Temporal and thermal profiling of the Toxoplasma proteome implicates parasite Protein Phosphatase 1 in the regulation of Ca2+-responsive pathways
Figures
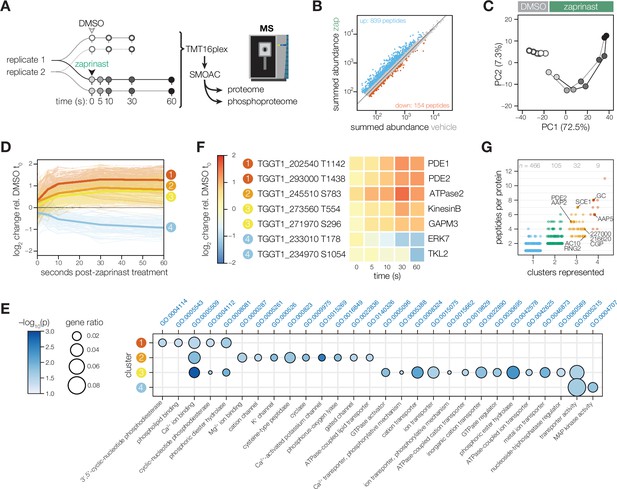
Phosphoregulation triggered by Ca2+ release.
(A) Schematic of the sub-minute phosphoproteomics experiments with the Ca2+ signaling agonist zaprinast. (B) The summed abundances of unique phosphopeptides during zaprinast or vehicle (DMSO) treatment. The abundance ratios were transformed into a modified Z score and were used to threshold increasing (Z>3; blue) or decreasing (Z<–1.8; orange) phosphopeptides. (C) Principal component analysis of phosphopeptides identified as significantly changing. Symbols follow the schematic in A. (D) Gaussian mixture-model-based clustering of phosphopeptides changing during zaprinast treatment. Solid lines show the median relative abundance of each cluster. Opaque lines show the individual phosphopeptides belonging to each cluster. (E) GO terms enriched among phosphopeptides changing with zaprinast treatment, grouped by cluster. Gene ratio is the proportion of proteins with the indicated GO term divided by the total number of proteins belonging to each cluster. Significance was determined with a hypergeometric test; only GO terms with p<0.05 are shown. Redundant GO terms were removed. (F) Examples of phosphopeptides belonging to each cluster. (G) The number of clusters each phosphoprotein belongs to plotted against the number of changing phosphopeptides belonging to each protein. Gene names or IDs indicate proteins discussed in the text.
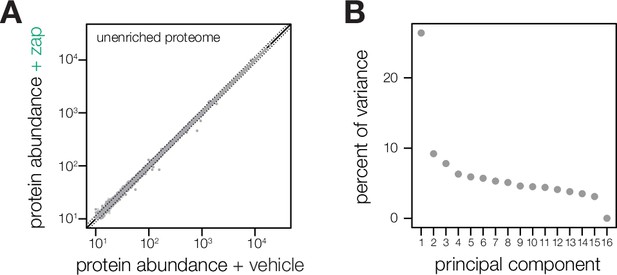
Metrics describing the zaprinast-dependent phosphoproteome.
(A) Aggregate protein abundances for all time points from the non-phosphopeptide enriched samples of parasites treated with zaprinast or the corresponding vehicle (DMSO). Proteins quantified by a single peptide or more are shown in light and dark gray, respectively. Dotted lines correspond to two median absolute deviations. (B) Proportion of the variance explained by each principal component, as described in Figure 1C.
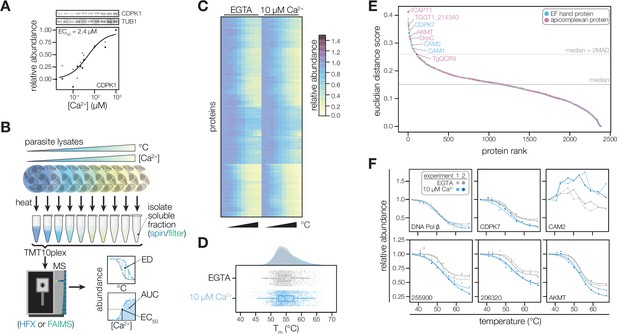
Thermal profiling identifies proteins that change stability in response to Ca2+.
(A) Thermal shift assays can detect Ca2+-dependent stability of CDPK1 in extracts. Parasite lysates were combined with 10 concentrations of Ca2+ spanning the nanomolar to micromolar range. After denaturation at 58 °C, the soluble fraction was separated by SDS-PAGE and probed for CDPK1. Band intensity was normalized to the no-Ca2+ control and scaled. Points in shades of gray represent two different replicates. A dose-response curve was calculated for the mean abundances. (B) Schematic of the thermal profiling workflow. In the temperature-range experiment, parasite lysates were combined with EGTA or 10 µM [Ca2+]free and heated at 10 temperatures spanning 37–67°C. In the concentration-range experiment, parasite lysates were combined with 10 different [Ca2+]free (nM–mM range) and heated at 50, 54, or 58 °C. Temperature-range shifts were quantified by the Euclidean distance (ED) score, a weighted ratio of thermal stability differences between treatments and replicates. Concentration-range shifts were summarized by pEC50, area under the curve (AUC), and goodness of fit (R2). (C) Heat map of protein thermal stability relative to the lowest temperature (37 °C) in 0 or 10 µM Ca2+. The mean relative abundance at each temperature was calculated for 2381 proteins. Proteins are plotted in the same order in both treatments. (D) Raincloud plots summarizing the distribution of Tm in lysates with EGTA (gray) or 10 µM [Ca2+]free (blue). The average melting temperatures of proteins identified in two replicates were plotted. (E) Proteins rank-ordered by euclidean distance score quantifying the Ca2+-dependent shift in thermal stability. Solid and dotted lines represent the median ED score and two modified Z scores above the median, respectively. Highlighted proteins have EF hand domains (blue) or are conserved in apicomplexans (pink). (F) Thermal profiles of individual proteins: DNA polymerase β (TGGT1_233820); the EF hand domain-containing proteins CDPK7 (TGGT1_228750) and the calmodulin-like protein CAM2 (TGGT1_262010); potential Ca2+-leak channels TGGT1_255900 and TGGT1_206320; and AKMT (TGGT1_216080).
-
Figure 2—source data 1
This file contains the source data that was quantified to make the graph presented in Figure 2.
TUB1, LICOR 700 channel.
- https://cdn.elifesciences.org/articles/80336/elife-80336-fig2-data1-v3.zip
-
Figure 2—source data 2
This file contains the source data that was quantified to make the graph presented in Figure 2.
CDPK1, LICOR 800 channel.
- https://cdn.elifesciences.org/articles/80336/elife-80336-fig2-data2-v3.zip
-
Figure 2—source data 3
This file contains the annotated source data that was quantified to make the graph presented in Figure 2.
TUB1, LICOR 700 channel and CDPK1, LICOR 800 channel.
- https://cdn.elifesciences.org/articles/80336/elife-80336-fig2-data3-v3.zip
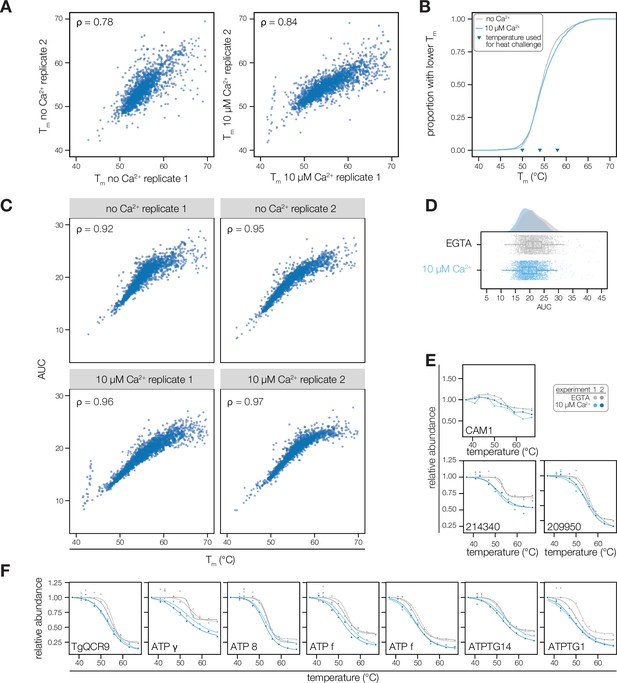
Extended data for thermal profiling experiments.
(A) Comparison of the melting points of proteins with standard melting behavior (R2 >0.8) across replicate experiments and treatment conditions. The correlation of melting temperatures is given by Spearman’s rho. (B) Cumulative distribution function of average measured melting temperatures in lysates with 10 µM or no Ca2+. (C) Correlation between Tm of proteins with standard melting behavior (R2 >0.8) and AUC in each experiment. Correlation is given by Spearman’s rho. (D) Distribution of AUC (as in Figure 1D). (E) Melting curve of proteins discussed in the text: CAM1 (TGGT1_246930), an ICAP (TGGT1_214340, Sidik et al., 2016a), and a putative thioredoxin (TGGT1_209950). (F) Melting curves of TgQCR9 and ATP synthase subunits (ATP) destabilized by Ca2+.
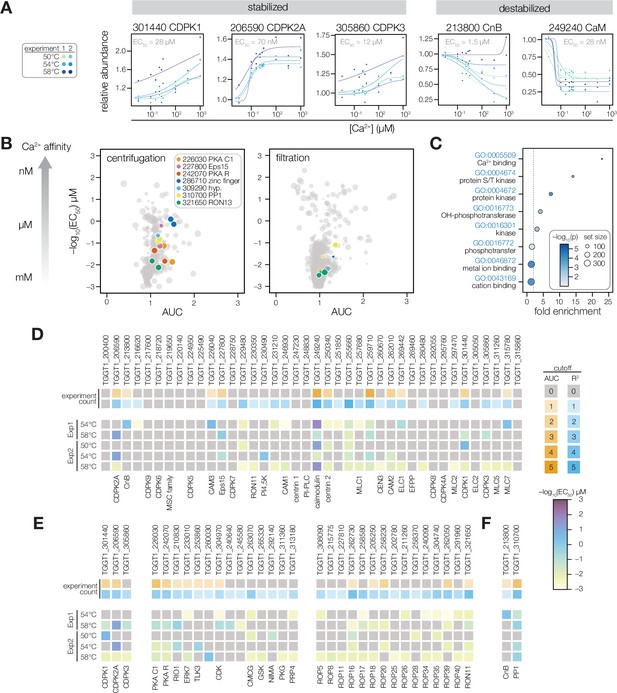
Thermal profiling identifies anticipated and unexplored Ca2+-responsive proteins.
(A) Mass spectrometry-derived thermal profiles of EF hand-containing proteins stabilized or destabilized by Ca2+. Relative abundance is calculated relative to the protein abundance at 0 µM Ca2+. EC50 is the median of the EC50 values of the curves displayed on the plots. (B) The magnitude of Ca2+-dependent stabilization (AUC) plotted against the sensitivity (pEC50) for protein abundances exhibiting a dose-response trend with an R2 >0.8. Point size is scaled to R2. Summary parameters for the different separation methods (ultracentrifugation or filtration) are plotted separately. Colors identify candidates with Ca2+-responsive behavior validated in Figure 4. (C) Gene ontology (GO) terms enriched among candidate Ca2+-responsive proteins (AUC greater than two modified Z scores and R2 dose-response >0.8). Fold enrichment is the frequency of Ca2+-responsive proteins in the set relative to the frequency of the GO term in the population of detected proteins. Significance was determined with a hypergeometric test; only GO terms with p<0.05 are shown. (D–F) EF hand domain proteins (D), protein kinases (E), and protein phosphatases (F) detected in the thermal profiling mass spectrometry datasets. The top rows indicate if a protein passed the AUC cutoff (orange) or R2 cutoff (blue) for dose-response behavior. The opacity of the band represents the number of experiments in which the protein exhibited the behavior (out of five). The five rows below summarize the pEC50 of each experiment in which the protein exhibited a dose-response trend with R2 >0.8. Kinases are loosely grouped as CDPK’s (included as a reference), non-rhoptry kinases, and secretory pathway kinases.
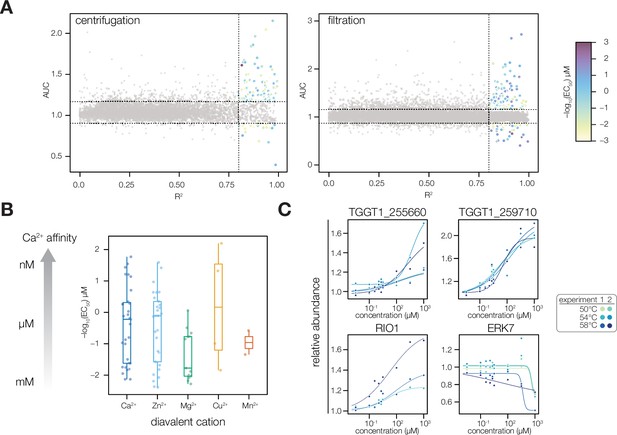
Extended analysis of thermal profiling experiments.
(A) Plots of protein curve fit R2 vs. AUC, a measure of stability change, for each set of MS experiments. Dotted lines indicate thresholds for designated Ca2+-responsive behavior: R2 >0.8 and an AUC two modified Z scores from the median. Each point corresponds to an average of two replicates at each thermal challenge temperature (50, 54, or 58 °C). Color denotes pEC50 in µM. (B) A comparison of the pEC50 values of proteins predicted to bind different divalent metal cations. Specificity was predicted via the presence of Interpro domains and through manual annotation. (C) Plots of individual protein melting curves, as described in the text: the EF hand domain-containing proteins TGGT1_255660 and TGGT1_259710; and the kinases RIO1 (TGGT1_210830) and ERK7 (TGGT1_233010).
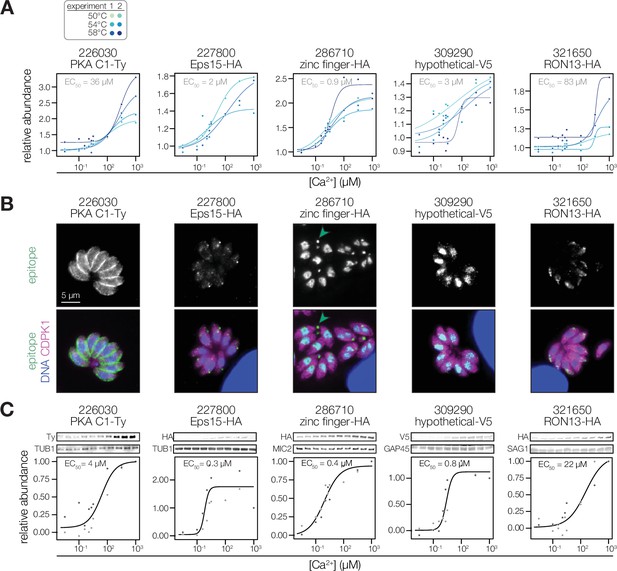
Validation of Ca2+-dependent thermal stability.
(A) Mass spectrometry-derived thermal profiles of the candidates, as in Figure 3A. (B) Immunofluorescence images of fixed intracellular parasites expressing the indicated proteins with C-terminal epitopes at endogenous loci. Hoechst and anti-CDPK1 were used as counterstains in the merged image. Green arrowheads highlight an example of TGGT1_286710 residual body staining. In the case of PKA C1/R, the stain of the R subunit is shown, as both subunits colocalize. (C) Immunoblot-derived thermal profiles of the candidates. Colors correspond to two independent replicates. Uncropped blots are shown in the Figure 4—figure supplement 1.
-
Figure 4—source data 1
This file contains the source data that was quantified to make the graph presented in Figure 4.
TUB1, LICOR 700 channel.
- https://cdn.elifesciences.org/articles/80336/elife-80336-fig4-data1-v3.zip
-
Figure 4—source data 2
This file contains the source data that was quantified to make the graph presented in Figure 4.
PKA C1-Ty, LICOR 800 channel.
- https://cdn.elifesciences.org/articles/80336/elife-80336-fig4-data2-v3.zip
-
Figure 4—source data 3
This file contains the source data that was quantified to make the graph presented in Figure 4.
TUB1, LICOR 700 channel.
- https://cdn.elifesciences.org/articles/80336/elife-80336-fig4-data3-v3.zip
-
Figure 4—source data 4
This file contains the source data that was quantified to make the graph presented in Figure 4.
Eps15-HA LICOR 800 channel.
- https://cdn.elifesciences.org/articles/80336/elife-80336-fig4-data4-v3.zip
-
Figure 4—source data 5
This file contains the source data that was quantified to make the graph presented in Figure 4.
286710 HA, LICOR 700 channel.
- https://cdn.elifesciences.org/articles/80336/elife-80336-fig4-data5-v3.zip
-
Figure 4—source data 6
This file contains the source data that was quantified to make the graph presented in Figure 4.
MIC2, LICOR 800 channel.
- https://cdn.elifesciences.org/articles/80336/elife-80336-fig4-data6-v3.zip
-
Figure 4—source data 7
This file contains the source data that was quantified to make the graph presented in Figure 4.
GAP45, LICOR 700 channel.
- https://cdn.elifesciences.org/articles/80336/elife-80336-fig4-data7-v3.zip
-
Figure 4—source data 8
This file contains the source data that was quantified to make the graph presented in Figure 4.
309290-V5, LICOR 800 channel.
- https://cdn.elifesciences.org/articles/80336/elife-80336-fig4-data8-v3.zip
-
Figure 4—source data 9
This file contains the source data that was quantified to make the graph presented in Figure 4.
SAG1, LICOR 700 channel.
- https://cdn.elifesciences.org/articles/80336/elife-80336-fig4-data9-v3.zip
-
Figure 4—source data 10
This file contains the source data that was quantified to make the graph presented in Figure 4.
RON13-HA, LICOR 800 channel.
- https://cdn.elifesciences.org/articles/80336/elife-80336-fig4-data10-v3.zip
-
Figure 4—source data 11
This file contains the annotated source data that was quantified to make the graph presented in Figure 4.
- https://cdn.elifesciences.org/articles/80336/elife-80336-fig4-data11-v3.zip
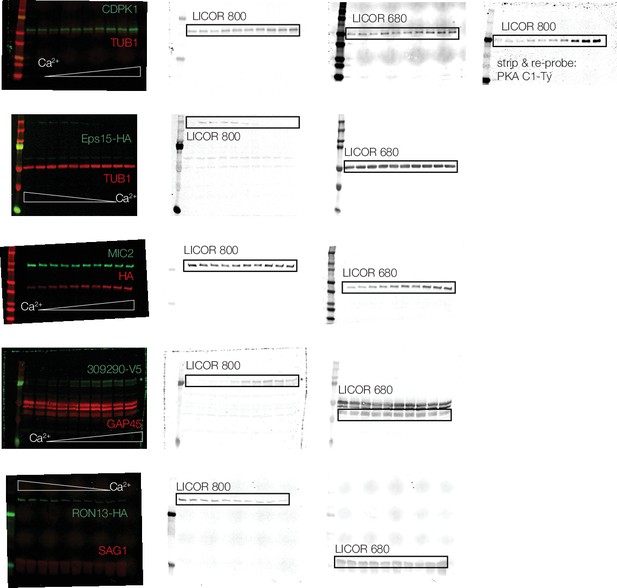
Uncropped immunoblots, as in Figure 4C.
Parasite lysates were incubated at 10 Ca2+ concentrations and were thermally challenged at 58 °C. Following centrifugation, the supernatant containing the soluble protein fraction was separated by SDS-PAGE. Following transfer onto a nitrocellulose membrane, the blots were probed with the indicated primary antibodies. The direction of the Ca2+ gradient is indicated on each blot. Representative blots from two biological replicates are shown.
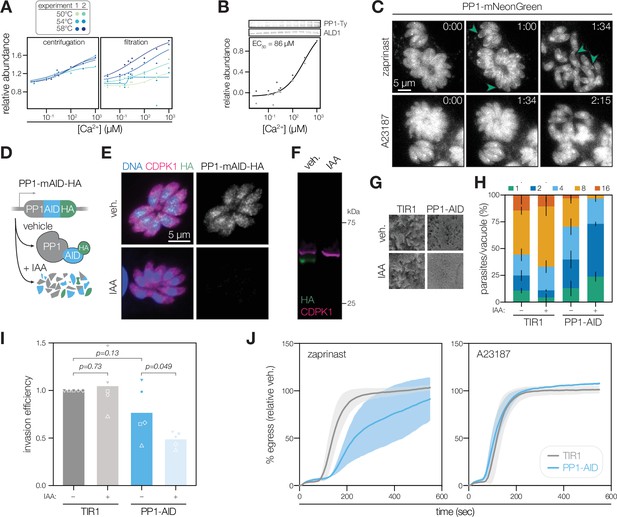
PP1 is a Ca2+-responsive enzyme involved in T. gondii egress and invasion.
(A) The Ca2+-dependent stabilization of PP1 (TGGT1_310700) in each mass spectrometry experiment. (B) Immunoblotting for endogenously tagged PP1-mNG-Ty at different Ca2+ concentrations and thermal challenge at 58 °C. Abundance is calculated relative to the band intensity at 0 µM Ca2+ and scaled. Points in shades of gray represent different replicates. A dose-response curve was calculated for the mean abundances. (C) Parasites expressing endogenously tagged PP1-mNG egress after treatment with 500 µM zaprinast or 4 µM A23187. Arrows show examples of PP1 enrichment at the apical end. Time after treatment is indicated in m:ss. (D) Rapid regulation of PP1 by endogenous tagging with mAID-HA. IAA, Indole-3-acetic acid (IAA). (E) PP1-mAID-HA visualized in fixed intracellular parasites by immunofluorescence after 3 hr of 500 µM IAA or vehicle treatment. Hoechst and anti-CDPK1 are used as counterstains (Waldman et al., 2020). (F) PP1-mAID-HA depletion, as described in (E), monitored by immunoblotting. The expected molecular weights of PP1-mAID-HA and CDPK1 are 48 and 65 kDa, respectively. (G) Plaque assays of 1,000 TIR1 and PP1-mAID-HA parasites infected onto a host cell monolayer and allowed to undergo repeated cycles of invasion, replication, and lysis for 7 days in media with or without IAA. (H) The number of parasites per vacuole measured for PP1-mAID-HA and the TIR1 parental strain after 24 hr of 500 µM IAA treatment. Mean counts (n=3) are expressed as a percentage of all vacuoles counted. (I) Invasion assays PP1-mAID-HA or TIR1 parental strains treated with IAA or vehicle for 3 hr. Parasites were incubated on host cells for 60 min prior to differential staining of intracellular and extracellular parasites. Parasite numbers were normalized to host cell nuclei for each field. Means graphed for n=5 biological replicates (different shapes), Welch’s t-test. (J) Parasite egress stimulated with 500 µM zaprinast or 8 µM A23187 following 3 h of treatment with vehicle or IAA. Egress was monitored by the number of host cell nuclei stained with DAPI over time and was normalized to egress in the vehicle-treated strain. Mean ±S.D. graphed for n=3 biological replicates.
-
Figure 5—source data 1
This file contains the source data that was quantified to make the graph presented in Figure 4.
ALD1, LICOR 700 channel.
- https://cdn.elifesciences.org/articles/80336/elife-80336-fig5-data1-v3.zip
-
Figure 5—source data 2
This file contains the source data that was quantified to make the graph presented in Figure 4.
PP1-Ty, LICOR 800 channel.
- https://cdn.elifesciences.org/articles/80336/elife-80336-fig5-data2-v3.zip
-
Figure 5—source data 3
This file contains the annotated source data that was quantified to make the graph presented in Figure 5.
- https://cdn.elifesciences.org/articles/80336/elife-80336-fig5-data3-v3.zip
-
Figure 5—source data 4
This file contains the source data that was presented in Figure 5.
CDPK1, LICOR 700 channel (magenta) and PP1-HA, LICOR 800 channel (green).
- https://cdn.elifesciences.org/articles/80336/elife-80336-fig5-data4-v3.zip
-
Figure 5—source data 5
This file contains the annotate source data that was presented in Figure 5.
CDPK1, LICOR 700 channel (magenta) and PP1-HA, LICOR 800 channel (green).
- https://cdn.elifesciences.org/articles/80336/elife-80336-fig5-data5-v3.zip
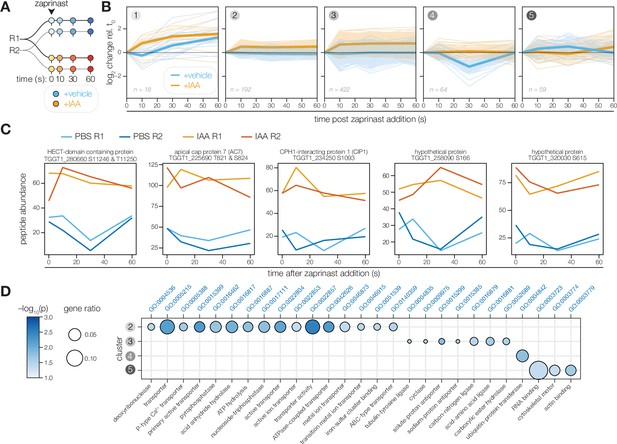
The PP1-dependent phosphoproteome.
(A) Schematic of the phosphoproteomics time course. PP1-AID parasites were treated with IAA or vehicle for 3 hr. Extracellular parasites were then treated with zaprinast, and samples were collected during the first minute after stimulation. The experiment was performed in biological replicate (R1 and R2). (B) Five clusters were identified with respect to phosphopeptide dynamics and PP1-dependence. (C) Examples of phosphopeptides dynamically regulated by zaprinast and exhibiting PP1-dependent dephosphorylation. (D) GO terms enriched among phosphopeptides, grouped by cluster. Gene ratio is the proportion of proteins with the indicated GO term divided by the total number of proteins belonging to each cluster. Significance was determined with a hypergeometric test; only GO terms with p<0.05 and represented by more than one protein are shown. Redundant GO terms were removed. Cluster 1 lacked enough peptides for enrichment analysis.
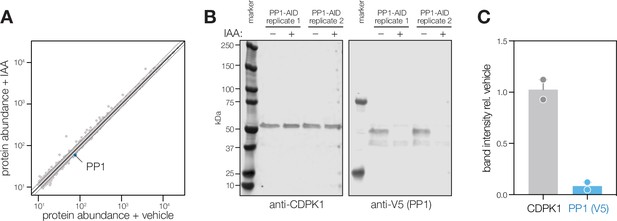
Extended analysis of PP1 phosphoproteomics experiment.
(A) Aggregate protein abundances from the non-phosphopeptide enriched samples of parasites treated with IAA or vehicle. Proteins quantified by a single peptide or more are shown in light and dark gray, respectively. Lines correspond to two median absolute deviations. (B) Immunoblot of samples used for the PP1 phosphoproteomics experiment. (C) Quantification of immunoblot band intensity. Intensity was normalized relative to the signal of the vehicle-treated lane for each replicate. Mean ± SD plotted for two independent replicates.
-
Figure 6—figure supplement 1—source data 1
This file contains the source data that was presented in Figure 6—figure supplement 1.
CDPK1, LICOR 700 channel.
- https://cdn.elifesciences.org/articles/80336/elife-80336-fig6-figsupp1-data1-v3.zip
-
Figure 6—figure supplement 1—source data 2
This file contains the source data that was presented in Figure 6—figure supplement 1.
PP1-HA, LICOR 800 channel.
- https://cdn.elifesciences.org/articles/80336/elife-80336-fig6-figsupp1-data2-v3.zip
-
Figure 6—figure supplement 1—source data 3
This file contains the annotated source data that was presented in Figure 6—figure supplement 1.
- https://cdn.elifesciences.org/articles/80336/elife-80336-fig6-figsupp1-data3-v3.zip
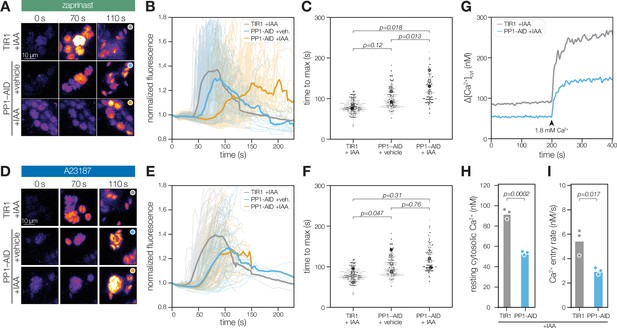
PP1 ensures rapid Ca2+ mobilization prior to zaprinast-induced egress.
(A) Selected frames of time-lapse images of PP1-AID parasites expressing the genetically encoded Ca2+ indicator GCaMP following treatment with 500 µM zaprinast. (B) Normalized GCaMP fluorescence of individual vacuoles was tracked after zaprinast treatment and prior to egress (opaque lines). The solid line represents the mean normalized fluorescence of all vacuoles across n=3 biological replicates. (C) The time to maximum normalized fluorescence of individual vacuoles after zaprinast treatment. Different replicates are shown in different shades of gray. Small points correspond to individual vacuoles; large points are the mean for each replicate. p Values were calculated from a two-tailed t-test. (D) Selected frames of time-lapse images of PP1-AID parasites expressing the genetically encoded Ca2+ indicator GCaMP following treatment with 4 µM A23187. (E) Normalized GCaMP fluorescence of individual vacuoles was tracked after A23187 treatment and prior to egress (opaque lines). The solid line represents the mean normalized fluorescence of all vacuoles across n=3 biological replicates. (F) The time to maximum normalized fluorescence of individual vacuoles after A23187 treatment. Small points correspond to individual vacuoles; large points are the mean for each replicate. p Values were calculated from a two-tailed t-test. (G) Fluorescence intensity of Fura2/AM-loaded TIR1 or PP1-AID parasites treated with IAA for 5 hr before and after addition of the 1.8 mM Ca2+. Representative traces from three biological replicates. (H) Resting cytoplasmic [Ca2+] prior to incubation in buffers with elevated [Ca2+]. p Values were calculated from an ANOVA. (I) The rate of Ca2+ entry in the first 20 s after addition of 1.8 mM Ca2+ to parasites p values were calculated from an ANOVA. Entry rates following addition of other concentrations are shown in Figure 7—figure supplement 1.
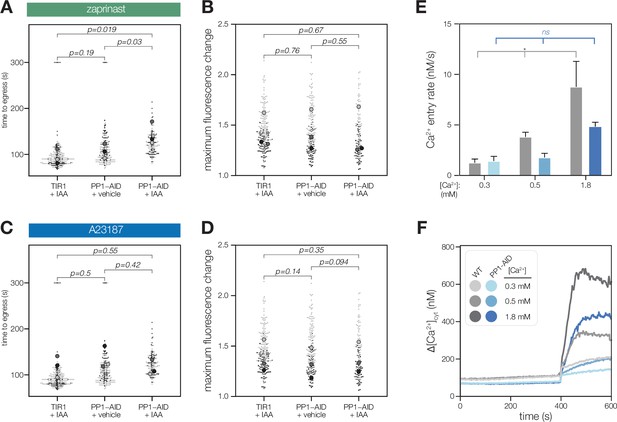
PP1-AID parasites egress, as quantified by video microscopy.
(A) The time to vacuole egress after zaprinast treatment was manually scored. Different replicates are shown in different shades of gray. Small points represent individual vacuoles; large points are the mean for each replicate. p Values were calculated from a two-tailed t-test. (B) The normalized fluorescence change of individual vacuoles after zaprinast treatment. Different replicates are shown in different shades of gray. Small points correspond to individual vacuoles; large points are the mean for each replicate. p Values were calculated from a two-tailed t-test. (C) The time to vacuole egress after A23187 treatment was manually scored. Different replicates are shown in different shades of gray. Small points correspond to individual vacuoles; large points are the mean for each replicate. p Values were calculated from a two-tailed t-test. (D) The normalized fluorescence change of individual vacuoles after A23187 treatment. Different replicates are shown in different shades of gray. Small points represent individual vacuoles; large points are the mean for each replicate. p Values were calculated from a two-tailed t-test. (E) Ca2+ entry rates corresponding to (F). The slope of the trace at the time of addition of Ca2+ was measured as the change in the concentration of Ca2+ during the initial 20 s after addition of Ca2+. Each bar represents the average of a minimum of three biological replicates. ANOVA was used for the statistical analyses. * p<0.01. (F) Fluorescence intensity of Fura2/AM-loaded TIR1 or PP1-AID parasites treated with IAA for 5 hours upon incubation with buffers of the indicated [Ca2+]. Representative traces from at least three biological replicates.
Videos
Representative image series of parasites expressing endogenously tagged PP1-mNG following treatment with 500 µM zaprinast.
Representative image series of parasites expressing endogenously tagged PP1-mNG following treatment with 4 µM A23187.
Representative image series of PP1-AID parasites expressing the genetically encoded Ca2+ indicator GCaMP following treatment with 500 µM zaprinast.
Representative image series of PP1-AID parasites expressing the genetically encoded Ca2+ indicator GCaMP following treatment with 4 µM A23187.
Tables
Gene IDs of proteins discussed in the text.
NS, not significant (using thresholds defined in the text). ND, not detected. TR, temperature range. CR, concentration range.
| Gene ID | Description in text | Reference | Phospho cluster | Thermal profiling |
|---|---|---|---|---|
| TGGT1_202540 | PDE1 | Jia et al., 2017 | 1, 3 | NS |
| TGGT1_293000 | PDE2 | Jia et al., 2017; Moss et al., 2022; Vo et al., 2020 | 1, 2, 4 | NS |
| TGGT1_245730 | PI4,5K | Garcia et al., 2017 | 1, 2 | NS |
| TGGT1_276170 | PI3,4K | Garcia et al., 2017 | 1, 2 | NS |
| TGGT1_248830 | PI-PLC | Bullen et al., 2016; Fang et al., 2006 | 1, 3 | NS |
| TGGT1_288800 | Phosphatidylinositol-3,4,5-triphosphate 5-phosphatase | 1 | NS | |
| TGGT1_254390 | Putative Sec14 | 1 | NS | |
| TGGT1_206590 | CDPK2A | Billker et al., 2009 | 1, 2, 3 | CR |
| TGGT1_228750 | CDPK7 | Bansal et al., 2021 | 1, 3 | TR |
| TGGT1_267100 | PPM2B | Yang et al., 2019; Yang and Arrizabalaga, 2017 | 1 | NS |
| TGGT1_238995 | Ca2+-activated K+ channel | 2 | ND | |
| TGGT1_273380 | Ca2+-activated K+ channel | 2, 3 | ND | |
| TGGT1_259200B | Na+/H+ exchanger | Arrizabalaga et al., 2004 | 2, 3 | NS |
| TGGT1_305180 | Na+/H+ exchanger | Francia et al., 2011 | 2, 3, 4 | NS |
| TGGT1_245510 | ATPase2 | Chen et al., 2021 | 2 | ND |
| TGGT1_254370 | Guanylyl cyclase | Bisio et al., 2019; Brown and Sibley, 2018 | 1, 2, 3, 4 | NS |
| TGGT1_312100 | Calcium ATPase TgA1 | Luo et al., 2005; Luo et al., 2001 | 3 | ND |
| TGGT1_201150 | Copper transporter CuTP | Kenthirapalan et al., 2014 | 3 | NS |
| TGGT1_318460 | Putative P5B-ATPase | Møller et al., 2008 | 2 | NS |
| TGGT1_289070 | Putative E1-E2 ATPase | 3 | NS | |
| TGGT1_278660 | TgATP4 | Lehane et al., 2019 | 3, 4 | NS |
| TGGT1_226020 | MFS transporter | 3 | NS | |
| TGGT1_230570 | MFS transporter | 3 | NS | |
| TGGT1_253700 | MFS transporter | 3, 4 | NS | |
| TGGT1_257530 | Tyrosine transporter ApiAT5-3 | Parker et al., 2019; Wallbank et al., 2019 | 3, 4 | NS |
| TGGT1_292110 | Formate transporter TgFNT2 | Erler et al., 2018; Zeng et al., 2021 | 3 | NS |
| TGGT1_270865 | Adenylyl cyclase | Brown and Sibley, 2018; Jia et al., 2017 | 2, 3 | NS |
| TGGT1_238390 | Unique guanylyl cyclase organizer UGO | Bisio et al., 2019 | 2, 3 | NS |
| TGGT1_309190 | ARO-interacting protein (adenylyl cyclase organizer) AIP | Mueller et al., 2016; Mueller et al., 2013 | 1, 3 | NS |
| TGGT1_273560 | Divergent kinesin KinesinB | Leung et al., 2017 | 2, 3 | NS |
| TGGT1_201230 | Divergent kinesin | Wickstead et al., 2010 | 3 | ND |
| TGGT1_247600 | Dynein light chain | 3 | NS | |
| TGGT1_255190 | MyoC | Frénal et al., 2017 | 3 | NS |
| TGGT1_278870 | MyoF | Heaslip et al., 2016; Jacot et al., 2013 | 1, 2, 3 | NS |
| TGGT1_257470 | MyoJ | Frénal et al., 2014 | 3 | CR |
| TGGT1_213325 | Uncharacterized TBC domain protein | 3 | ND | |
| TGGT1_221710 | Uncharacterized TBC domain protein | 3 | NS | |
| TGGT1_237280 | Uncharacterized TBC domain protein | 1, 3, 4 | NS | |
| TGGT1_274130 | Uncharacterized TBC domain protein | 2, 3, 4 | NS | |
| TGGT1_289820 | Uncharacterized TBC domain protein | 3 | NS | |
| TGGT1_206690 | GAPM2B | Harding et al., 2019 | 3 | NS |
| TGGT1_271970 | GAPM3 | Harding et al., 2019 | 3 | NS |
| TGGT1_233010 | ERK7 | O’Shaughnessy et al., 2020 | 3, 4 | CR |
| TGGT1_234970 | Tyrosine kinase-like protein TgTLK2 | Varberg et al., 2018 | 4 | NS |
| TGGT1_202900 | Putative K+ voltage-gated channel complex subunit | 4 | NS | |
| TGGT1_228200 | Vacuolar (H+)-ATPase G subunit | 1, 4 | NS | |
| TGGT1_233130 | Putative nucleoside transporter | 4 | NS | |
| TGGT1_258700 | MFS family transporter | 4 | ND | |
| TGGT1_269260 | SCE1 | McCoy et al., 2017 | 1, 2, 3 | NS |
| TGGT1_295850 | AAP2 | Engelberg et al., 2020 | 1, 2, 4 | CR |
| TGGT1_319900 | AAP5 | Engelberg et al., 2020 | 1, 2, 3, 4 | NS |
| TGGT1_227000 | Apical polar ring protein | Koreny et al., 2021 | 1, 3, 4 | ND |
| TGGT1_244470 | RNG2 | Katris et al., 2014 | 1, 2, 3 | ND |
| TGGT1_292950 | Apical cap protein AC10 | Back et al., 2020; Tosetti et al., 2020 | 1, 2, 3 | NS |
| TGGT1_240380 | Conoid gliding protein CGP | Li et al., 2022 | 1, 3, 4 | NS |
| TGGT1_216620 | Ca2+ influx channel with EF hands | Chang et al., 2019 | 1, 3, 4 | NS |
| TGGT1_246930 | Calmodulin-like protein CAM1 | Long et al., 2017b | ND | TR |
| TGGT1_262010 | ACalmodulin-like protein CAM2 | Long et al., 2017b | ND | TR, CR |
| TGGT1_216080 | apical lysine methyltransferase (AKMT) | Heaslip et al., 2011 | NS | TR |
| TGGT1_270690 | DrpC | Heredero-Bermejo et al., 2019; Melatti et al., 2019 | NS | TR |
| TGGT1_201880 | TgQCR9 | ND | TR | |
| TGGT1_204400 | ATP synthase subunit alpha | Hayward et al., 2021; Huet et al., 2018; Mühleip et al., 2021; Salunke et al., 2018; Seidi et al., 2018 | NS | TR |
| TGGT1_231910 | ATP synthase subunit gamma | Huet et al., 2018; Mühleip et al., 2021; Salunke et al., 2018; Seidi et al., 2018 | ND | TR |
| TGGT1_208440 | ATP synthase subunit 8/ASAP-15 | Huet et al., 2018; Mühleip et al., 2021; Salunke et al., 2018; Seidi et al., 2018 | ND | TR, CR |
| TGGT1_215610 | ATP synthase subunit f/ICAP11/ASAP-10 | Huet et al., 2018; Mühleip et al., 2021; Salunke et al., 2018; Seidi et al., 2018 | ND | TR |
| TGGT1_263080 | ATP synthase-associated protein ASAP-18/ATPTG14 | Huet et al., 2018; Mühleip et al., 2021; Salunke et al., 2018; Seidi et al., 2018 | ND | TR, CR |
| TGGT1_246540 | ATP synthase-associated protein ATPTG1 | Mühleip et al., 2021 | NS | TR |
| TGGT1_249240 | CaM | Paul et al., 2015 | ND | CR |
| TGGT1_213800 | CnB | Paul et al., 2015 | NS | CR |
| TGGT1_227800 | Eps15 | Birnbaum et al., 2020; Chern et al., 2021 | NS | CR |
| TGGT1_269442 | ELC1 | Nebl et al., 2011 | ND | CR |
| TGGT1_297470 | MLC1 | Gaskins et al., 2004 | ND | CR |
| TGGT1_297470 | MLC5 | Graindorge et al., 2016 | ND | CR |
| TGGT1_315780 | MLC7 | Graindorge et al., 2016 | ND | CR |
| TGGT1_226030 | PKA-C1 | Jia et al., 2017; Uboldi et al., 2018 | NS | CR |
| TGGT1_242070 | PKA-R | Jia et al., 2017; Uboldi et al., 2018 | NS | CR |
| TGGT1_210830 | Putative RIO1 kinase | NS | CR | |
| TGGT1_310700 | PP1 | Paul et al., 2020; Zeeshan et al., 2021 | ND | CR |
| TGGT1_207910 | Calcium-hydrogen exchanger TgCAX | Guttery et al., 2013 | NS | CR |
| TGGT1_311080 | Apicoplast two-pore channel TgTPC | Li et al., 2021 | NS | CR |
| TGGT1_204050 | Subtilisin 1 SUB1 | NS | CR | |
| TGGT1_206490 | Metacaspase 1 with a C2 domain | Li et al., 2015 | ND | CR |
| TGGT1_310810 | Ca2+-activated apyrase | ND | CR | |
| TGGT1_321650 | RON13 | Lentini et al., 2021 | ND | CR |
| TGGT1_286710 | Uncharacterized metal-binding protein with zinc fingers | ND | CR | |
| TGGT1_309290 | Uncharacterized metal-binding protein with HD domain | ND | CR | |
| TGGT1_225690 | Apical cap protein AC7 | Chen et al., 2015 | 2, 3 | NS |
| TGGT1_234250 | CHP interacting protein CIP1 | Long et al., 2017a | NS | ND |
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Strain, strain background (T. gondii) | TIR1 | PMID:28465425 | RH/TIR1/∆KU80/∆HXGPRT | |
| Strain, strain background (T. gondii) | TIR1/GCaMP6f | PMID:35484233 | RH/TIR1/pTUB1-GCaMP6f-3'DHFR/∆KU80/∆HXGPRT | |
| Strain, strain background (T. gondii) | DiCre | PMID:31577230 | RH/∆KU80::DiCre_T2A/∆HXGPRT | |
| Strain, strain background (T. gondii) | TIR1/MIC2-GLuc-P2A-GCaMP6f | This paper | RH/TIR1/∆KU80/∆HXGPRT/pMIC2-MIC2-GLuc-myc-P2A-GCaMP6f | |
| Strain, strain background (T. gondii) | PKA C1-Ty (Figure 4) | This paper | TGGT1_226030 | RH/TIR1/pTUB1-GCaMP6f-3'DHFR/∆KU80/PKA C1-mCherry-V5-mAID-Ty/PKA R-V5-3HA |
| Strain, strain background (T. gondii) | Eps15-HA (Figure 4) | This paper | TGGT1_227800 | RH/TIR1/pTUB1-GCaMP6f-3'DHFR/∆KU80/Eps15-V5-mCherry-mAID-HA |
| Strain, strain background (T. gondii) | zinc finger-HA (Figure 4) | This paper | TGGT1_286710 | RH/TIR1/∆KU80/∆HXGPRT/TGGT1_286470-V5-3HA |
| Strain, strain background (T. gondii) | hypothetical-V5 (Figure 4) | This paper | TGGT1_309290 | RH/TIR1/pTUB1-GCaMP6f-3'DHFR/∆KU80/∆HXGPRT/TGGT1_309290-V5-mNeonGreen-mAID-Ty |
| Strain, strain background (T. gondii) | RON13-HA (Figure 4) | This paper | TGGT1_321650 | RH/∆KU80::DiCre_T2A/∆HXGPRT/RON13-HA-U1 |
| Strain, strain background (T. gondii) | PP1-AID (Figure 5) | This paper | TGGT1_310700 | RH/TIR1/∆KU80/∆HXGPRT/pMIC2-MIC2-GLuc-myc-P2A-GCaMP6f/PP1-V5-mAID-HA |
| Strain, strain background (T. gondii) | PP1-AID/TIR1/MIC2-GLuc-P2A-GCaMP6f (Figure 7) | This paper | TGGT1_310700 | RH/TIR1/∆KU80/∆HXGPRT/pMIC2-MIC2-GLuc-myc-P2A-GCaMP6f/PP1-V5-mAID-HA |
| Cell line (Homo sapiens) | Human Foreskin Fibroblasts (HFFs) | ATCC | SCRC-1041 | |
| Antibody | Guinea pig monoclonal anti-CDPK1 | Covance | Custom antibody | IF (1/10000), WB (1/40000) |
| Antibody | Mouse monoclonal anti-TUB1 (clone 12G10) | Developmental Studies Hybridoma Bank at the University of Iowa | RRID:AB_1157911 | WB (1/5000) |
| Antibody | Mouse monoclonal anti-Ty1 (clone BB2) | PMID:8813669 | IF (1/1000), WB(1/2000) | |
| Antibody | Rabbit polyclonal anti-HA | Invitrogen | Invitrogen:71–5500 | WB (1/1000) |
| Antibody | Mouse monoclonal anti-MIC2 (6D10) | PMID:10799515 | WB (1:2000) | |
| Antibody | Mouse monoclonal anti-V5 | Invitrogen | Invitrogen:R960-25 | IF (1:1000), WB (1:2000) |
| Antibody | Rabbit polyclonal | PMID:18312842 | WB (1:2000) | |
| Antibody | Mouse monoclonal anti-HA (16B12) | Biolegend | Biolegend:901533 | IF (1:1000) |
| Antibody | Mouse polyclonal anti-SAG1 | PMID:3183382 | WB (1/1000) | |
| Antibody | Alexa Fluor 594 polyclonal goat anti-guinea pig | Life Technologies | Life Technologies:A11076 | IF (1/1000) |
| Antibody | Alexa Fluor 488 polyclonal goat anti-mouse | Life Technologies | Life Technologies:A11029 | IF (1/1000) |
| Antibody | IRDye 800CW polyclonal Goat anti-Mouse IgG1-Specific Secondary Antibody | LICOR | LICOR:926–32350 | WB (1/10000) |
| Antibody | IRDye 680LT polyclonal Goat anti-Mouse IgG Secondary Antibody | LICOR | LICOR:926–68020 | WB (1/10000) |
| Antibody | IRDye 800CW polyclonal Donkey anti-Guinea Pig IgG Secondary Antibody | LICOR | LICOR:926–32411 | WB (1/10000) |
| Antibody | IRDye 680RD polyclonal Donkey anti-Guinea Pig IgG Secondary Antibody | LICOR | LICOR:926–68077 | WB (1/10000) |
| Antibody | IRDye 800CW polyclonal Goat anti-Rabbit IgG Secondary Antibody | LICOR | LICOR:926–32211 | WB (1/10000) |
| Antibody | IRDye 680LT polyclonal Goat anti-Rabbit IgG Secondary Antibody | LICOR | LICOR:926–68021 | WB (1/10000) |
| Chemical compound, drug | Hoechst | Santa Cruz | Santa Cruz:sc-394039 | IF (1/20000) |
| Chemical compound, drug | Prolong Diamond | Thermo Fisher | Thermo Fisher:P36965 | |
| Chemical compound, drug | zaprinast | Calbiochem | Calbiochem:684500 | |
| Chemical compound, drug | A23187 | Calbiochem | Calbiochem:100105 | |
| Commercial assay or kit | S-trap micro | Protifi | Protific:C02-micro-80 | |
| Commercial assay or kit | TMT10plex Isobaric Label Reagent Set | Thermo Fisher Scientific | Thermo Fisher Scientific:90111 | |
| Commercial assay or kit | TMTpro 16plex Label Reagent Set | Thermo Fisher Scientific | Thermo Fisher Scientific:A44522 | |
| Commercial assay or kit | High-Select TiO2 Phosphopeptide Enrichment Kit | Thermo Fisher Scientific | Thermo Fisher Scientific:A32993 | |
| Commercial assay or kit | High-Select Fe-NTA Phosphopeptide Enrichment Kit | Thermo Fisher Scientific | Thermo Fisher Scientific:A32992 | |
| Commercial assay or kit | Pierce High pH Reversed-Phase Peptide Fractionation Kit | Thermo Fisher Scientific | Thermo Fisher Scientific:84868 | |
| Commercial assay or kit | Calcium Calibration Buffer Kit #1, zero and 10 mM CaEGTA | Life Technologies | Life Technologies:C3008MP | |
| Commercial assay or kit | Hydrophobic Sera-Mag Speed Beads | GE Healthcare | GE Healthcare:65152105050250 | |
| Commercial assay or kit | Hydrophilic Sera-Mag Speed Beads | GE Healthcare | GE Healthcare:45152105050250 | |
| Recombinant DNA reagent | All plasmids used in this study are listed in Supplementary file 7 | |||
| Sequence-based reagent | All primers and oligonucleotides used in this study are listed in Supplementary file 7 | |||
| Software, algorithm | Proteome Discoverer 4.2 | Thermo Fisher | ||
| Software, algorithm | R version 4.0 | R Foundation for Statistical Computing | ||
| Software, algorithm | mineCETSA version 1.1.1 | Dziekan et al., 2020 | https://github.com/nkdailingyun/mineCETSA | |
| Software, algorithm | Prism 8 | GraphPad | ||
| Software, algorithm | HHPRED | PMID:29258817 | ||
| Other | Halt protease inhibitor | Thermo Fisher | Thermo Fisher:87786 | Materials and Methods: lysis buffer |
| Other | Halt protease and phosphatase inhibitor | Thermo Fisher | Thermo Fisher:PI78440 | Materials and Methods: lysis buffer |
| Other | Benzonase | Sigma Aldrich | Sigma Aldrich:E1014 | Materials and Methods: lysis buffer |
Additional files
-
Supplementary file 1
Sub-minute phosphoproteomics time course protein and abundance assignments from Proteome Discoverer 2.4.
- https://cdn.elifesciences.org/articles/80336/elife-80336-supp1-v3.txt
-
Supplementary file 2
Sub-minute phosphoproteomics time course peptide and abundance assignments from Proteome Discoverer 2.4.
Mclust cluster assignments (column 118) of phosphopeptides dynamically changing during zaprinast treatment.
- https://cdn.elifesciences.org/articles/80336/elife-80336-supp2-v3.txt
-
Supplementary file 3
Data pertaining to the temperature range thermal profiling experiment.
1. Protein and abundance assignments from Proteome Discoverer 2.4 for samples with 0 µM Ca2+, replicate 1. 2. Protein and abundance assignments from Proteome Discoverer 2.4 for samples with 0 µM Ca2+, replicate 2. 3. Protein and abundance assignments from Proteome Discoverer 2.4 for samples with 10 µM Ca2+, replicate 1. 4. Protein and abundance assignments from Proteome Discoverer 2.4 for samples with 10 µM Ca2+, replicate 2. 5. Curve fit output from the mineCETSA package. 6. Area under the euclidean distance score calculations from the mineCETSA package.
- https://cdn.elifesciences.org/articles/80336/elife-80336-supp3-v3.xlsx
-
Supplementary file 4
Data pertaining to the concentration range thermal profiling experiments.
1. Protein and abundance assignments from Proteome Discoverer 2.4 for Experiment 1 samples with 54 °C, replicate 1. 2. Protein and abundance assignments from Proteome Discoverer 2.4 for Experiment 1 samples with 54 °C, replicate 2. 3. Protein and abundance assignments from Proteome Discoverer 2.4 for Experiment 1 samples with 58 °C, replicate 1. 4. Protein and abundance assignments from Proteome Discoverer 2.4 for Experiment 1 samples with 58 °C, replicate 2. 5. Curve fit output for concentration range Experiment 1 from the mineCETSA package. 6. Area under the curve score calculations from the mineCETSA package for concentration range Experiment 2. 7. Protein and abundance assignments from Proteome Discoverer 2.4 for Experiment 2 samples with 50 °C, replicate 1. 8. Protein and abundance assignments from Proteome Discoverer 2.4 for Experiment 2 samples with 50 °C, replicate 2. 8. Protein and abundance assignments from Proteome Discoverer 2.4 for Experiment 2 samples with 54 °C, replicate 1. 9. Protein and abundance assignments from Proteome Discoverer 2.4 for Experiment 2 samples with 54 °C, replicate 2. 10. Protein and abundance assignments from Proteome Discoverer 2.4 for Experiment 2 samples with 58 °C, replicate 1. 11. Protein and abundance assignments from Proteome Discoverer 2.4 for Experiment 2 samples with 58 °C, replicate 2. 12. Curve fit output for concentration range Experiment 2 from the mineCETSA package. 13. Area under the curve score calculations from the mineCETSA package for concentration range Experiment 2.
- https://cdn.elifesciences.org/articles/80336/elife-80336-supp4-v3.xlsx
-
Supplementary file 5
PP1 depletion zaprinast phosphoproteomics time course protein and abundance assignments from Proteome Discoverer 2.4.
- https://cdn.elifesciences.org/articles/80336/elife-80336-supp5-v3.txt
-
Supplementary file 6
PP1 depletion zaprinast phosphoproteomics time course peptide and abundance assignments from Proteome Discoverer 2.4.
Mclust cluster assignments (column 2) of phosphopeptides dynamically changing during zaprinast treatment when PP1 is depleted.
- https://cdn.elifesciences.org/articles/80336/elife-80336-supp6-v3.txt
-
Supplementary file 7
Sequences and accessions of oligonucleotides and plasmids used in this study.
- https://cdn.elifesciences.org/articles/80336/elife-80336-supp7-v3.xlsx
-
MDAR checklist
- https://cdn.elifesciences.org/articles/80336/elife-80336-mdarchecklist1-v3.docx





