Inflammatory response in hematopoietic stem and progenitor cells triggered by activating SHP2 mutations evokes blood defects
Figures
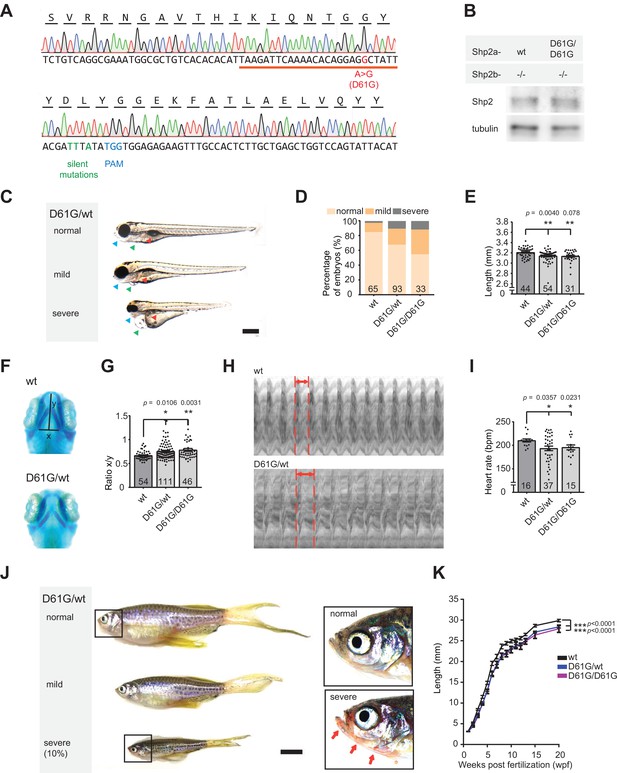
Shp2D61G zebrafish display Noonan syndrome (NS)-like traits.
(A) Sequencing trace derived from Shp2D61G/D61G zebrafish. Oligonucleotide sequence used to generate the model is underlined. Nucleotide substitutions for D61G mutation (red), silent mutations close to the PAM site (green), and the PAM site (blue) are indicated. (B) Immunoblot of Shp2 levels from five pooled Shp2awtShp2b-/- or Shp2aD61G/D61GShp2b-/- embryos using antibodies for Shp2 and tubulin (loading control) (Figure 1—source data 1). (C) Representative images of typical Shp2D61G zebrafish embryonic phenotypes at 5 days post fertilization (dpf). Blue arrows: jaw, green arrows: heart, red arrows: swim bladder. (D) Quantification of phenotypes of Shp2wt, Shp2D61G/wt, and Shp2D61G/D61G embryos scored as in (C) normal, mild, and severe. (E) Body axis length of Shp2wt, Shp2D61G/wt, and Shp2D61G/D61G embryos at 5 dpf. (F) Representative images of Alcian blue-stained head-cartilage of 4dpf Shp2wt and Shp2D61G/wt embryos (x) width of ceratohyal, (y) distance to Meckel’s cartilage. (G) Quantified craniofacial defects (x/y ratio). (H) Representative ventricular kymographs derived from high-speed video recordings of beating hearts of 5 dpf Shp2wt and Shp2D61G/wt embryos. Red dotted lines indicate one heart period. (I) Heart rates derived from the ventricular kymographs. (J) Representative images of typical Shp2D61G zebrafish adult phenotypes at 24 weeks post fertilization (wpf). Red arrows indicate skin redness in the jaw region. Scale bar, 0.5 cm. Insets, zoom-in of boxed regions. (K) Body axis lengths of 10 Shp2wt, 25 Shp2D61G/wt, and 10 Shp2D61G/D61G zebrafish measured weekly between 5 dpf and 20 wpf of age. (D,E,G,I) Measurements originate from three distinct experiments. Number on bars: number of embryos. (E,G,I,K) Error bars: standard error of the mean (SEM), *p < 0.05; **p < 0.01, ***p < 0.001, ANOVA complemented by Tukey’s HSD.
-
Figure 1—source data 1
Immunoblot raw data showing uncropped and unedited blots of Figure 1B.
- https://cdn.elifesciences.org/articles/73040/elife-73040-fig1-data1-v2.pdf
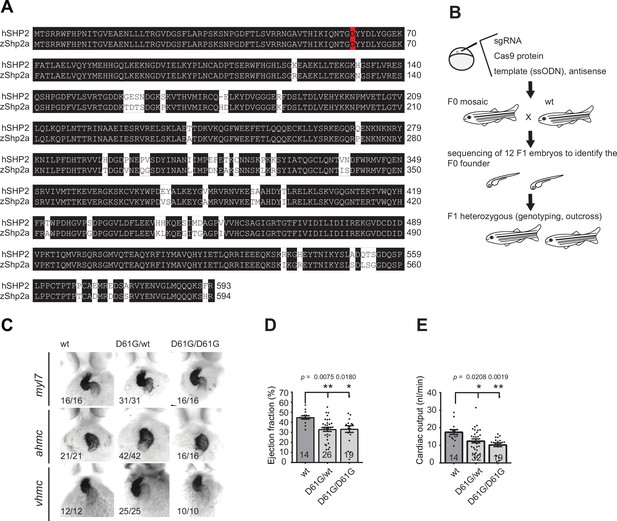
Defective heart function, but not heart morphology in Shp2D61G zebrafish embryos.
(A) Sequence alignment of human SHP2 and zebrafish Shp2a polypeptides performed by ClustalW. The residue D61 is highlighted in red, while the identical amino acids are highlighted in black. The amino acid number is marked to the right of each row. (B) Schematic representation of the procedure used to establish the Shp2D61G mutant line, as described in the Materials and methods section. (C) Representative images of the WISH staining for myl7, ahmc, and vhmc expression in the hearts of 3 days post fertilization (dpf) old Shp2wt, Shp2D61G/wt, and Shp2D61G/D61G zebrafish embryos. Numbers in the pictures indicate the number of embryos with the phenotype represented in the image. Ejection fraction (D) and cardiac output (E) determined from the high-speed videos of the heart of 5 dpf old Shp2wt, Shp2D61G/wt, and Shp2D61G/D61G zebrafish embryos. Measurements originate from at least three distinct experiments. Numbers on the bars depict the number of embryos. Error bars represent standard error of the mean (SEM).*p < 0.05; **p < 0.01, ***p < 0.001, NS, non-significant, ANOVA complemented by Tukey’s HSD.
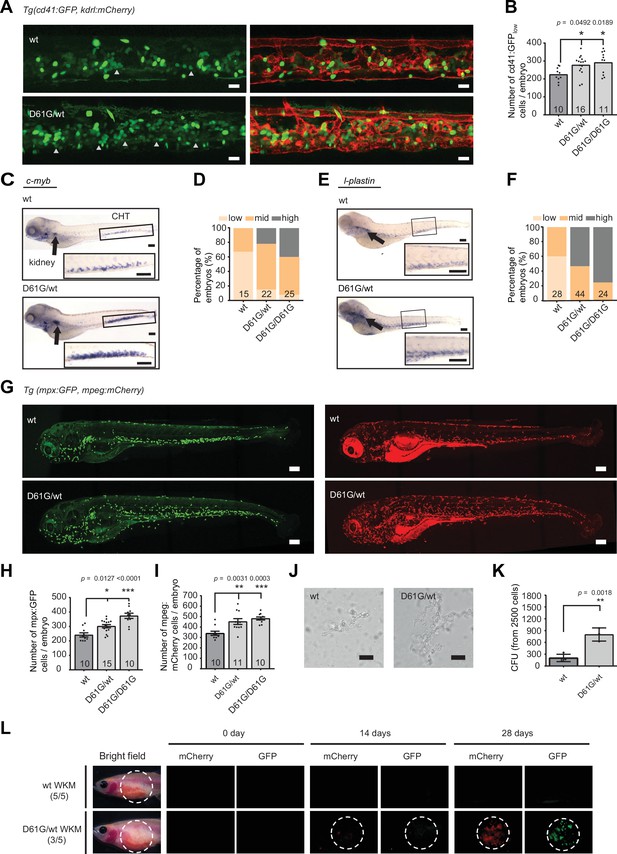
Shp2D61G mutant zebrafish embryos display juvenile myelomonocytic leukemia (JMML)-like myeloproliferative neoplasm (MPN).
(A) Representative images of the caudal hematopoietic tissue (CHT) region of Shp2wt and Shp2D61G zebrafish embryos in Tg(cd41:GFP, kdrl:mCherry-CAAX) background at 5 days post fertilization (dpf). cd41:GFPlow cells mark hematopoietic stem and progenitor cells (HSPCs) and cd41:GFPhigh cells thrombocytes. Gray arrow heads indicate cd41:GFPlow cells. Scale bar, 20 μm. (B) The low-intensity cd41:GFP-positive cells in the CHT region were counted. (C) Whole-mount in situ hybridization (WISH) of 5 dpf Shp2wt and Shp2D61G/wt embryos using c-myb-specific probe. Head kidney (arrow) and CHT (box) are indicated; zoom-in in inset. Scale bars, 150 μm. (D) Quantification of c-myb WISH. C-myb expression in Shp2wt, Shp2D61G/wt, and Shp2D61G/D61G embryos was scored as low, mid, and high. (E) WISH of 5 dpf Shp2wt and Shp2D61G/wt embryos using l-plastin-specific probe. Head kidney (arrow) and CHT (box) are indicated and zoom-in in inset. Scale bars, 150 μm. (F) Quantification of l-plastin expression in Shp2wt, Shp2D61G/wt, and Shp2D61G/D61G embryos scored as low, mid, and high. (G) Representative images of Shp2wt and Shp2D61G zebrafish embryos in Tg(mpx:GFP, mpeg:mCherry) background at 5 dpf. Mpx:GFP marks neutrophils and mpeg:mCherry macrophages. Scale bars, 150 μm. (H,I) Number of mpx:GFP and mpeg:mCherry-positive cells per embryo. (J) Representative images of colonies developed from cd41:GFPlow cells isolated from the CHT of 5 dpf Shp2wt and Shp2D61G/wt zebrafish embryos, grown in methylcellulose with zebrafish cytokine granulocyte colony stimulating factor a (Gcsfa) for 2 days. Scale bar, 50 μm. (K) Quantification of number of colonies from J, t-test. (L) WKM cells harvested from Shp2wt and Shp2D61G zebrafish in the Tg(mpx:GFP, mpeg:mCherry) background were injected into the peritoneum of adult prkdc-/- zebrafish. Recipients were monitored by fluorescence imaging. (B,D,F,H,I,K) Measurements originate from at least three distinct experiments. Number on bars: number of embryos. (B,H,I,K) Error bars represent SEM. *p < 0.05, **p < 0.01, ***p < 0.001. (B,H,I) ANOVA complemented by Tukey’s HSD.
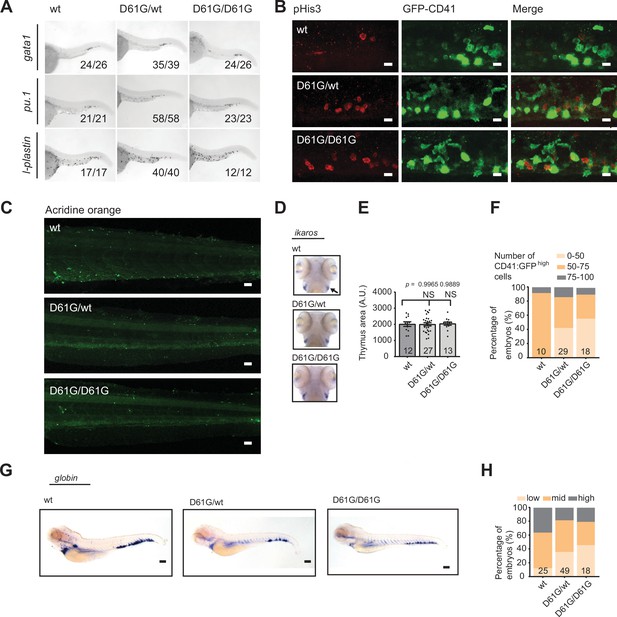
Hematopoiesis in Shp2D61G zebrafish.
(A) Representative images of the whole-mount in situ hybridization (WISH) staining for gata1, pu.1, and l-plastin expression in the tail region of 48 hr post fertilization (hpf) old Shp2wt, Shp2D61G/wt, and Shp2D61G/D61G zebrafish embryos. Scale bar, 250 µm. Numbers in the pictures indicate the number of embryos with the phenotype represented in the image. (B) Cell proliferation was assessed in the caudal hematopoietic tissue (CHT) region of 5 days post fertilization (dpf) old Shp2wt, Shp2D61G/wt, and Shp2D61G/D61G embryos in the Tg(cd41:GFP) background by immunohistochemistry using antibodies specific for phosphohistone H3 (pHis3) and GFP. Cells positive for both GFP and pHis3 are indicated with white arrows in the merge panel. Scale bar, 10 μm. (C) Representative images of the Acridine orange staining of the CHT region of the 5 dpf old Shp2wt, Shp2D61G/wt, and Shp2D61G/D61G embryos. Scale bar, 50 μm. (D) WISH of 5 dpf Shp2wt, Shp2D61G/wt, and Shp2D61G/D61G embryos using ikaros-specific probe. Thymus is indicated (arrow). (E) Size of ikaros-positive thymus, NS, non-significant, ANOVA complemented by Tukey’s HSD. (F) Number of cd41:GFPhigh cells in Shp2wt, Shp2D61G/wt, and Shp2D61G/D61G zebrafish embryos at 5 dpf were counted and percentage of embryos with either 0–50, 50–75, or 75–100 cd41:GFPhigh cells was plotted. (G) WISH of 5 dpf Shp2wt, Shp2D61G/wt, and Shp2D61G/D61G embryos using β-globin-specific probe. Scale bar, 150 μm. (H) Quantification of β-globin expression in Shp2wt, Shp2D61G/wt, and Shp2D61G/D61G embryos scored as low, mid, and high. (E,F,H) Measurements originate from at least three distinct experiments. Numbers on the bars depict the number of embryos.
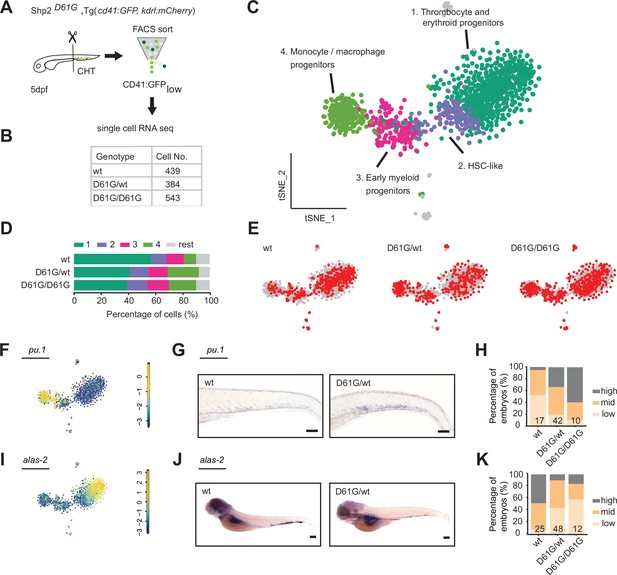
Single-cell RNA sequencing of hematopoietic stem and progenitor cells (HSPCs) reveals myeloid bias in Shp2D61G embryos.
(A) Schematic representation of the experimental procedure. At 5 days post fertilization (dpf), caudal hematopoietic tissues (CHTs) from Shp2wt, Shp2D61G/wt, and Shp2D61G/D61G embryos in Tg(cd41:GFP, kdrl:mCherry-CAAX) background were isolated. Cells were dissociated and separated by fluorescence-activated cell sorting (FACS), based on cd41:GFPlow expression, prior to single-cell RNA sequencing, as described in the Materials and methods section. (B) Number of cells of distinct genotypes used in single-cell RNA sequencing analysis. (C) Combined t-distributed stochastic neighbor embedding (t-SNE) map generated using the cells of all three genotypes (Shp2wt, Shp2D61G/wt, and Shp2D61G/D61G). Single cells from four major clusters are marked in dark green, blue, pink, and green, and their identities based on marker gene expression are indicated. Minor clusters are marked in gray. (D) Barplots showing the percentage of cells of Shp2wt, Shp2D61G/wt, and Shp2D61G/D61G genotype in distinct clusters. (E) Cells of distinct genotypes Shp2wt, Shp2D61G/wt, and Shp2D61G/D61G are visualized in t-distributed stochastic neighbor embedding (t-SNE) maps in red. (F) t-SNE maps showing log2-transformed read-counts of pu.1. (G) Representative images of the WISH staining for pu.1 expression in 5 dpf Shp2wt and Shp2D61G zebrafish embryos. Scale bar, 100 μm. (H) Expression of the pu.1 marker scored as low, mid, and high. (I) t-SNE maps showing log2-transformed read-counts of alas-2. (J) Representative images of the WISH staining for alas-2 expression in the tail region of 5 dpf Shp2wt and Shp2D61G zebrafish embryos. Scale bar, 100 μm. (K) Expression of the alas-2 marker scored as low, mid, and high. (H,K) Number on bars: number of embryos.
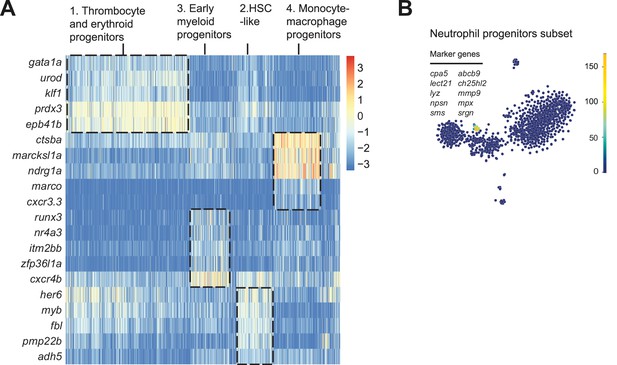
Identification of different hematopoietic stem and progenitor cells (HSPCs) subpopulations based on differential expression of representative genes.
(A) Heat map showing scaled expression (log TPM [transcripts per million] values) of representative genes (y-axis) in all cells (x-axis). Cells belonging to distinct clusters are squared with a dashed line and cluster identity is indicated on the top of the heat map. Cluster 1 is characterized by expression of genes characteristic for thrombocyte and erythroid differentiation, such as gata1a and klf1. Cells from cluster 2 express typical hematopoietic stem cell markers, such as c-myb and her6 (HES1 analogue in human). Cells from cluster 3 express early myeloid progenitor markers, such as runx3 and cxcr4b. Cells from cluster 4 express genes typical for both monocyte and macrophages, such as marco and ctsba. (B) t-Distributed stochastic neighbor embedding (t-SNE) maps showing the sum of total read-counts of selected neutrophil-specific genes.
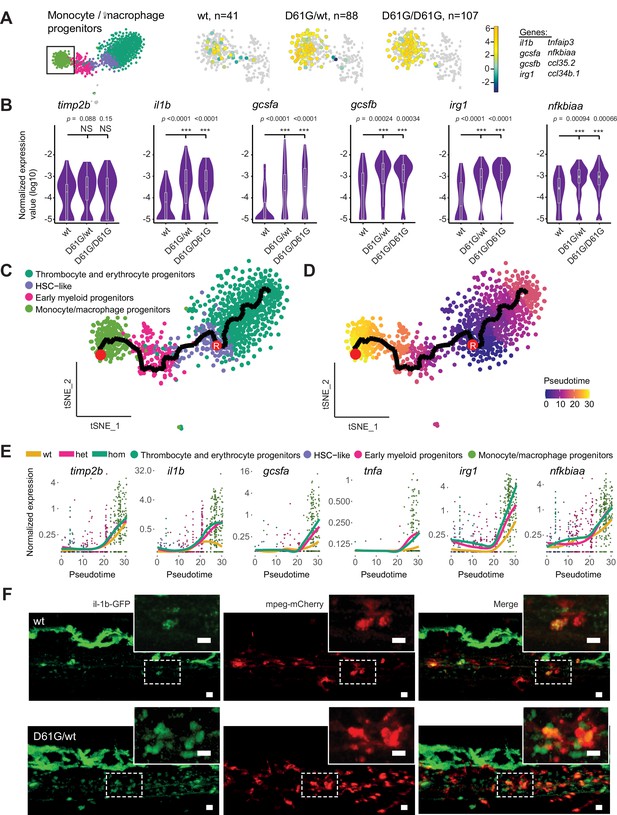
Inflammatory response in monocyte/macrophage progenitors in Shp2D61G embryos.
(A) The monocyte/macrophage progenitors cluster (boxed on the t-distributed stochastic neighbor embedding [t-SNE] map on left) was analyzed in detail. log2-transformed sum of read-counts of selected inflammation-related genes from the top 50 differentially expressed genes in the cells of the monocyte/macrophage progenitors cluster, with genotype and number of cells (n) indicated above. (B) Violin plots show the expression of specific genes in monocyte/macrophage progenitors of Shp2wt, Shp2D61G/wt, and Shp2D61G/D61G genotypes. NS, not significant, ***p < 0.001, t-test. (C,D) Monocle pseudotime trajectory of Shp2wt, Shp2D61G/wt, and Shp2D61G/D61G hematopoietic stem and progenitor cells (HSPCs) differentiation superimposed on the four clusters and indicated by color in D. A route leading from hematopoietic stem cell (HSC)-like cells to monocyte/macrophage progenitors was selected and its root (R) and end are marked with red circles. (E) Pseudotemporal expression dynamics of specific genes along the selected route. The lines represent the smoothened regression of the moving average for Shp2wt, Shp2D61G/wt, and Shp2D61G/D61G genotypes. (F) In vivo imaging of the caudal hematopoietic tissue (CHT) region of Shp2wt and Shp2D61G/wt zebrafish embryos in Tg(il1b:eGFP, mpeg:mCherry) background at 5 days post fertilization (dpf). Representative images are shown. The dashed line boxed region of CHT is zoom-in in inset. Scale bar, 10 μm.
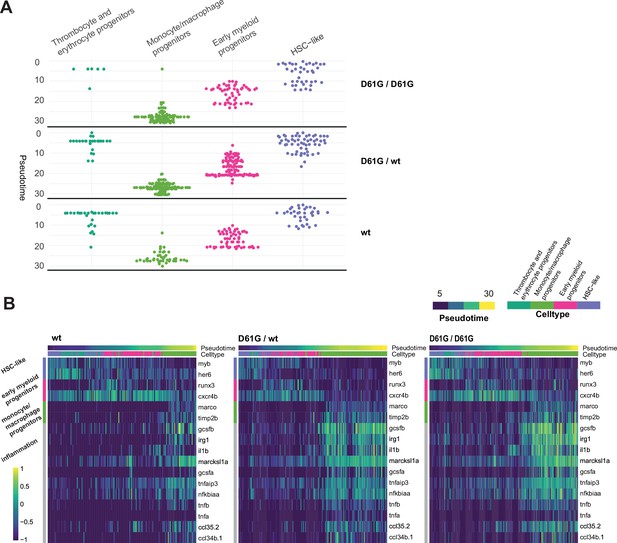
Distinct expression profiles of mutant zebrafish embryos in pseudotime.
(A) Identities of the four clusters of HSPCs of Shp2wt, Shp2D61G/wt and Shp2D61G/D61G genotypes from the trajectory in Figure 4, ordered in pseudotime. (B) Heat map showing scaled expression (log TPM [transcripts per million] values) of representative genes (y-axis) in cells of Shp2wt, Shp2D61G/wt, and Shp2D61G/D61G genotypes from the selected trajectory route, ordered along the pseudotime (x-axis). The cell type and pseudotime are annotated above the heat map.
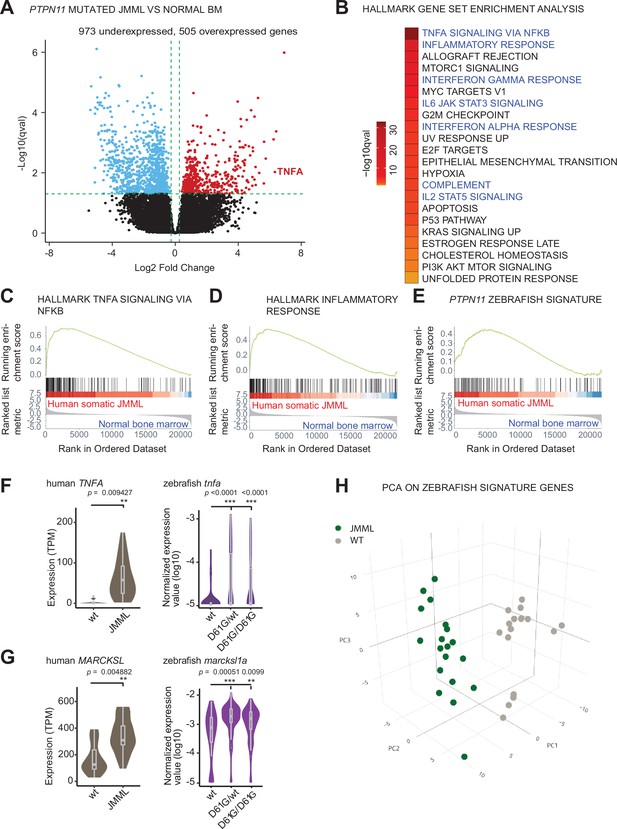
Similar molecular signatures in hematopoietic stem and progenitor cells (HSPCs) from human juvenile myelomonocytic leukemia (JMML) patients and zebrafish Shp2D61G embryos.
(A) Volcano plot of differentially expressed genes of HSPCs derived from bone marrow of JMML patients with PTPN11 mutations (n = 5) and healthy bone marrow (n = 7). Underexpressed genes are marked in blue and overexpressed genes are marked in red. TNFA expression is highlighted. Green dashed lines indicate the significance level. (B) Gene set enrichment analysis (GSEA) for the MSigDB’s hallmark gene sets in HSPCs from JMML compared to normal human age-matched bone marrow. GSEA plots for TNFA_SIGNALING_VIA_NFKB (C), INFLAMMATORY RESPONSE (D), and the custom zebrafish signature based on the top 100 human orthologous of genes upregulated in monocyte/macrophage progenitor cluster of zebrafish HSPCs (E). Violin plots show the expression of TNFA (F) and MARCKSL (G) in either human wild type (wt) vs. JMML HSPCs, and zebrafish Shp2wt vs. Shp2D61G/wt and Shp2D61G/D61G monocyte/macrophage HSPC progenitors. **p < 0.01, ***p < 0.001, t-test. (H) Principal component analysis (PCA) for the 100 genes included in the custom zebrafish signature. Green dots represent PTPN11 mutated JMML, gray ones represent normal human age-matched bone marrow. PC1: 19% of the variance, PC2: 16% of the variance, PC3: 14% of the variance.
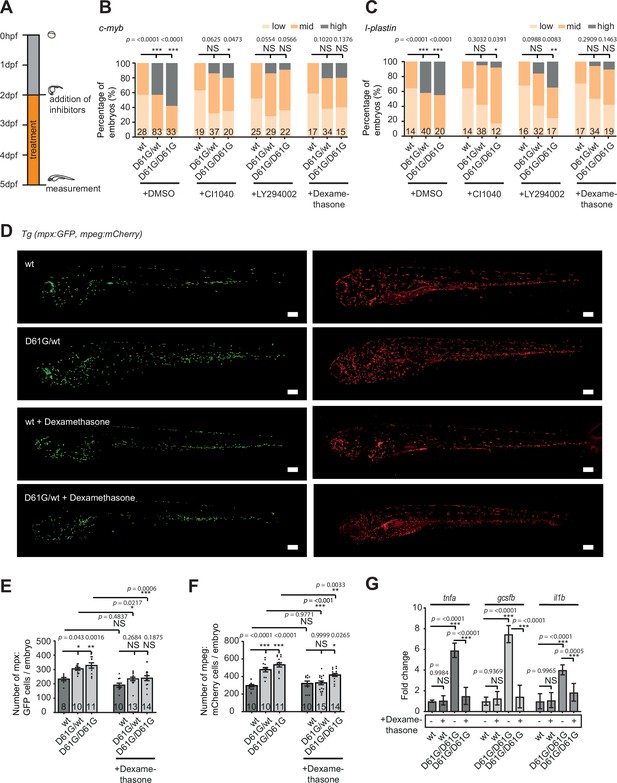
Anti-inflammatory treatment of zebrafish Shp2D61G embryos ameliorates the juvenile myelomonocytic leukemia (JMML)-like myeloproliferative neoplasm (MPN) phenotype.
(A) Schematic overview of the treatments with MEK inhibitor CI1040, PI3K inhibitor LY294002, and anti-inflammatory corticosteroid dexamethasone. Embryos were continuously treated from 48 hr post fertilization (hpf) until 5 days post fertilization (dpf), when either whole-mount in situ hybridization (WISH), confocal imaging, or quantitative reverse transcription PCR (RT-qPCR) was performed. (B,C) WISH staining for the expression of the c-myb and l-plastin markers was scored as low, mid, and high. Measurements originate from at least three distinct experiments. Number on bars: number of embryos. NS, not significant; *p < 0.05, **p < 0.01, ***p < 0.001, chi-squared test. (D) Representative images of 5 dpf Shp2wt and Shp2D61G zebrafish embryos in Tg(mpx:GFP, mpeg:mCherry) background without and with dexamethasone treatment. Scale bars, 150 μm. (E,F) Number of mpx:GFP and mpeg:mCherry-positive cells per embryo. Number on bars: number of embryos. Error bars represent SEM. (G) Expression of tnfa, gcsfb and il1b genes determined by RT-qPCR in Shp2wt and Shp2D61G/D61G zebrafish embryos without and with dexamethasone treatment at 5 dpf, normalized to ef1a expression. Standard deviation of four samples in duplicates for each condition are shown. (E,F,G) *p < 0.05, **p < 0.01, ***p < 0.001. ANOVA complemented by Tukey’s HSD.
Additional files
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/73040/elife-73040-transrepform1-v2.pdf
-
Supplementary file 1
List of differentially expressed genes (DE) and enriched gene ontology terms (GO) in each of the 4 major clusters of zebrafish HSPCs.
- https://cdn.elifesciences.org/articles/73040/elife-73040-supp1-v2.xlsx
-
Supplementary file 2
Characteristics of JMML PTPN11 mutated patients and healty subjects.
- https://cdn.elifesciences.org/articles/73040/elife-73040-supp2-v2.xlsx
-
Supplementary file 3
List of differentially expressed genes (DE) in JMML patient vs. normal bone marrow derived HSPCs.
- https://cdn.elifesciences.org/articles/73040/elife-73040-supp3-v2.xlsx
-
Supplementary file 4
GSEA analysis of differentially expressed genes in JMML patient derived HSPCs.
- https://cdn.elifesciences.org/articles/73040/elife-73040-supp4-v2.xlsx





