Tumor Metastasis: Fishing for drugs
About 90% of all cancer-related deaths are caused by metastasis, which is when cancer cells spread to other parts of the body to form new tumors (Chaffer and Weinberg, 2011; Suhail et al., 2019). Yet, the majority of currently available therapeutics do not inhibit metastasis, and only target the primary tumor where the cancer initially arises from.
To screen anti-cancer drugs, researchers often carry out experiments on mice or cells grown in the laboratory. While these model systems have led to effective treatments, they have limitations when it comes to testing drugs that block metastasis. For instance, cells cultured in the laboratory cannot accurately replicate tumor progression in humans (Katt et al., 2016), and metastasis can take at least several weeks to appear in mouse models, which are expensive to create and maintain (Simons and Brayton, 2017). Now, in eLife, Joji Nakayama, Zhiyuan Gong and co-workers report an innovative zebrafish model for screening anti-metastasis drugs (Nakayama et al., 2021).
The zebrafish was introduced to the research field in 1972, and has become a powerful model system for cancer research, due to its relative transparency, high reproduction rates, and genetic similarity to humans (Brown et al., 2017; Chen et al., 2021; Fazio et al., 2020; Gamble et al., 2021; Hason and Bartůněk, 2019). Early in development, cells in the zebrafish embryo undergo a morphological change and migrate inwards via a process called epiboly (Bruce and Heisenberg, 2020). The way these healthy cells move is similar to how cancer cells travel across tissues during metastasis. Hence, Nakayama et al. proposed that small-molecule inhibitors that interrupt epiboly may also suppress metastasis.
The team (who are based at the National University of Singapore, the National Cancer Center in Japan and other institutes in Singapore and Japan) found that some of the genes expressed during zebrafish epiboly are also activated during tumor metastasis. This finding provides the experimental support that zebrafish epiboly can serve as a model for tumor cell movement. So, Nakayama et al. developed a zebrafish screening platform, which they used to test 1,280 drugs that had already been approved by a government agency, such as the US Food and Drug Administration (FDA) or the European Medicines Agency (EMA).
The screen was carried out on zebrafish embryos exposed to a specific drug at four hours post-fertilization (Figure 1A). Nakayama et al. found that 132 of the drugs tested induced a delay in epiboly after five hours of treatment. Several of these drugs had previously been reported to inhibit molecular mechanisms associated with metastasis (Liu et al., 2013; Nakayama et al., 2020).
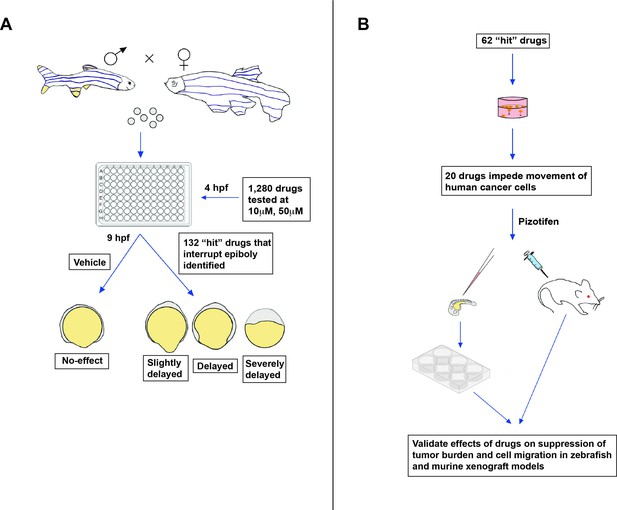
New approach for screening anti-metastasis drugs.
(A) Zebrafish were bred and their embryos were collected and plated into individual wells. Each embryo was treated four hours post-fertilization (hpf) with either the vehicle (an inactive substance that the drug is mixed with to facilitate administration) or a drug that had been approved by the FDA, EMA, or another government agency for cancer treatment: two concentrations were administered (10 µm and 50 µm). Out of the 1,280 drugs tested, 132 interrupted or delayed epiboly five hours after the drug was administered. (B) 62 of these positive hits were then tested on tumor cells cultured in the laboratory. This revealed that 20 of these drugs also impeded the migration of cancer cells, in addition to disrupting epiboly in zebrafish. One of the identified drugs, called Pizotifen, was then administered to zebrafish and mice models that had been injected with fluorescently labelled human cancer cells (commonly referred to as xenografts). This showed that the drug can also suppress metastasis in vivo.
Image credit: Emily Hill; the 6-well plate is adapted from an image by DataBase Center for Life Science (DBCLS; CC BY 4.0).
Nakayama et al. then used cell-based assays to test whether 62 of these 132 ‘positive hits’ (which also delay epiboly in vitro) can suppress the migration of cancer cells (Figure 1B). The tumor cells were placed in a chamber with or without the drug, and the team recorded how many could crawl into the neighboring compartment after a few hours of treatment. This revealed that 20 of the drugs that disrupted epiboly also impeded the movement of human cancer cells.
Finally, Nakayama et al. tested if one of the epiboly-interrupting drugs called Pizotifen could also impair tumor cell movement in living animals: this drug was selected because its primary target (serotonin receptor 2C) is highly expressed in human cancer cells during metastasis. To do this, they injected fluorescently labelled cancer cells into zebrafish embryos, and found that fish exposed to Pizotifen experienced significantly less metastasis than fish treated with a placebo. Similar observations were made in mice that had cancer cells injected into their breast-like tissue, half of which were treated with a daily dose of Pizotifen, and half of which received a placebo (Figure 1B).
The screening platform created by Nakayama et al. makes it easy to rapidly find new drugs that suppress metastasis, while circumventing the limitations of cell culture and mouse model systems. In addition, zebrafish injected with human cancer cells can serve as an additional means for narrowing down which drugs to test in mouse models. Having zebrafish join the drug discovery platform will hopefully result in more and better treatments for patients with metastatic cancers.
References
-
Zebrafish xenograft models of cancer and metastasis for drug discoveryExpert Opinion on Drug Discovery 12:379–389.https://doi.org/10.1080/17460441.2017.1297416
-
Mechanisms of zebrafish epiboly: a current viewCurrent Topics in Developmental Biology 136:319–341.https://doi.org/10.1016/bs.ctdb.2019.07.001
-
A perspective on cancer cell metastasisScience 331:1559–1564.https://doi.org/10.1126/science.1203543
-
Benefits of zebrafish xenograft models in cancer researchFrontiers in Cell and Developmental Biology 9:616551.https://doi.org/10.3389/fcell.2021.616551
-
Zebrafish patient avatars in cancer biology and precision cancer therapyNature Reviews Cancer 20:263–273.https://doi.org/10.1038/s41568-020-0252-3
-
In vitro tumor models: advantages, disadvantages, variables, and selecting the right platformFrontiers in Bioengineering and Biotechnology 4:12.https://doi.org/10.3389/fbioe.2016.00012
-
BookChapter 3 - Challenges and Limitations of Mouse Xenograft Models of CancerIn: Simons BW, editors. Patient Derived Tumor Xenograft Models: Promise, Potential and Practice. Academic Press. pp. 25–36.https://doi.org/10.1016/B978-0-12-804010-2.00003-5
-
Systems biology of cancer metastasisCell Systems 9:109–127.https://doi.org/10.1016/j.cels.2019.07.003
Article and author information
Author details
Acknowledgements
HF acknowledges grants from the National Institutes of Health (NIH; CA215059), the American Cancer Society (RSG-17-204-01-TBG), the National Science Foundation (1911253), and Boston University; CK thanks the diversity grant support from the National Institutes of Health (NIH; CA215059S); and EH is grateful for the Undergraduate Research of Program Award from Boston University.
Publication history
Copyright
© 2022, Kemet et al.
This article is distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use and redistribution provided that the original author and source are credited.
Metrics
-
- 1,001
- views
-
- 97
- downloads
-
- 0
- citations
Views, downloads and citations are aggregated across all versions of this paper published by eLife.
Download links
Downloads (link to download the article as PDF)
Open citations (links to open the citations from this article in various online reference manager services)
Cite this article (links to download the citations from this article in formats compatible with various reference manager tools)
Further reading
-
- Developmental Biology
The evolutionarily conserved Hippo (Hpo) pathway has been shown to impact early development and tumorigenesis by governing cell proliferation and apoptosis. However, its post-developmental roles are relatively unexplored. Here, we demonstrate its roles in post-mitotic cells by showing that defective Hpo signaling accelerates age-associated structural and functional decline of neurons in Caenorhabditis elegans. Loss of wts-1/LATS, the core kinase of the Hpo pathway, resulted in premature deformation of touch neurons and impaired touch responses in a yap-1/YAP-dependent manner, the downstream transcriptional co-activator of LATS. Decreased movement as well as microtubule destabilization by treatment with colchicine or disruption of microtubule-stabilizing genes alleviated the neuronal deformation of wts-1 mutants. Colchicine exerted neuroprotective effects even during normal aging. In addition, the deficiency of a microtubule-severing enzyme spas-1 also led to precocious structural deformation. These results consistently suggest that hyper-stabilized microtubules in both wts-1-deficient neurons and normally aged neurons are detrimental to the maintenance of neuronal structural integrity. In summary, Hpo pathway governs the structural and functional maintenance of differentiated neurons by modulating microtubule stability, raising the possibility that the microtubule stability of fully developed neurons could be a promising target to delay neuronal aging. Our study provides potential therapeutic approaches to combat age- or disease-related neurodegeneration.
-
- Developmental Biology
Wing dimorphism is a common phenomenon that plays key roles in the environmental adaptation of aphid; however, the signal transduction in response to environmental cues and the regulation mechanism related to this event remain unknown. Adenosine (A) to inosine (I) RNA editing is a post-transcriptional modification that extends transcriptome variety without altering the genome, playing essential roles in numerous biological and physiological processes. Here, we present a chromosome-level genome assembly of the rose-grain aphid Metopolophium dirhodum by using PacBio long HiFi reads and Hi-C technology. The final genome assembly for M. dirhodum is 447.8 Mb, with 98.50% of the assembled sequences anchored to nine chromosomes. The contig and scaffold N50 values are 7.82 and 37.54 Mb, respectively. A total of 18,003 protein-coding genes were predicted, of which 92.05% were functionally annotated. In addition, 11,678 A-to-I RNA-editing sites were systematically identified based on this assembled M. dirhodum genome, and two synonymous A-to-I RNA-editing sites on CYP18A1 were closely associated with transgenerational wing dimorphism induced by crowding. One of these A-to-I RNA-editing sites may prevent the binding of miR-3036-5p to CYP18A1, thus elevating CYP18A1 expression, decreasing 20E titer, and finally regulating the wing dimorphism of offspring. Meanwhile, crowding can also inhibit miR-3036-5p expression and further increase CYP18A1 abundance, resulting in winged offspring. These findings support that A-to-I RNA editing is a dynamic mechanism in the regulation of transgenerational wing dimorphism in aphids and would advance our understanding of the roles of RNA editing in environmental adaptability and phenotypic plasticity.






