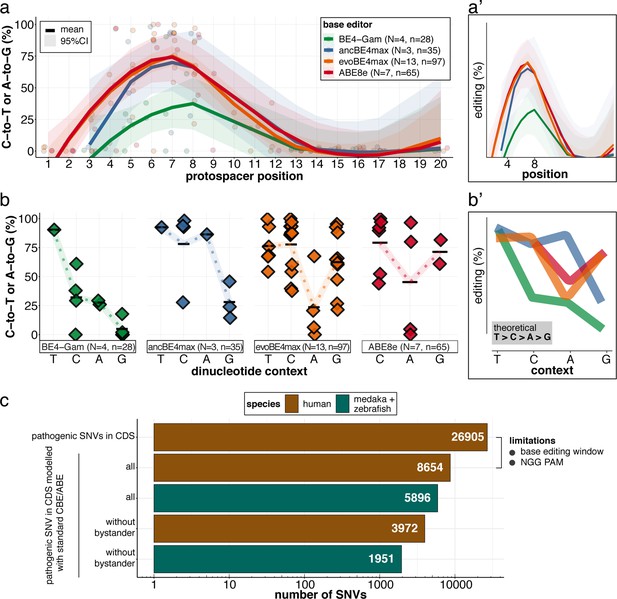
(a) Transition efficiencies for CBEs and ABE8e tested in this study in medaka across the entire protospacer are shown as mean with 95% confidence interval (CI). Each data point represents the mean efficiency for the locus (sgRNA) tested. N = represents the number of loci tested with the respective editor. n = total number of genomes used for quantification of editing efficiencies (a pool of five embryos was counted as five genomes). (a’) Simplified scheme only showing mean and CI. (b) Summary of dinucleotide preference for the tested base editors, calculated for the standard editing window (4-8). Each data point represents the mean editing efficiency of the corresponding editor for a particular protospacer position. (b’) Simplified scheme of overlaid dinucleotide logic. (c) Analysis of human pathogenic CDS SNVs annotated in ClinVar reveals that a remarkable portion of these SNVs have orthologous sequences in medaka or zebrafish that can be mimicked by CBEs or ABEs following editing window (4-8) and NGG PAM restrictions. Modeling SNVs mutations may be achieved with stringent criteria (no bystander mutations accepted, n = 1951) or less stringent selection (allowing bystander mutations ‘all’, n = 5896). Note: the number of SNVs shown for medaka + zebrafish, corresponds to a set-up in which these species are complementing each other.
Videos
evoBE4max introduced premature STOP codon in O.latipes tnnt2a results in silent heart phenotype.
Time-lapse movie (10 seconds) of the beating medaka heart. Scale bar = 400 µm.
evoBE4max introduced premature STOP codon in O.latipes kcnh6a results in ventricular asystole accompanied by morphological alterations.
Time-lapse movie (10 seconds) of the beating medaka heart. Scale bar = 400 µm.
ABE8e driven installation of the R512G missense mutation in O.latipes kcnh6a results in ventricular asystole accompanied by morphological alterations.
Time-lapse movie (10 s) of the beating medaka heart. Scale bar = 400 µm.
Tables
Estimated editing windows and dinucleotide preference affecting editing efficiencies.
Comparison of literature estimates (in vitro) and in vivo metrics observed in this study.
| Base editor | Highest average editing efficiency (site tested) | Editing window on protospacer: overall (peak) activity | Dinucleotide sequence preference | ||
|---|---|---|---|---|---|
| In vitro* | This study | In vitro* | This study | ||
| BE4-Gam | 61.0 ± 10.4(kcnh6a-p.Q11X) | 3–10 (4-8) | 4–8 (NA) | canonical | canonical |
| ancBE4max | 93.8% ± 7.9% (oca2-p.Q333X) | 3–9 (4-7) | NA (5-8) | << GC | << GC |
| evoBE4max | 99.7% ± 0.9% (usp44-p.E68K) | 1–11 (4-8) | 3–12 (5-8) | << AC | << AC |
| ABE8e | 100% (oca2-p.Q256R) | 3–11 (4-8) | 3–11 (4-8) | - | < AC |
-
NA – not sufficient data to estimate.
-
*
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Strain, strain background (Oryzias latipes) | Cab | Loosli et al., 2000 | N/A | medaka Southern wild type population; Wittbrodt lab |
| Strain, strain background (Oryzias latipes) | Cab (myl7::EGFP) | Gierten et al., 2020 | N/A | Wittbrodt lab |
| Strain, strain background (Oryzias latipes) | HdrR (myl7::EGFP myl7::H2A-mCherry) | Hammouda et al., 2021 | N/A | Wittbrodt lab |
| Strain, strain background (Danio rerio) | AB | ZIRC | ZFIN: ZBD-GENO-960809–7, RRID:ZIRC_ZL1 | Wildtype zebrafish strain |
| Recombinant DNA reagent | pGGEV_4_BE4-Gam (plasmid) | Thumberger et al., 2022 | N/A | Plasmid vector for in vitro transcription of BE4-Gam mRNA; Wittbrodt lab |
| Recombinant DNA reagent | pCMV_AncBE4max, (plasmid) | Addgene | Addgene plasmid #112094; http://n2t.net/addgene:112094; RRID:Addgene_112094 | Plasmid vector for expression or in vitro transcription of ancBE4max mRNA from pCMV; Liu lab |
| Recombinant DNA reagent | pBT281(evoAPOBEC1-BE4max) (plasmid) | Addgene | Addgene plasmid #122611; http://n2t.net/addgene:122611; RRID:Addgene_122611 | Plasmid vector for expression of evoBE4max in mammalian cells; Liu lab |
| Recombinant DNA reagent | pCS2+ (plasmid) | Rupp et al., 1994 | Xenbase: XB-VEC-1221270 | high-level transient expression for mRNA synthesis and injection numerous aquatic organisms |
| Recombinant DNA reagent | pCS2+_evoBE4max | This paper | N/A | See Materials and methods |
| Recombinant DNA reagent | pBABE8e (plasmid) | Addgene | Addgene plasmid #138489; http://n2t.net/addgene:138489; RRID:Addgene_138489 | Plasmid vector for expression or in vitro transcription of ABE8e mRNA from pCMV; Liu lab |
| Recombinant DNA reagent | DR274 (plasmid) | Addgene | Addgene plasmid #42250; http://n2t.net/addgene:42250; RRID:Addgene_42250 | sgRNA expression vector to create sgRNA to a specific sequence with T7 promoter for in vitro transcription |
| Recombinant DNA reagent | oligonucleotides | Eurofins Genomics | PCR primers, sgRNA cloning primers, sequencing primers | see Materials and Methods |
| Gene (Oryzias latipes) | oca2 | Ensemble genome browser | Ensemble (release 96): ENSORLG00000015893 CDS | Medaka OCA2 melanosomal transmembrane protein |
| Gene (Danio rerio) | oca2 | Ensemble genome browser | Ensemble (release 103):ENSDARG00000061303.8 CDS | Zebrafish OCA2 melanosomal transmembrane protein |
| Gene (Homo sapiens) | oca2 | Ensemble genome browser | Ensemble (release 103):ENSG00000104044.16 CDS | OCA2 melanosomal transmembrane protein |
| Gene (Oryzias latipes) | tnnt2a | Ensemble genome browser | Ensemble (release 95): ENSORLG00000024544.1 CDS | Medaka cardiac muscle-like troponin T |
| Gene (Oryzias latipes) | kcnh6a | Ensemble genome browser | Ensemble (release 93): ENSORLG00000002317.1 CDS | Encodes Ol ERG |
| Gene (Danio rerio) | kcnh6a | Ensemble genome browser | Ensemble (release 103): ENSDARG00000001803.12 CDS | Encodes Dr ERG |
| Gene (Homo sapiens) | kcnh2 | Ensemble genome browser | Ensemble (release 103): ENSG00000055118.16 CDS | Encodes Hs ERG |
| Gene (Oryzias latipes) | tnnt2c | Ensemble genome browser | Ensemble (release 93): ENSORLG00000016386.1 CDS | Medaka cardiac troponin T2c |
| Gene (Oryzias latipes) | s1pr2 | Ensemble genome browser | Ensemble (release 93): ENSORLG00000005560.1 CDS | Medaka sphingosine-1-phosphate receptor 2 |
| Gene (Oryzias latipes) | dapk3 | Ensemble genome browser | Ensemble (release 103): ENSORLG00000017965.2 CDS | Medaka death associated protein kinase 3 |
| Gene (Homo sapiens) | dapk3 | Ensemble genome browser | Ensemble (release 103): ENSG00000167657.14 CDS | Human death associated protein kinase 3 |
| Gene (Oryzias latipes) | ube2b | Ensemble genome browser | Ensemble (release 103): ENSORLG00000000951.2 CDS | Medaka ubiquitin conjugating enzyme E2 B |
| Gene (Homo sapiens) | ube2b | Ensemble genome browser | Ensemble (release 103): ENSG00000119048, CCDS4174 | Human ubiquitin conjugating enzyme E2 B |
| Gene (Oryzias latipes) | usp44 | Ensemble genome browser | Ensemble (release 103): ENSORLG00000016627.3 CDS | Medaka ubiquitin specific peptidase 44 |
| Gene (Homo sapiens) | usp44 | Ensemble genome browser | Ensemble (release 103): ENSG00000136014.12 CDS | Human ubiquitin specific peptidase 44 |
| Gene (Oryzias latipes) | ptpn11 | Ensemble genome browser | Ensemble (release 103): ENSORLG00000000470.2 CDS | Medaka ptpn11a - protein tyrosine phosphatase non-receptor type 11 |
| Gene (Homo sapiens) | ptpn11 | Ensemble genome browser | Ensemble (release 103): ENSG00000179295.18 CDS | Human protein tyrosine phosphatase non-receptor type 11 |
| Commercial assay, kit | NEBuilder HiFi DNA Assembly Cloning kit | New England Biolabs | Catalog #E5520S | |
| Commercial assay, kit | mMessage mMachine Sp6 Transcription Kit | Thermo Fisher Scientific | Catalog #AM1340 | |
| Commercial assay, kit | mMessage mMachine T7 Transcription Kit | Thermo Fisher Scientific | Catalog #AM1344 | |
| Commercial assay, kit | T7 MEGAscript Kit | Thermo Fisher Scientific | Catalog #AM1334 | |
| Commercial assay, kit | InnuPREP Gel Extraction Kit | Analytik Jena | Catalog #845-KS-5030250 | |
| Commercial assay, kit | Monarch DNA Gel Extraction Kit | New England Biolabs | Catalog #T1020 | |
| Commercial assay, kit | RNeasy Mini Kit | Qiagen | Catalog #74,106 | |
| Peptide, recombinant protein | Q5 High-Fidelity DNA Polymerase | New England Biolabs | Catalog #M0491 | |
| Peptide, recombinant protein | Q5 Hot High-Fidelity DNA Polymerase | New England Biolabs | Catalog #M0493 | |
| Chemical compound, drug | 2,3-Butanedione 2-monoxime (BDM) | Abcam | Catalog #ab120616 | |
| Chemical compound, drug | N-Phenylthiourea (PTU) | Sigma-Aldrich | Catalog #P7629 | |
| Chemical compound, drug | Ethyl 3-aminobenzoate methanesulfonate salt (Tricaine) | Sigma-Aldrich | Catalog #A5040 | |
| Software, algorithm | ACEofBASEs | This paper | https://aceofbases.cos.uni-heidelberg.de | |
| Software, algorithm | EditR | Kluesner et al., 2018 | https://moriaritylab.shinyapps.io/editr_v10/ | |
| Software, algorithm | Geneious | Biomatters | Version 8.1.9 | |
| Software, algorithm | Fiji distribution of ImageJ | Schindelin et al., 2012 | Version 2.0.0 | |
| Software, algorithm | Adobe Illustrator | Adobe | Version 23.2.1 | |
| Software, algorithm | R, R studio | R Development Core Team, 2020 | https://www.R-project.org/ | |
| Software, algorithm | R package | Wickham et al., 2019 | Tidyverse | |
| Software, algorithm | R package | Wickham, 2016 | ggplot2 | |
| Software, algorithm | R package | Kassambara, 2020 | ggpubr | |
| Software, algorithm | R package | Wickham, 2011 | plyr | |
| Software, algorithm | R package | Wickham et al., 2020 | dplyr |
Additional files
-
Supplementary file 1
Cytosine base editing efficiencies.
Shows nucleotide position (of CDS) and corresponding amino acid with changes. Note: only cytosines on the protospacer with clear editing are shown.
*sgRNA on complementary strand
- https://cdn.elifesciences.org/articles/72124/elife-72124-supp1-v2.docx
-
Supplementary file 2
Adenine base editing efficiencies for ABE8e.
Shows nucleotide position (of CDS) and corresponding amino acid with changes. Note: only adenines on the protospacer with clear editing are shown.
*sgRNA on complementary strand
#averaged over single oca2-Q333 and pooled injections
- https://cdn.elifesciences.org/articles/72124/elife-72124-supp2-v2.docx
-
Supplementary file 3
Cytosine base editing efficiencies at GWAS validation genes.
Shows nucleotide position (of CDS) and corresponding amino acid with changes. Note: only cytosines on the protospacer with clear editing are shown.
*sgRNA on complementary strand
- https://cdn.elifesciences.org/articles/72124/elife-72124-supp3-v2.docx
-
Supplementary file 4
Editing efficiency estimation by Sanger vs Illumina sequencing.
Comparison of target nucleotide editing efficiency and overview of indel frequency from Illumina data.
- https://cdn.elifesciences.org/articles/72124/elife-72124-supp4-v2.docx
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/72124/elife-72124-transrepform1-v2.docx
-
Source code 1
ACEofBASEs source code.
- https://cdn.elifesciences.org/articles/72124/elife-72124-code1-v2.zip