Comparative analysis of two Caenorhabditis elegans kinesins KLP-6 and UNC-104 reveals a common and distinct activation mechanism in kinesin-3
Figures
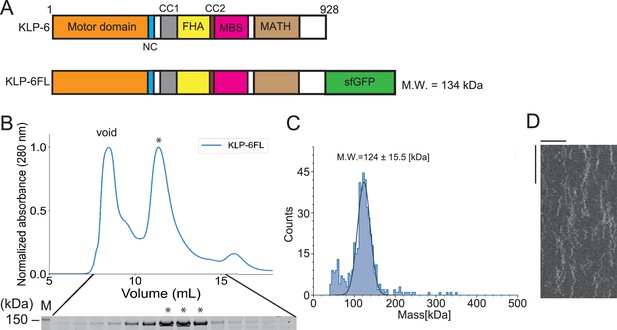
Full-length KLP-6 shows diffusive motion on microtubules.
(A) Schematic drawing of the domain organization of KLP-6 motor protein and the full length of KLP-6 fused with sfGFP (KLP-6FL). NC, neck coiled-coil domain. CC1, coiled-coil 1 domain. FHA, forkhead-associated domain. CC2, coiled-coil 2 domain. MBS, membrane-associated guanylate kinase homolog (MAGUK)-binding stalk domain. MATH, meprin and TRAF homology domain. Calculated molecular weight (MW) is shown at the right side. (B) Size exclusion chromatography of KLP-6FL. The SDS-PAGE of the elution fractions is shown beneath the profile. Asterisks indicate fractions used for mass photometry and single molecule assays. The void volume of the column is indicated. Asterisks indicate fractions that are used for mass photometry and total internal reflection fluorescent (TIRF) assays. Number shown at the left side indicates a molecular weight standard. (C) Mass photometry of KLP-6FL. Histogram shows the particle count of KLP-6FL at 20 nM. The line shows a Gaussian fit (mean ± standard deviation [SD]: 124±15.5 kDa). (D) Representative kymographs showing the motility of 5 pM KLP-6FL in the presence of 2 mM ATP. Note that KLP-6FL shows only one-dimensional diffusion on microtubules but does not show any processive runs. Horizontal and vertical bars shows 10 µm and 10 s, respectively.
-
Figure 1—source data 1
Original file for the SDS-PAGE analysis in Figure 1B (KLP-6FL).
- https://cdn.elifesciences.org/articles/89040/elife-89040-fig1-data1-v1.zip
-
Figure 1—source data 2
PDF containing Figure 1B and a original scan of the relevant SDS-PAGE analysis (KLP-6FL) with a highlighted band.
- https://cdn.elifesciences.org/articles/89040/elife-89040-fig1-data2-v1.zip
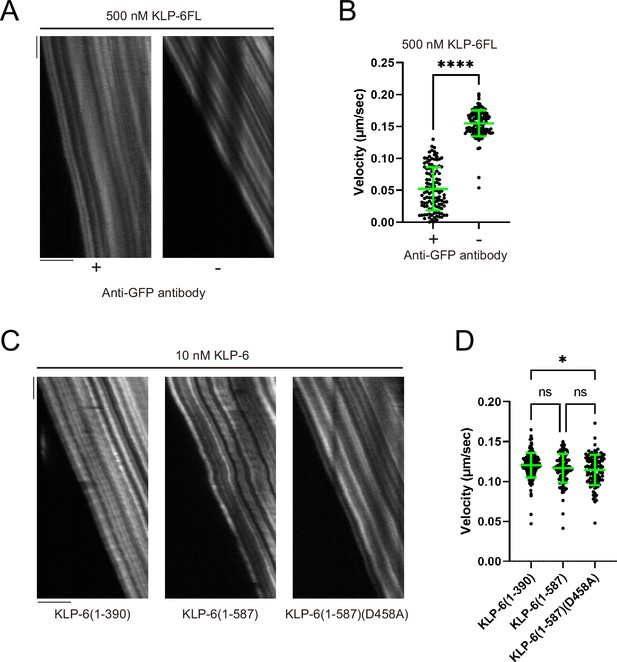
Microtubule gliding assays using KLP-6.
(A) Representative kymographs showing the microtubule gliding with 500 nM KLP-6FL. KLP-6FL was attached to the glass surface using anti-GFP antibody (+) or directly (-). Horizontal and vertical bars show 5 µm and 10 s, respectively. (B) Dot plots showing the griding velocity of KLP-6FL when attached to the glass surface with (+) or without (-) anti-GFP antibody. Green bars represent mean ± SD, n=115 and 127 microtubules for KLP-6FL with and without anti-GFP antibody. Student’s t-test. ****, p<0.0001. (C) Representative kymographs showing the microtubule gliding with 10 nM KLP-6 (1–390), KLP-6 (1–587), and KLP-6 (1–587)(D458A). The motor was attached to the glass surface using anti-GFP antibody. Horizontal and vertical bars show 5 µm and 10 s, respectively. (D) Dot plots showing the griding velocity of 10 nM KLP-6 (1–390), KLP-6 (1–587), and KLP-6 (1–587)(D458A). Each dot shows a single datum point. Each dot shows a single datum point. Green bars represent mean ± SD, n=180, 107, and 103 microtubules for KLP-6 (1–390), KLP-6 (1–587), and KLP-6 (1–587)(D458A). One-way ANOVA test followed by Tukey’s multiple comparisons test. *, p<0.05. ns, p>0.05 and statistically not significant.
-
Figure 1—figure supplement 1—source data 1
Velocities in the microtubule-gliding assays.
The velocity of microtubule gliding assays are shown. Dot plots are shown in Figure 1—figure supplement 1B, D, respectively.
- https://cdn.elifesciences.org/articles/89040/elife-89040-fig1-figsupp1-data1-v1.xlsx

KLP-6 has a second microtubule binding domain.
Schematic drawing of the domain organization of KLP-6 analyzed and representative total internal reflection fluorescent (TIRF) images showing the bindings of GFP fused KLP-6 deletion mutant proteins (GFP, green) to microtubules (MT, cyan). The tail domain alone binds to microtubules. Bar, 10 µm.
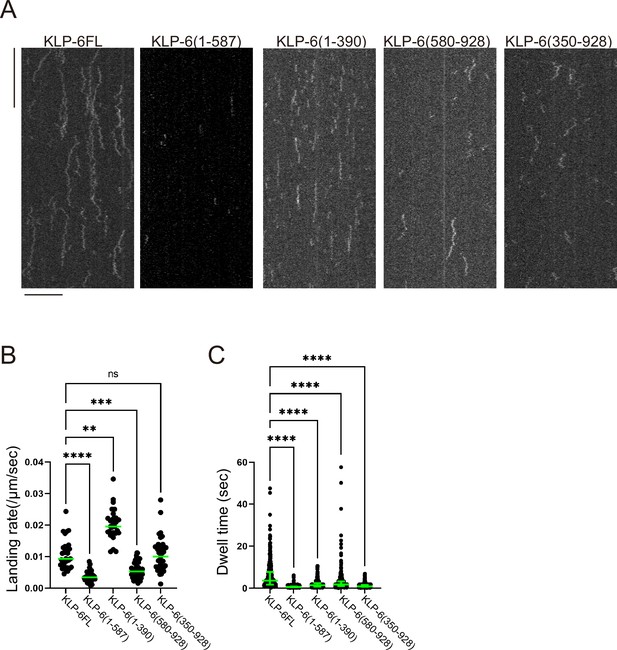
Purified KLP-6 and its deletion mutants do not show processive runs.
(A) Representative kymographs showing the motility of 5 pM KLP-6FL, KLP-6 (1–587), KLP-6 (1–390), KLP-6 (350–928), and KLP-6 (580–928) on microtubules in the presence of 2 mM ATP. Horizontal and vertical bars show 10 µm and 10 s, respectively. None of the KLP-6 proteins shows directional movements on microtubules. (B) Dot plots showing the landing rate of KLP-6FL, KLP-6 (1–587), KLP-6 (1–390), KLP-6 (350–928), and KLP-6 (580–928). Each dot shows a single datum point. Green bars represent median value. n=31, 33, 27, 42, and 39 microtubules for KLP-6FL, KLP-6 (1–587), KLP-6 (1–390), KLP-6 (350–928), and KLP-6 (580–928). Kruskal-Wallis test followed by Dunn’s multiple comparison test. **, p<0.01, p<0.001, and ****, p<0.0001. (C) Dot plots showing the dwell time on microtubules of KLP-6FL, KLP-6 (1–587), KLP-6 (1–390), KLP-6 (350–928), and KLP-6 (580–928). Each dot shows a single datum point. Green bars represent median value and interquartile range. n=348 and 338, 326, 412, and 403 particles in KLP-6FL, KLP-6 (1–587), KLP-6 (1–390), KLP-6 (350–928), and KLP-6 (580–928), respectively. Kruskal-Wallis test followed by Dunn’s multiple comparison test. ****, p<0.0001.
-
Figure 2—figure supplement 1—source data 1
Landing rate and dwell time of KLP-6 mutants.
The result of single molecule assays using KLP-6 mutant proteins. Landing rate and dwell time are shown. Dot plots are shown in Figure 2—figure supplement 1B, C, respectively.
- https://cdn.elifesciences.org/articles/89040/elife-89040-fig2-figsupp1-data1-v1.xlsx
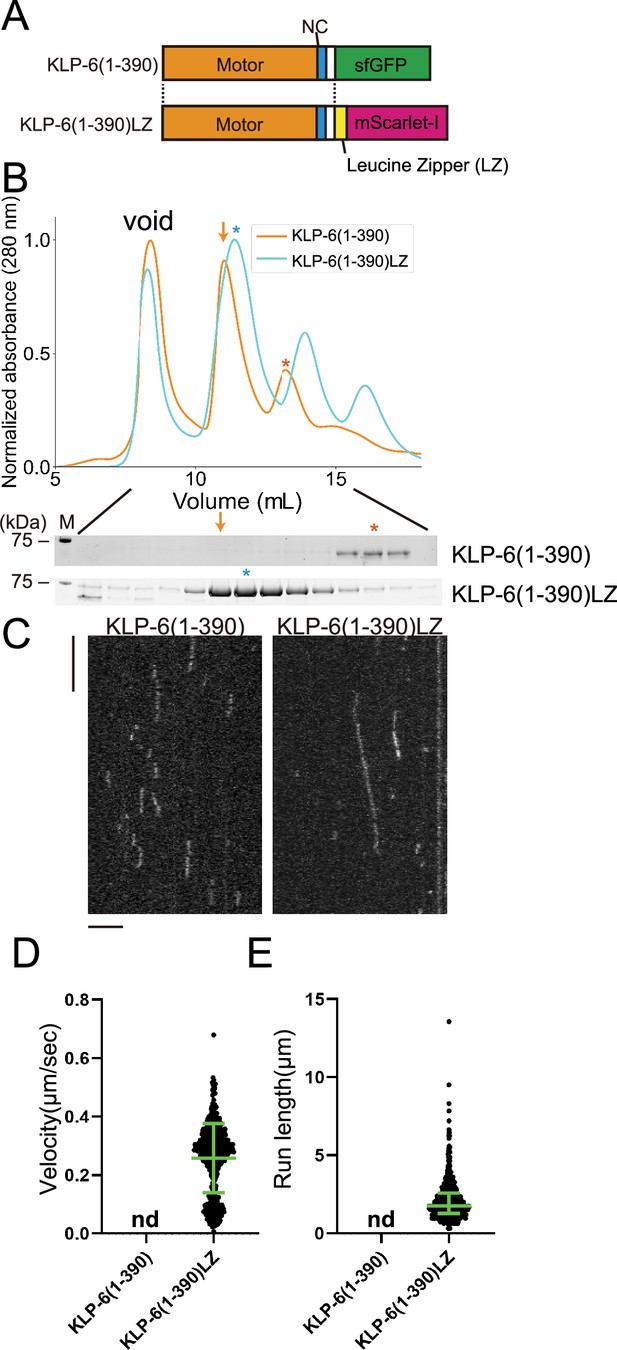
KLP-6 is converted to a processive motor upon dimerization.
(A) Schematic drawing of the domain organization of KLP-6 (1–390) and KLP-6 (1–390)LZ. (B) Size exclusion chromatography of KLP-6 (1–390) (orange) and KLP-6 (1–390)LZ (cyan). The SDS-PAGE of the elution fractions are shown beneath the profiles. The number shown at the left side indicates molecular weight standard. Orange and cyan asterisks indicate fractions used for single molecule assays. Note that the fraction indicated by an orange arrow does not contain KLP-6 (1–390) protein. (C) Representative kymographs showing the motility of 5 pM KLP-6 (1–390) and KLP-6 (1–390)LZ in the presence of 2 mM ATP. Horizontal and vertical bars show 5 s and 5 µm, respectively. (D) Dot plots showing the velocity of KLP-6 (1–390) and KLP-6 (1–390)LZ. Each dot shows a single datum point. Green bars represent mean ± SD, n=642 for KLP-6 (1–390)LZ. n.d., no directional movement was detected in KLP-6 (1–390). (E) Dot plots showing the run length of KLP-6 (1–390) and KLP-6 (1–390)LZ. Each dot shows a single datum point. Green bars represent median value and interquartile range. n=642 for KLP-6 (1–390)LZ. n.d., no directional movement was detected in KLP-6 (1–390).
-
Figure 3—source data 1
Velocity and run length of KLP-6 (1–390)LZ.
The result of single molecule assays using KLP-6 (1–390)LZ. Velocity and run length are shown. Dot plots are shown in Figure 3D and E, respectively.
- https://cdn.elifesciences.org/articles/89040/elife-89040-fig3-data1-v1.xlsx
-
Figure 3—source data 2
Original file for the SDS-PAGE analysis in Figure 3B (KLP-6(1-390) and KLP-6(1-390)LZ).
- https://cdn.elifesciences.org/articles/89040/elife-89040-fig3-data2-v1.zip
-
Figure 3—source data 3
PDF containing Figure 3B and original scans of the relevant SDS-PAGE analysis (KLP-6(1-390) and KLP-6(1-390)LZ) with highlighted bands.
- https://cdn.elifesciences.org/articles/89040/elife-89040-fig3-data3-v1.zip

KLP-6 is a constitutive monomer.
(A) Schematic drawing of the domain organization of KLP-6 (1–587) and UNC-104(1–653). Calculated molecular weight is written at the right side. (B) Size exclusion chromatography of KLP-6 (1–587) (plum) and KLP-6 (1–587)(D458A) (purple). The SDS-PAGE of the elution fractions are shown beneath the profiles. Asterisks indicate fractions used for mass photometry and single molecule assays. The number shown at the left side indicates molecular weight standard. (C) Mass photometry of KLP-6 (1–587) and KLP-6 (1–587)(D458A). Histograms show the normalized particle count of KLP-6 (1–587) (blue) and KLP-6 (1–587)(D458A) (orange) at 40 nM. Lines show Gaussian fits (mean ± SD: 94±44 kDa and 88±35 kDa for KLP-6 (1–587) and KLP-6 (1–587)(D458A), respectively). (D) Representative kymographs showing the motility of 5 pM KLP-6 (1–587) and KLP-6 (1–587)(D458A) in the presence of 2 mM ATP. Note that no directional movement was detected in either case. Horizontal and vertical bars show 10 µm and 10 s, respectively. (E) Dot plots showing the landing rate of KLP-6 (1–587) and KLP-6 (1–587)(D458A). Each dot shows a single datum point. Green bars represent median value. n=33 microtubules. Mann-Whitney U test. ****, p<0.0001. (F) Dot plots showing the dwell time of KLP-6 (1–587) and KLP-6 (1–587)(D458A) on microtubules. Each dot shows a single datum point. Green bars represent median value and interquartile range. n=338 and 351 particles for KLP-6 (1–587) and KLP-6 (1–587)(D458A), respectively. Mann-Whitney U test. ****, p<0.0001.
-
Figure 4—source data 1
Landing rate and dwell time of KLP-6 (1–587) and KLP-6 (1–587)(D458A).
The result of single molecule assays using KLP-6 (1–587) and KLP-6 (1–587)(D458A). Landing rate and dwell time are shown. Dot plots are shown in Figure 4E and F, respectively.
- https://cdn.elifesciences.org/articles/89040/elife-89040-fig4-data1-v1.xlsx
-
Figure 4—source data 2
Original file for the SDS-PAGE analysis in Figure 4B (KLP-6(1-587) and KLP-6(1-587)(D458A)).
- https://cdn.elifesciences.org/articles/89040/elife-89040-fig4-data2-v1.zip
-
Figure 4—source data 3
PDF containing Figure 4B and original scans of the relevant SDS-PAGE analysis (KLP-6(1-587) and KLP-6(1-587)(D458A)) with highlighted bands.
- https://cdn.elifesciences.org/articles/89040/elife-89040-fig4-data3-v1.zip
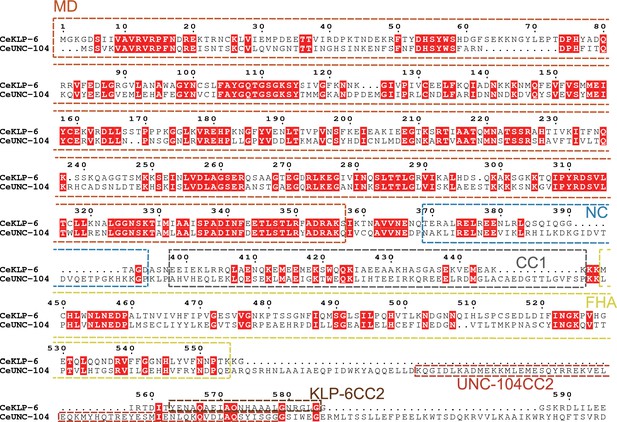
Comparison of KLP-6 and UNC-104.
Amino acid sequences of KLP-6 and UNC-104 are aligned using CLUSTALW on ESPript 3 (https://espript.ibcp.fr/ESPript/cgi-bin/ESPript.cgi). The motor domain, neck coil domain, coiled-coil 1 (CC1) domain, forkhead-associated (FHA) domain, and coiled-coil 2 (CC2) domains are indicated.
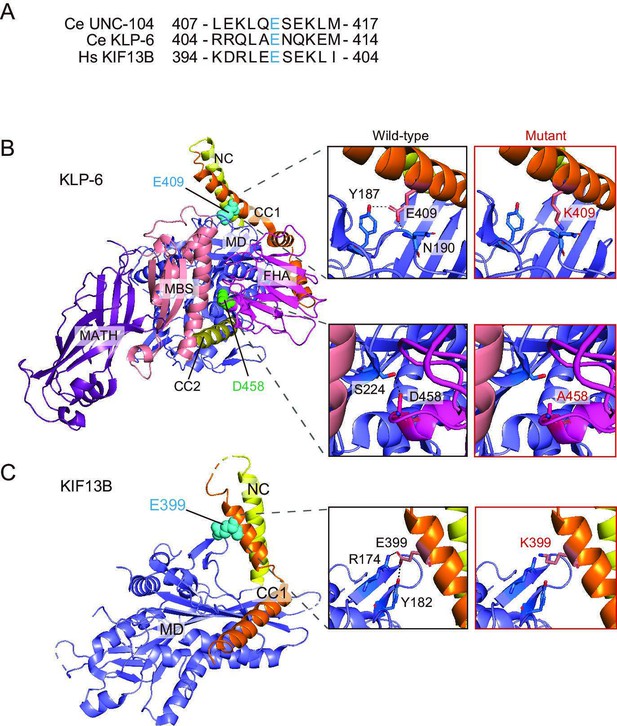
Structural model of KLP-6 and KIF13B.
(A) Sequence comparison between Ce (C. elegans) UNC-104, Ce (C. elegans) KLP-6, and Hs (Homo sapiens) KIF13B. Ce UNC-104(E412) is conserved in Ce KLP-6(E409) and Hs KIF13B(E399). (B) Crystal structure of full-length KLP-6 (PDB 7WRG). The cyan sphere represents E409 from the coiled-coil 1 (CC1) domain, which interacts with Y187 and N190 from the motor domain (MD), while the mutation K409 does not interact with those residues. This mutation disrupts the MD-CC1 domain interaction and autoinhibition of KLP-6. The green sphere represents D458 from the forkhead-associated (FHA) domain, which interacts with S224 from the MD, while the mutation A458 does not interact with this residue. This mutation disrupts the MD-FHA domain interaction and autoinhibition of KLP-6. (C) Crystal structure of MD-NC-CC1 of KIF13B (PDB 6A20). The cyan sphere represents E399 from the CC1 domain, which interacts with R174 and Y182 from the MD, while the mutation K399 does not interact with those residues. This mutation disrupts the MD-CC1 domain interaction and autoinhibition of KIF13B. The mutations were generated using PyMOL.
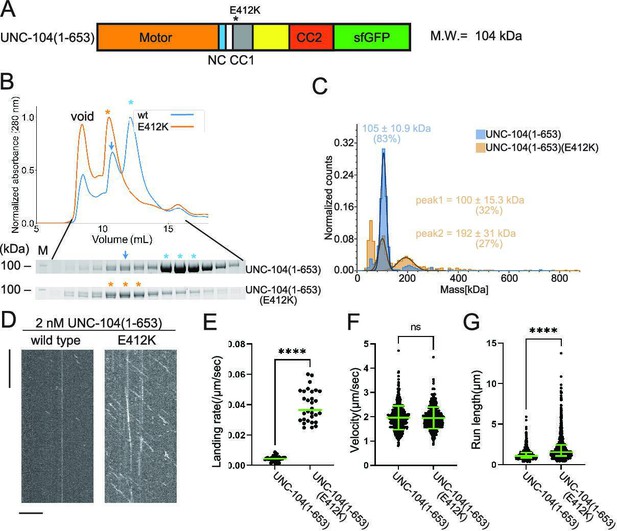
UNC-104 is converted to a processive dimer upon autoinhibition release.
(A) Schematic drawing of the domain organization of UNC-104(1–653). Calculated molecular weight is written at the right side. (B) Size exclusion chromatography of UNC-104(1–653) (blue) and UNC-104(1–653)(E412K) (orange). The SDS-PAGE of the elution fractions are shown beneath the profiles. Blue and orange asterisks indicate fractions used for mass photometry and single molecule assays. The number shown at the left side indicates molecular weight standard. Blue arrows indicate expected dimer fraction of wild-type UNC-104(1–653). (C) Mass photometry of UNC-104(1–653) and UNC-104(1–653)(E412K). Histograms show the normalized particle count of UNC-104(1–653) (blue) and UNC-104(1–653)(E412K) (orange) at 10 nM. Lines show Gaussian fits. UNC-104(1–653) is distributed within 100±15.3 kDa range (mean ± SD). UNC-104(1–653)(E412K) shows two peaks consisting of 32% and 27% of particles which are distributed within 100±15.3 kDa and 192±31 kDa range, respectively (mean ± SD). Note that wild-type UNC-104(1–653) also has a very small peak around 200 kDa, but the number of datum point is too little for Gaussian fitting. (D) Representative kymographs showing the motility of 2 nM UNC-104(1–653) and UNC-104(1–653)(E412K) in the presence of 2 mM ATP. Horizontal and vertical bars show 10 µm and 10 s, respectively. (E) Dot plots showing the landing rate of UNC-104(1–653) and UNC-104(1–653)(E412K). Each dot shows a single datum point. Green bars represent median value n=31 and 30 microtubules for UNC-104(1–653) and UNC-104(1–653)(E412K), respectively. Mann-Whitney U test. ****, p<0.0001. (F) Dot plots showing the velocity of UNC-104(1–653) and UNC-104(1–653)(E412K). Each dot shows a single datum point. Green bars represent mean ± SD, n=603 and 624 particles for UNC-104(1–653) and UNC-104(1–653)(E412K). Student’s t-test, ns, p=0.7 and statistically not significant. (G) Dot plots showing the run length of UNC-104(1–653) and UNC-104(1–653)(E412K). Each dot shows a single datum point. Green bars represent median value and interquartile range. n=603 and 624 particles for UNC-104(1–653) and UNC-104(1–653)(E412K). Mann-Whitney U test. ****, p<0.0001.
-
Figure 5—source data 1
Landing rate, run length, and velocity of UNC-104(1–653) and UNC-104(1–653)(E412K).
The result of single molecule assays using UNC-104(1–653) and UNC-104(1–653)(E412K). Landing rate, run length, and velocity are shown. Dot plots are shown in Figure 5E, F, and G, respectively.
- https://cdn.elifesciences.org/articles/89040/elife-89040-fig5-data1-v1.xlsx
-
Figure 5—source data 2
Original file for the SDS-PAGE analysis in Figure 5B (UNC-104(1-653) and UNC-104(1-653)(E412K)).
- https://cdn.elifesciences.org/articles/89040/elife-89040-fig5-data2-v1.zip
-
Figure 5—source data 3
PDF containing Figure 5B and original scans of the relevant SDS-PAGE analysis (UNC-104(1-653) and UNC-104(1-653)(E412K)) with highlighted bands.
- https://cdn.elifesciences.org/articles/89040/elife-89040-fig5-data3-v1.zip
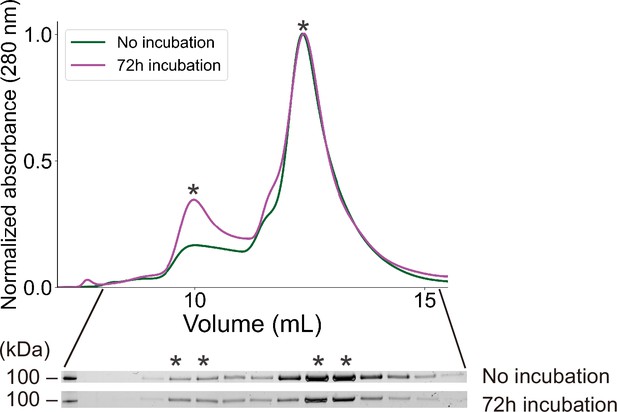
Monomer-dimer re-equilibrium of monomeric UNC-104(1–653).
Size exclusion chromatography of the monomer fraction of UNC-104(1–653), indicated by the blue asterisk in Figure 5B in the main text. Without incubation (green), the dimer peak (left asterisk) was minimal, but after 72 hr of incubation at 4°C (plum), the peak increased. The SDS-PAGE of the elution fractions are shown beneath the profile. The number shown at the left side indicates molecular weight standard.
-
Figure 5—figure supplement 1—source data 1
Original file for the SDS-PAGE analysis in Figure 5—figure supplement 1 (UNC-104(1-653)).
- https://cdn.elifesciences.org/articles/89040/elife-89040-fig5-figsupp1-data1-v1.zip
-
Figure 5—figure supplement 1—source data 2
PDF containing Figure 5-figure supplement 1 and original scans of the relevant SDS-PAGE analysis (UNC-104(1-653)) with highlighted bands.
- https://cdn.elifesciences.org/articles/89040/elife-89040-fig5-figsupp1-data2-v1.zip
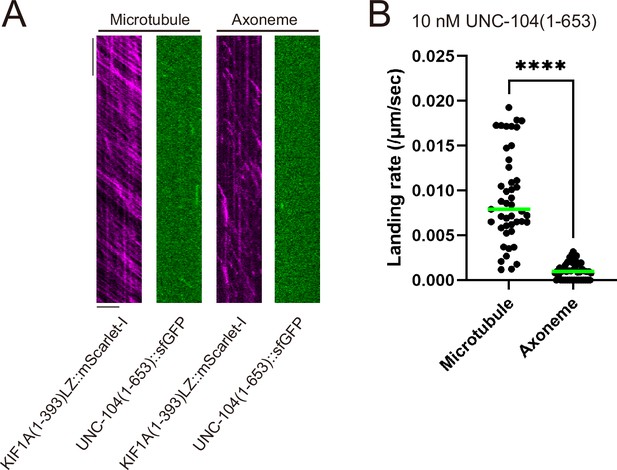
Single molecule analysis using Chlamydomonas axonemes.
(A) Representative kymographs showing the motility of 0.2 nM KIF1A(1–393)LZ::mScarlet-I and 10 nM UNC-104(1–653)::sfGFP on purified porcine microtubules within the same chamber, as well as the motility of 0.2 nM KIF1A(1–393)LZ::mScarlet-I and 10 nM UNC-104(1–653)::sfGFP on Chlamydomonas axonemes within the same chamber. Horizontal and vertical bars show 5 µm and 5 s, respectively. UNC-104(1–653)::sfGFP shows no processive runs on Chlamydomonas axonemes. (D) Dot plots showing the landing rate of UNC-104(1–653)::sfGFP on purified porcine microtubules and Chlamydomonas axonemes. Each dot shows a single datum point. Green bars represent median value. n=45 and 46 microtubules for UNC-104(1–653)::sfGFP on purified porcine microtubules and Chlamydomonas axonemes, respectively. Mann-Whitney U test. ****, p<0.0001.
-
Figure 5—figure supplement 2—source data 1
Landing rate of UNC-104(1–653) on microtubules and axonemes.
The property of microtubules and axonemes are compared using UNC-104(1–653). Landing rate is shown. A dot plot is shown in Figure 5—figure supplement 2B.
- https://cdn.elifesciences.org/articles/89040/elife-89040-fig5-figsupp2-data1-v1.xlsx
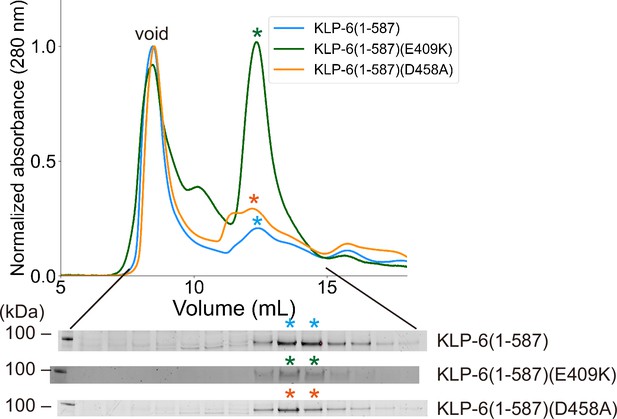
KLP-6(E409K) does not form a dimer.
Size exclusion chromatography of KLP-6 (1–587) (plum), KLP-6 (1–587)(E409K) (green), and KLP- 6 (1–587)(D458A) (purple). The SDS-PAGE of the elution fractions are shown beneath the profile. Data for KLP-6 (1–587) and KLP-6 (1–587)(D458A) are replotted and reshown from Figure 4B. Note that no significant differences are observed among the elution profiles of the three proteins. The number shown at the left side indicates molecular weight standard.
-
Figure 5—figure supplement 3—source data 1
Original file for the SDS-PAGE analysis in Figure 5—figure supplement 3 (KLP-6(1-587)(E409K)).
- https://cdn.elifesciences.org/articles/89040/elife-89040-fig5-figsupp3-data1-v1.zip
-
Figure 5—figure supplement 3—source data 2
PDF containing Figure 5—figure supplement 3 and a original scan of the relevant SDS-PAGE analysis (KLP-6(1-587)(E409K)) with a highlighted band.
- https://cdn.elifesciences.org/articles/89040/elife-89040-fig5-figsupp3-data2-v1.zip

Coiled-coil 2 (CC2) domain of UNC-104, but not KLP-6, is capable to form a dimer.
(A) Coiled coil prediction of KLP-6 (aa 1–587) and UNC-104 (aa 1–653). NC, CC1, and CC2 domains are indicated. Prediction was performed using the Marcoil algorism. (B) Schematic drawing of the domain organization of mScarlet-I(mSca), KLP-6CC2-mSca, and UNC-104CC2-mSca analyzed in panel C. (C) Size exclusion chromatography of mSca (blue), KLP-6CC2-mSca (plum), and UNC-104CC2-mSca (orange). The SDS-PAGE of the elution fractions are shown beneath the profiles. The number shown at the left side indicates molecular weight standard.
-
Figure 6—source data 1
Original file for the SDS-PAGE analysis in Figure 6C (mSca, KLP-6CC2-mSca, and UNC-104CC2-mSca).
- https://cdn.elifesciences.org/articles/89040/elife-89040-fig6-data1-v1.zip
-
Figure 6—source data 2
PDF containing Figure 6C and original scans of the relevant SDS-PAGE analysis (mSca, KLP-6CC2-mSca, and UNC-104CC2-mSca) with highlighted bands.
- https://cdn.elifesciences.org/articles/89040/elife-89040-fig6-data2-v1.zip

The coiled-coil 2 (CC2) domain is essential for the stable dimer formation in UNC-104.
(A) Schematic drawing of the domain organization of UNC-104(1–653) and UNC-104(1–594). Calculated molecular weight is shown at the right side. (B) Size exclusion chromatography of UNC-104(1–594) (blue) and UNC-104(1–594)(E412K) (orange). The SDS-PAGE of the elution fractions are shown beneath the profiles. The number shown at the left side indicates molecular weight standard. Both proteins show almost identical profile. (C) Mass photometry of UNC-104(1–594) (blue) and UNC-104(1–594)(E412K) (orange) at 10 nM. Lines show Gaussian fits. Both UNC-104(1–594) and UNC-104(1–594)(E412K) have a single peak which is Gaussian distributed within 98±14.3 kDa range and within 99±13.6 kDa range, respectively (mean ± SD). (D) Representative kymographs showing the motility of 2 nM UNC-104(1–594) and UNC-104(1–594)(E412K) in the presence of 2 mM ATP. Horizontal and vertical bars show 10 µm and 10 s, respectively. (E) Dot plots showing the landing rate of UNC-104(1–594), UNC-104(1–594)(E412K), UNC-104(1–653), and UNC-104(1–653)(E412K). For comparison, data for UNC-104(1–653) and UNC-104(1–653)(E412K) are replotted from Figure 5E. Each dot shows a single datum point. Green bars represent median value and interquartile range. N=31, 32, 31, 30 microtubules for UNC-104(1–594), UNC-104(1–594)(E412K), UNC-104(1–653), and UNC-104(1–653)(E412K), respectively. Kruskal-Wallis test followed by Dunn’s multiple comparison test. ***, p<0.001. ****, p<0.0001. ns, p>0.05 and statistically not significant. (F) Dot plots showing the velocity of UNC-104(1–594), UNC-104(1–594)(E412K), UNC-104(1–653), and UNC-104(1–653)(E412K). For comparison, data for UNC-104(1–653) and UNC-104(1–653)(E412K) are replotted from Figure 5F. Green bars represent mean ± SD, n=467, 609, 603, 624 particles for UNC-104(1–594), UNC-104(1–594)(E412K), UNC-104(1–653), and UNC-104(1–653)(E412K), respectively. One-way ANOVA test followed by Šidák’s multiple comparison test. ns, p>0.05 and statistically not significant. (G) Dot plots showing the run length of UNC-104(1–594), UNC-104(1–594)(E412K), UNC-104(1–653), and UNC-104(1–653)(E412K). For comparison, data for UNC-104(1–653) and UNC-104(1–653)(E412K) are replotted from Figure 5G. Each dot shows a single datum point. Green bars represent median value and interquartile range. Kruskal-Wallis test followed by Dunn’s multiple comparison test. n=467, 609, 603, 624 particles for UNC-104(1–594), UNC-104(1–594)(E412K), UNC-104(1–653), and UNC-104(1–653)(E412K), respectively. ****, p<0.0001. ns, p>0.05 and statistically not significant.
-
Figure 7—source data 1
Landing rate, run length, and velocity of UNC-104(1–594) and UNC-104(1–594)(E412K).
The result of single molecule assays using UNC-104(1–594) and UNC-104(1–594)(E412K). Landing rate, run length, and velocity are shown. Dot plots are shown in Figure 7E, F, and G, respectively.
- https://cdn.elifesciences.org/articles/89040/elife-89040-fig7-data1-v1.xlsx
-
Figure 7—source data 2
Original file for the SDS-PAGE analysis in Figure 7B (UNC-104(1-594) and UNC-104(1-594)(E412K)).
- https://cdn.elifesciences.org/articles/89040/elife-89040-fig7-data2-v1.zip
-
Figure 7—source data 3
PDF containing Figure 7B and original scans of the relevant SDS-PAGE analysis (UNC-104(1-594) and UNC-104(1-594)(E412K)) with highlighted bands.
- https://cdn.elifesciences.org/articles/89040/elife-89040-fig7-data3-v1.zip
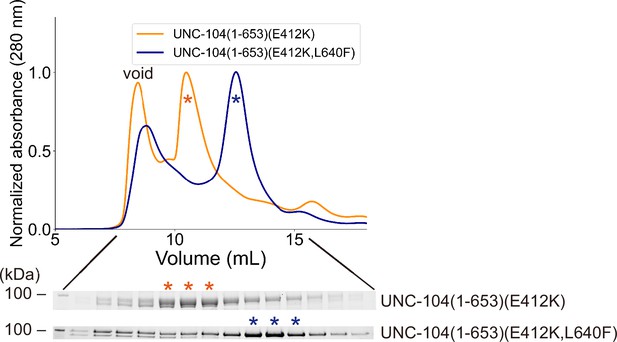
UNC-104(E412K,L640F) does not form a dimer.
Size exclusion chromatography of UNC-104(1–653)(E412K) (orange) and UNC-104(1–653)(E412K,L640F) (navy blue). The SDS-PAGE of the elution fractions are shown beneath the profile. Data for UNC-104(1–653)(E412K) are replotted and reshown from Figure 5B. Note that UNC-104(1–653)(E412K,L640F) predominantly eluted in the monomer peak, in contrast to UNC-104(1–653)(E412K), which predominantly eluted in the dimer peak. The number shown at the left side indicates molecular weight standard.
-
Figure 7—figure supplement 1—source data 1
Original file for the SDS-PAGE analysis in Figure 7—figure supplement 1 (UNC-104(1-653)(E412K,L640F)).
- https://cdn.elifesciences.org/articles/89040/elife-89040-fig7-figsupp1-data1-v1.zip
-
Figure 7—figure supplement 1—source data 2
PDF containing Figure 7—figure supplement 1 and a original scan of the relevant SDS-PAGE analysis (UNC-104(1-653)(E412K,L640F)) with a highlighted band.
- https://cdn.elifesciences.org/articles/89040/elife-89040-fig7-figsupp1-data2-v1.zip
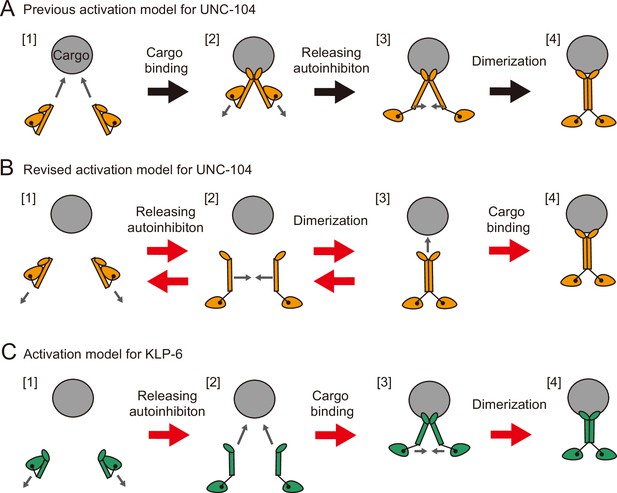
Activation models for UNC-104 and KLP-6.
(A) Previous activation model for UNC-104. UNC-104 initially exists in a monomeric state in solution and subsequently binds to cargo (transition 1 → 2), triggering the release of its autoinhibition (transition 2 → 3). On the cargo membrane, the motor can then undergo dimerization (transition 3 → 4), initiating processive transport on microtubules. (B) Proposed activation model for UNC-104. Upon release of autoinhibition (transition 1 → 2), UNC-104 can dimerize independently of cargo binding (transition 2 → 3). Note that the transitions 1 → 2 and 2 → 3 are reversible. Following dimerization, the motor can bind to cargo and initiate processive transport on microtubules. (C) Proposed activation model for KLP-6. KLP-6 is strongly autoinhibited and primarily exists as a monomer in solution. Upon release of autoinhibition (transition 1 → 2), KLP-6 engages with cargo (transition 2 → 3), facilitating subsequent dimerization of the motor (transition 3 → 4). Subsequently, the motor can initiate processive transport on microtubules.
Proteins analyzed in this study.
Full scan images of SDS-PAGE showing the purified proteins analyzed in this study. Lane M, molecular weight markers. Numbers shown at the left side indicate the molecular weight standard. (A) KLP-6 proteins. (B) UNC-104 proteins.
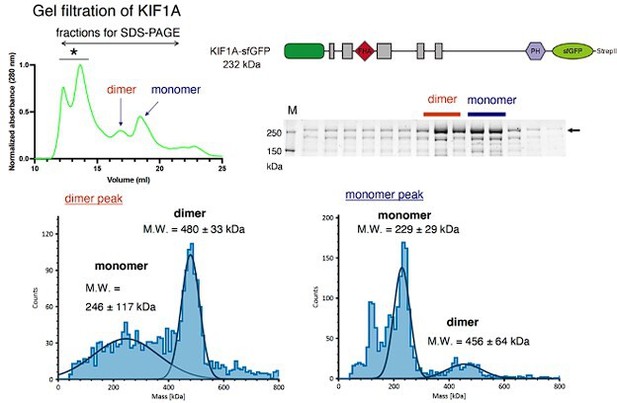
Upper left, A representative result of size exclusion chromatography obtained from the analysis of full-length human KIF1A fused with sfGFP.
Upper right, A schematic drawing showing the structure of KIF1A fused with sfGFP and a result of SDS-PAGE recovered from SEC analysis. Presumable dimer and monomer peaks are indicated.Lower left, Presumable dimer fractions in SEC were collected and analyzed by mass photometry. The result confirms that the fraction contains considerable amount of dimer KIF1A.Lower right, Presumable monomer fractions were collected and analyzed by mass photometry. The result confirms that the fraction mainly consists of monomer KIF1A.Note that these results obtained from full-length KIF1A protein are similar to those of UNC-104(1-653) protein shown in Figure 5A-C.





