Pest Control: Hiding in plain smell
If you look carefully at a plant, you may start to notice a few telltale signs of feeding insects: holes chewed in a leaf, little mazes of trails, shiny spots of honeydew. You might even catch a caterpillar hiding along a leaf’s midvein – one of many strategies that plant-eating insects have evolved to camouflage themselves (Duncan, 1922). If you lean in, you will smell a planty scent, which herbivores use to choose the plants they eat (Bruce et al., 2005). At the same time, plants also release smells to attract species that prey on these herbivores (Turlings and Erb, 2018).
In fact, recent evidence suggests that plant odors are the subject of an ‘information arms race’, which plants seem to be winning so far. In this arms race, plants evolve new scents to become harder for herbivores to 'sniff out' in a crowd, while herbivores evolve to use more odors to find the plants they eat (Zu et al., 2020). In addition, plants also attract predators of herbivores, using smells that change depending on the herbivores feeding on the plant (Dicke and Baldwin, 2010). Together, these observations may explain why all plants studied so far produce rich, situation-dependent odor bouquets.
Now, in eLife, Yunhe Li from the Chinese Academy of Agricultural Sciences and colleagues from Switzerland and China – including Xiaoyun Hu as first author – report that a common crop pest can use the plant odors released by the feeding of another herbivore to hide from its own enemies (Figure 1; Hu et al., 2020). This strategy is known as ‘olfactory camouflage’ (Ruxton, 2009).
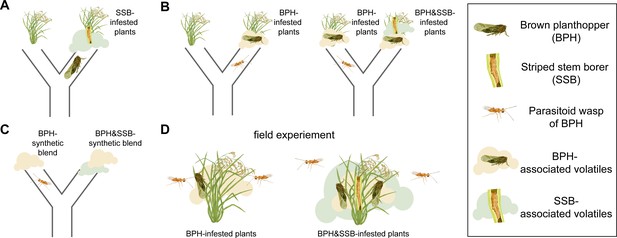
The level of infestation by striped stem borer caterpillars influences how brown planthoppers and the parasitoid wasp Anagrus nilaparvatae choose the rice plants on which to settle.
(A) Brown planthoppers placed in the middle of a tube between an intact plant and a plant infested with striped stem borer caterpillars (SSB) usually choose the infested plant. This choice is likely based on the smell that the plant releases when being eaten by the caterpillars. (B) The parasitoid wasp Anagrus nilaparvatae, which lays its eggs inside planthopper eggs, prefers plants infested with ten brown planthoppers (BPH) over those infested with just five (left). However, if a striped stem borer caterpillar is added to the plant with ten brown planthoppers, the wasp has no significant preference for either plant. If a second caterpillar is added to the plant with ten brown planthoppers, the wasp then prefers the plant with five brown planthoppers (right). (C) Wasps placed in the middle of a tube between two synthetic scent blends never preferred the blends that smelled like striped stem borer caterpillars. (D) The results were consistent both in the wild and in the glasshouse, with wasps always preferring plants with brown planthoppers that were not infested with striped stem borer caterpillars.
Hu et al. focused on two widespread rice pests: the striped stem borer caterpillar and the brown planthopper. Rice can make different blends of odors to attract animals that rid the plant of feeding herbivores. One such animal is a species of wasp called Anagrus nilaparvatae, which lays its eggs inside planthopper eggs. Hu et al. first observed that brown planthoppers preferred to lay their eggs on caterpillar-infested rice plants rather than undamaged plants (Figure 1A). Next, experiments were performed to test whether Anagrus nilaparvatae wasps chose the plant on which to lay their eggs based on the presence of caterpillars. The results showed that, in the absence of caterpillars, wasps preferred plants with more planthoppers. However, when the plants had both planthoppers and caterpillars, the wasps instead preferred plants with fewer caterpillars (Figure 1B). This indicates that one or more odors emitted by the caterpillar-infested plants were masking the presence of planthoppers.
To test this hypothesis, Hu et al. identified 20 odor compounds whose levels varied depending on the densities of planthoppers and caterpillars on the plants. These compounds were then used to test which odors the wasps preferred. Finally, to test whether odor alone was sufficient to explain the wasps’ choice, Hu et al. made synthetic blends of 13 odors that affected the wasps’ behavior. This experiment had exactly the same results as using infested plants: wasps did not choose any odor blends that smelled like plants eaten by caterpillars, with or without planthoppers (Figure 1C). In fact, both in the glasshouse and in the field, wasps parasitize a smaller share of eggs on caterpillar-infested plants, even when the larger number of eggs on those plants is accounted for (Figure 1D). This corresponds to wasps’ preference for the odors of plants hosting only planthoppers, and not caterpillars.
Hu et al.’s results suggest that planthoppers currently have the advantage in their information arms race with rice plants and wasps. How did this happen? The blend of odors that rice produces to encode ‘caterpillar’ appears to be more complex than the blend for ‘planthopper’, so when both are present, information about the planthoppers may be lost. However, the information arms race model indicates that rice plants should evolve a counter-strategy (Zu et al., 2020); and indeed, Hu et al. further showed that olfactory camouflage is less effective in wild rice than in cultivated plants. Unlike wild rice, cultivated rice is under artificial selection pressure by humans and is not free to respond to natural selection.
These results indicate that reducing striped stem borer caterpillar infestations in rice can yield additional benefits, as it may promote biological control of the brown planthopper by parasitoid wasps. From an evolutionary perspective, however, this is shortsighted: if efforts to reduce caterpillar populations fail, planthoppers will continue using the caterpillars as camouflage unless rice plants and wasps evolve ways to elude this mechanism. Unfortunately, waiting for rice plants to evolve a response would entail several generations of reduced rice yield – a disaster for our food supply. An alternative may be to artificially select plants with an advantage in this evolutionary arms race. Doing so will first require dissecting exactly how the planthopper’s olfactory camouflage works, and better understanding how plant odors direct interactions between species.
References
-
Insect host location: a volatile situationTrends in Plant Science 10:269–274.https://doi.org/10.1016/j.tplants.2005.04.003
-
The evolutionary context for herbivore-induced plant volatiles: beyond the 'cry for help'Trends in Plant Science 15:167–175.https://doi.org/10.1016/j.tplants.2009.12.002
-
Non-visual crypsis: a review of the empirical evidence for camouflage to senses other than visionPhilosophical Transactions of the Royal Society B: Biological Sciences 364:549–557.https://doi.org/10.1098/rstb.2008.0228
Article and author information
Author details
Publication history
Copyright
© 2020, Joo and Schuman
This article is distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use and redistribution provided that the original author and source are credited.
Metrics
-
- 1,338
- views
-
- 154
- downloads
-
- 0
- citations
Views, downloads and citations are aggregated across all versions of this paper published by eLife.
Download links
Downloads (link to download the article as PDF)
Open citations (links to open the citations from this article in various online reference manager services)
Cite this article (links to download the citations from this article in formats compatible with various reference manager tools)
Further reading
-
- Ecology
Global change is causing unprecedented degradation of the Earth’s biological systems and thus undermining human prosperity. Past practices have focused either on monitoring biodiversity decline or mitigating ecosystem services degradation. Missing, but critically needed, are management approaches that monitor and restore species interaction networks, thus bridging existing practices. Our overall aim here is to lay the foundations of a framework for developing network management, defined here as the study, monitoring, and management of species interaction networks. We review theory and empirical evidence demonstrating the importance of species interaction networks for the provisioning of ecosystem services, how human impacts on those networks lead to network rewiring that underlies ecosystem service degradation, and then turn to case studies showing how network management has effectively mitigated such effects or aided in network restoration. We also examine how emerging technologies for data acquisition and analysis are providing new opportunities for monitoring species interactions and discuss the opportunities and challenges of developing effective network management. In summary, we propose that network management provides key mechanistic knowledge on ecosystem degradation that links species- to ecosystem-level responses to global change, and that emerging technological tools offer the opportunity to accelerate its widespread adoption.
-
- Ecology
- Evolutionary Biology
Eurasia has undergone substantial tectonic, geological, and climatic changes throughout the Cenozoic, primarily associated with tectonic plate collisions and a global cooling trend. The evolution of present-day biodiversity unfolded in this dynamic environment, characterised by intricate interactions of abiotic factors. However, comprehensive, large-scale reconstructions illustrating the extent of these influences are lacking. We reconstructed the evolutionary history of the freshwater fish family Nemacheilidae across Eurasia and spanning most of the Cenozoic on the base of 471 specimens representing 279 species and 37 genera plus outgroup samples. Molecular phylogeny using six genes uncovered six major clades within the family, along with numerous unresolved taxonomic issues. Dating of cladogenetic events and ancestral range estimation traced the origin of Nemacheilidae to Indochina around 48 mya. Subsequently, one branch of Nemacheilidae colonised eastern, central, and northern Asia, as well as Europe, while another branch expanded into the Burmese region, the Indian subcontinent, the Near East, and northeast Africa. These expansions were facilitated by tectonic connections, favourable climatic conditions, and orogenic processes. Conversely, aridification emerged as the primary cause of extinction events. Our study marks the first comprehensive reconstruction of the evolution of Eurasian freshwater biodiversity on a continental scale and across deep geological time.






